Chronic Conditions Dominate this Year’s List of Most Common Pet Health Problems
Nationwide analyzed more than 3.3 million pet insurance claims submitted in 2025 from over one million insured dogs and cats. This year’s analysis underscores a clear and ongoing trend: chronic diseases dominate the most common claims across both species, mirroring what many veterinary healthcare teams observe in practice. These conditions not only contribute to repeat visits, diagnostics, and multimodal management, but also can represent significant cumulative cost over time.
“Each year these findings remind us just how much our pets’ health is shaped by ongoing, chronic conditions,” said Dr. Emily Tincher, chief veterinary officer at Nationwide. “While not every illness can be prevented, early recognition and proactive management can improve outcomes. Our goal is to equip pet families, and the veterinary teams who partner with them, with insights that help support better long-term care.”
Canine Trends: Allergies top the list again and chronic issues dominate the top 10
- Skin allergies rank #1 again, for the 15th year running.
- No movement in the top 9 conditions, signaling persistent, widespread health patterns among U.S. canines
- Seizures enter the top 10 for the first time but are very closely followed by kidney disease and cranial cruciate ligament tears.
- Chronic conditions make up 6 of the top 10 and may require long-lasting care and follow families for years, underscoring the importance of ongoing care and budgeting.
Feline trends: Digestive issues lead the pack again with chronic conditions climbing
- For the third consecutive year, digestive issues stay firmly in the top spot.
- The top six conditions remain unchanged from last year, showing consistent health patterns across the feline population.
- A striking 7 out of 10 conditions for cats are chronic, meaning long-term costs can far exceed a single visit.
- The bottom half of the list sees some reshuffling among inflammatory bowel disease (#7 in 2024), diabetes (#8 in 2024), and respiratory infection (#9 in 2024).
USDA approes drug for canine allergic and atopic dermatitis
A newly approved injectable therapy expands treatment options for allergic and atopic dermatitis in dogs, with a U.S. launch planned in the first half of 2026.
January 6, 2026
Elanco Animal Health has received U.S. Department of Agriculture approval for Befrena (tirnovetmab), an anti–IL-31 monoclonal antibody injection for the treatment of canine allergic and atopic dermatitis. According to the company, the product is intended for use in dogs of any age and is recommended at dosing intervals of six to eight weeks following treatment. Elanco expects to launch the product in the first half of 2026.
The approval marks Elanco’s second dermatology-related product clearance in less than 18 months, following Zenrelia (ilunocitinib tablets), an oral JAK inhibitor indicated for itching and inflammation associated with canine skin allergies. Elanco noted that recent updates to the Zenrelia label removed a specific vaccine-related risk statement, while existing boxed warnings on vaccination timing remain in place.
Elanco also cited findings from its “America’s Itchy Dogs Report,” which highlights the prevalence of itch-related skin conditions in dogs and delays in seeking veterinary care.
_____________________________________________________________________________
The dental is in the details: CT scan in veterinary dentistry
January 7, 2026
By Robert Whitaker
The increase in accessibility and capabilities of computed tomography (CT) technology has understandably led to a growth in its use in the veterinary industry across a variety of applications. Dentistry is an area where CT imaging can be particularly useful, capturing small details to provide an intricate diagnostic resource.
CT in dentistry is likely to continue gaining popularity, and by reviewing the available technology, clinical applications and benefits to veterinarians, patients and owners, that trend makes perfect sense.
Technology with teeth
One of the driving factors in the increased presence of CT scanners in veterinary clinics is the recent addition of a new CT modality to the market. High-definition volumetric imaging (HDVI) builds a 2D and 3D image dataset of the animal patient’s whole density, rather than the traditional method of stacking individual image slices. This provides veterinarians with a much finer level of detail, capturing spatial resolution down to 100 microns within safe radiation levels.
Analyzing small and intricate anatomy is a well-known challenge in the veterinary industry, and dentistry is certainly no exception. An HDVI scan captures the detail necessary to diagnose conditions in tiny structures, like teeth roots in small animals, making it ideal for dental procedures, including:
- Tooth extraction planning. Identifying root fractures, ankylosis and abnormal root morphology, and locating retained roots after incomplete extraction.
- Periodontal disease assessment. Evaluating bone loss patterns around teeth and detecting early or hidden lesions not visible in an X-ray scan.
- Endodontics. Visualizing pulp chamber anatomy, root canals, and periapical lesions, and assessing healing after treatment.
- Oral tumor diagnosis and surgical planning.Determining tumor extent, invasion into the bone, and surgical margins, and mapping complex resections involving the mandible or maxilla.
- Jaw fracture repair. Precisely identifying fracture lines, fragment displacement, and dental involvement, and planning fixation without damaging the tooth roots.
- Orthodontics/malocclusion cases. Assessing unerupted or impacted teeth and measuring jaw symmetry and tooth alignment in 3D.
- Resorptive lesions.Detecting early lesions under the gumline and guiding decisions on extractions or crown amputations in this common feline condition.
- Pre- and post-surgical follow-up.Confirming full removal of diseased tissue and monitoring healing and bone regeneration.
As much of the damage from periodontal conditions happens below the surface of the gumline, it may be easy to forget just how big an impact it can have on veterinary patients. A study on dogs shows regular dental cleanings and treatments of periodontal disease are linked to patients living up to 20 percent longer compared to canine patients that did not receive dental care.1 This translates to an additional two to four years to the expected lifespan in some breeds.1 Cats with untreated dental disease are at a higher risk of kidney disease, heart problems and weight loss, conditions that can significantly shorten the animal’s life and increase pain and discomfort.2
This technology can also be applied to exotic patients.
“I’ve had some rabbits in the last year that had very vague signs, things like not eating very well. Looking at their teeth, their dental issues seemed mild, or their teeth looked fine on awake oral exams. I even did a sedated exam, and I didn’t see anything,” says Sari Kanfer, DVM, who practices at Exotic Animal Veterinary Center in Pasadena, Calif.
“Then on CT, I could see the last lower molar was a little infected and loose, so I was able to then put them under anesthesia, extract the tooth and the problems were fixed.”
Animals often hide dental pain, an instinct that can cause both owners and veterinarians to overlook potential issues that impact their quality of life. CT scans can help remove that barrier and provide a full assessment of the animal’s periodontal health, allowing earlier detection and intervention before the condition requires a major procedure.
More than just lip service
In addition to detailed imaging for these and other conditions in the mouth, an HDVI dental scan also captures the fine details of the ears, eyes, and nasal passage. Veterinarians can view the entire skull in 2D and 3D with clear resolution, including both hard and soft tissue details. This provides more context for the patient’s condition and can help further elevate care.
HDVI CT scans are also used for chest, abdominal, and orthopedic imaging in general practice clinics that provide dental services, creating more opportunities for the modality to be utilized and to establish its place in the imaging suite.
As CT technology has continued to evolve, it has also seen improvements in its usability in veterinary settings. Modern machines are significantly faster than previous iterations or other imaging systems used today for dental applications. Oral radiographs on a dental patient can take up to 30 minutes, while an HDVI scan can be completed in less than five minutes. This enables veterinarians to access more detailed diagnostic information more quickly, facilitating rapid assessment and treatment implementation.
The need for anesthesia, a hindrance that can occasionally impede veterinarians’ use of CT scanning, is also not an issue in dental care. Patients are already under anesthesia for the dental exam and cleaning, and a veterinarian can take advantage of this fact by capturing an HDVI scan to visualize everything below the gumline and other surrounding structures and tissues.
Lower cost has been another shift that increases accessibility to CT technology, giving veterinarians more information and power at the point of care. These savings are, in turn, passed on to the client, who can be assured their provider has the data they need to provide informed treatment without an impossible price tag attached to it.
Owners also benefit from the extra visibility provided by CT imaging, particularly HDVI CT. When a veterinarian can use an image to more clearly illustrate a pet’s condition to the owner, compliance with treatment recommendations can increase exponentially. Not only are HDVI scans high-resolution, capturing even the tiniest details, but they can also be rendered in 3D and colorized. The problem can be clearly visualized and explained, allowing the veterinarian to more easily obtain permission to complete the necessary treatment.
On the cuspid of greatness
The advancement of CT technology and its subsequent growth as a cornerstone in veterinary dentistry have already brought it into the spotlight as the gold standard for patient images, and this trend has every indication of continuing.
Ultrasound saw a similar trajectory over the past few decades. In the 1990s and early 2000s, only specialty clinics offered ultrasound machines; now, virtually all veterinary practices have an ultrasound system they use daily.
Dental CT is already a much more established clinic staple than it was just a few years ago. For example, one veterinary group with over 30 locations across the Western U.S. has made dental imaging with an HDVI unit a standard component of all new patient intakes, in addition to regularly using it for diagnosis on existing patients. This is a relatively new and niche feature to offer patients, but as technology continues to become more affordable with additional capabilities like pairing with surgical planning software, it will likely continue to be adopted into more clinics.
December of this year will mark 130 years since Wilhelm Röntgen took the first X-ray. In just a handful of decades, the capabilities of this technology have absolutely skyrocketed from blurry low-contrast pictures to interactive 3D models that accurately illustrate even the tiniest anatomical details. No one knows what the next innovation will be or how it will elevate the tools and information available to veterinarians. However, what we can say with some degree of certainty is that good things are coming in the realm of HDVI, and the next couple of decades will see an even greater improvement in accessibility, capability, client confidence, and patient outcomes.
With nearly three decades in the animal health industry, Robert Whitaker is an experienced animal imaging technician and veterinary business professional. Whitaker has made significant contributions to advancing imaging in food animal, large animal, and small animal applications since 1995. He is the business development director for Veterinary in North America at Imaginalis.
References
_____________________________________________________________________________
Kinesiology taping–does it work? If so, how?
January 20, 2026
By Narda G. Robinson, DO, DVM, MS, FAAMA
As the field of sports medicine and rehabilitation evolves and expands, more of us are recognizing the value of trying non-invasive modalities first, rather than rushing to surgery, for a range of orthopedic problems and mobility disorders.
One of the approaches currently gaining traction is kinesiology taping (KT), a hard-to-miss technique for humans and animals, with its bold colors and intriguing patterns. When audiences see KT worn by human athletes at the Olympics, it surges in popularity. Nonetheless, many skeptics dismiss it as “placebo”1 or “pseudoscience.”2 Fortunately, proponents remain undeterred, citing more and better research that elucidates its mechanisms of action and clinical value.
KT consists of thin, flexible, and “breathable” tape that stretches to allow for movement and, as a result, sensory stimulation. This differs from rigid athletic tape (AT) that limits motion and inhibits neuronal excitation.
For humans, KT attaches directly to skin, much like a Band-Aid. In contrast, veterinary kinesiology tape (VKT) is applied to areas that usually have some amount of fur between the skin and tape, making it harder to stay in place. Some manufacturers of VKT have addressed this concern by opting for thicker material with a stronger, medical-grade adhesive, offering more support and stability. To discourage chewing and ingestion, VKT may have a bitter or unpleasant flavor.
Taping patterns in swirling, crisscrossed, and circular arrangements suggest a rational and clinical intent designed to deliver a specific outcome. In the accompanying images, we see “anchored” Hestaband (a type of VKT) above and below longitudinal tapes over a dog’s tarsus
Research considerations
A common criticism of taping techniques pertains to the lack of consistent guidelines and basic standards.3 Determining “best practices” and testing those against other forms of care could dramatically improve the quality and reliability of KT research.
That said, it is nearly impossible to design an inert and convincing sham intervention for physical treatment modalities in the awake patient. That is, placebo surgery would typically require a skin incision. Massage, by definition, makes physical contact with the patient. Similarly, any form of “fake” acupuncture the patient feels impacts the nervous system.
As Kaptchuk et al. wrote, “[S]ham acupuncture is not inert or innocuous. And indeed, most methods of sham acupuncture have somatosensory properties and stimulate mechanoreceptors. Touching the skin anywhere induces a certain amount of sensory stimulation and thereby a possible therapeutic effect.”4
Uncertainties and methodological challenges aside, positive findings have emerged that support the premise and purported mechanisms of KT for a multitude of conditions:
- “Current evidence suggested that kinesiology taping could be recommended to improve upper limb function in patients with stroke in pain intensity, shoulder subluxation, general disability, upper extremity function, and the PROM of flexion.”5
- “We conclude that ankle balance taping that uses kinesiology tape instantly increased the walking ability of amateur soccer players with lateral ankle sprain. Therefore, ankle balance taping is a useful alternative to prevent and treat ankle sprain of soccer players.”6
- “The meta-analysis showed a significant improvement in gait functions (step velocity, step and stride length and reduction in the base of support in dynamics), reduction in the joint range of motion in inversion and eversion, decrease in the muscle activation of the long peroneus and decrease in the postural sway in movement in the mid-lateral direction. It is possible to conclude that KT provides a moderate stabilising (sic) effect on the ankles of the athletes of most popular contact sports with chronic ankle instability.”7
Other human conditions showing the value of KT include plantar fasciitis,8 hemiplegic shoulder subluxation,9 post-operative dental pain,10 quadriceps muscle strength,11 and low back pain.12 Patients may experience a variety of benefits following anterior cruciate ligament (ACL) reconstruction such as pain reduction,13 faster improvement of range of motion, resolution of edema, and increased thigh circumference.14 For patients with complete ACL rupture and no surgery, those receiving KT showed greater static stability,15 proprioception, strength,16 and functional performance.17
VKT studies are growing in number, too. For horses, the application of KT to the abdominal musculature increased longitudinal activity at the trot18 and reduced signs of thoracolumbar epaxial pain.19 VKT was superior to rigid taping in improving gait and weight transfer in dogs.20
As Christa Veinotte,21 the owner and CEO of Hestaband, noted, “Kinesiology taping has become an invaluable tool in veterinary rehabilitation— not just for performance, but for comfort and recovery. It was inevitable that taping would move from human athletics into animal therapy, but it’s rare to see it go the other way. That’s what makes Hestaband unique—we started in the equine and canine world, and now our work with Acadia University is taking those insights back into human research.”
Mechanisms of KT
Delving more into the mechanisms, we find that the benefits of KT fall into three main categories: facilitated blood22 and lymphatic fluid flow, mechanoreceptor-driven analgesia, and strengthened muscles and joints.
When sufficiently adherent to skin or fur, KT lifts the general “gmish” of what’s beneath the surface, giving more space for vessels, nerves, and lymphatic channels to function adequately. How does this help? Tissues traumatized by injury, surgery, and chronic inflammation may compress local vasculature due to concomitant edema, muscle tension, and fascial restriction. Circulation of blood and lymph slows, setting the stage for inadequate oxygenation, carbon dioxide buildup, tissue acidosis, and the detrimental accumulation of metabolic end-products.
Nerves affected by ongoing pressure and oxygen reduction turn to inefficient anaerobic glycolysis for day-to-day energy (ATP) needs. Longstanding energy deficits disrupt the function of sensory, motor, and autonomic nerves, leading to numbness, tingling, weakness, cramping, and thermoregulatory dysfunction. This is where KT comes in—its lifting aspects give nerves room to breathe, impelling fluids to flow and pain to subside. Moreover, when KT activates skin mechanoreceptors, it bolsters endogenous analgesic processes in the brain and spinal cord.
Unhappy nerves may also weaken muscles and destabilize joints. Muscles assist ligaments in preserving the mechanical integrity of arthrodial structures through balanced activation, which resists forces and excessive displacement.
By stimulating somatic afferent fibers in skin, muscles, and across joints, KT cultivates a more normal level of communication throughout the sensorimotor system.23 KT’s value in rehabilitation medicine is not about structurally supporting joints by limiting motion as athletic tape would do, but rather by physiologically bolstering innate processes that make motor control over joint integrity effective and meaningfully protective. Left uncorrected, impaired reflexes have less ability to prevent repeated microtrauma and, ultimately, joint destruction. Thus, KT’s value in the rehabilitation setting is less about limiting movement and more about bolstering innate injury-preventing processes.
From the larger perspective, with all the ways in which integrative rehabilitation and physical medicine measures—such as VKT—reduce pain, improve function, and support resolution of edema and circulatory compromise, why does our profession all too often fast-track animals to surgery when safer measures exist? The side effects, pain, cost, and trauma from VKT and other rehabilitation modalities are negligible in comparison. Plus, VKT has not been implicated in possibly raising the risk of osteosarcoma … unlike certain surgical procedures, such as the TPLO.24
Narda G. Robinson, DO, DVM, MS, FAAMA, practices osteopathic medicine and veterinary medicine. Dr. Robinson taught science-based integrative medicine at the Colorado State University College of Veterinary Medicine and Biomedical Sciences for 20 years. In 2016, Robinson established CuraCore VET in Fort Collins, Colo., where she teaches medical acupuncture, integrative rehabilitation, medical massage, and other integrative medical approaches. Dr. Robinson is planning to offer certification programs in Sidney, British Columbia, beginning in 2026. Columnists’ opinions do not necessarily reflect those of Veterinary Practice News.
References
- Luz Junior MA, Sousa MV, Neves LAFS, et al. Kinesio Taping® is not better than placebo in reducing pain and disability in patients with chronic non-specific low back pain: a randomized controlled trial. Braz J Phys Ther. 2015;19(6):482-490.
- Oakford GC. Elastic kinesiology tape is eye-catching, but is it effective? The Chronicle of the Horse. July 14 & 21, 2014. Accessed on 11-11-25 at https://thehorseinmotion.com/wp-content/uploads/2017/05/CHRONICLE-Equine-Kinesiology-Taping.pdf
- Andrýsková A, Lee JH. The Guidelines for Application of Kinesiology Tape for Prevention and Treatment of Sports Injuries. Healthcare (Basel). 2020 May 26;8(2):144.
- Kaptchuk TJ, Chen K-J, and Song J. Recent trials of acupuncture in the West: Responses from practitioners. Chin J Integr Med. 2010 Aug 8;16(3):197–203.
- Wang Y, Li X, Sun C, Xu R. Effectiveness of kinesiology taping on the functions of upper limbs in patients with stroke: a meta-analysis of randomized trial. Neurol Sci. 2022 Jul;43(7):4145-4156.
- Kim MK, Shin YJ. Immediate Effects of Ankle Balance Taping with Kinesiology Tape for Amateur Soccer Players with Lateral Ankle Sprain: A Randomized Cross-Over Design. Med Sci Monit. 2017 Nov 21;23:5534-5541.
- Biz C, Nicoletti P, Tomasin M, Bragazzi NL, Di Rubbo G, Ruggieri P. Is Kinesio Taping Effective for Sport Performance and Ankle Function of Athletes with Chronic Ankle Instability (CAI)? A Systematic Review and Meta-Analysis. Medicina (Kaunas). 2022 Apr 29;58(5):620.
- García-Gomariz C, García-Martínez MT, Alcahuz-Griñán M, Hernández-Guillén D, Blasco JM. Effects on pain of kinesiology tape in patients with plantar fasciitis: a randomized controlled study. Disabil Rehabil. 2024 Nov;46(23):5490-5496.
- Yim J, Kim B. Effectiveness of Shoulder Taping in Treating Hemiplegic Shoulder Subluxation: A Randomized Controlled Study of 35 Patients. Med Sci Monit. 2024 May 31;30:e944222.
- Zheng X, Luo S, Huang C, Wang Z, Lin X. Comparison of kinesiology tape and cryotherapy on postoperative reaction following third molar extraction: a randomized clinical trial. Quintessence Int. 2022 Sep 16;53(9):772-777.
- Choi IR, Lee JH. Effect of kinesiology tape application direction on quadriceps strength. Medicine (Baltimore). 2018 Jun;97(24):e11038.
- Abbasi S, Hadian Rasanani MR, Ghotbi N, Olyaei GR, Bozorgmehr A, Rasouli O. Short-term effect of kinesiology taping on pain, functional disability and lumbar proprioception in individuals with nonspecific chronic low back pain: a double-blinded, randomized trial. Chiropr Man Therap. 2020 Nov 20;28(1):63.
- Chan MC, Wee JW, Lim MH. Does Kinesiology Taping Improve the Early Postoperative Outcomes in Anterior Cruciate Ligament Reconstruction? A Randomized Controlled Study. Clin J Sport Med. 2017 May;27(3):260-265.
- Boguszewski D, Tomaszewska I, Adamczyk JG, Białoszewski D. Evaluation of effectiveness of kinesiology taping as an adjunct to rehabilitation following anterior cruciate ligament reconstruction. Preliminary report. Ortop Traumatol Rehabil. 2013 Oct 31;15(5):469-78.
- Ogrodzka-Ciechanowicz K, Głąb G, Ślusarski J, Gądek A, Nawara J. Does kinesiotaping can improve static stability of the knee after anterior cruciate ligament rupture? A randomized single-blind, placebo-controlled trial. BMC Sports Sci Med Rehabil. 2021 Mar 16;13(1):24.
- Kielė D, Solianik R. Four-Week Application of Kinesiotaping Improves Proprioception, Strength, and Balance in Individuals with Complete Anterior Cruciate Ligament Rupture. J Strength Cond Res. 2023 Jan 1;37(1):213-219.
- Liu K, Qian J, Gao Q, Ruan B. Effects of Kinesio taping of the knee on proprioception, balance, and functional performance in patients with anterior cruciate ligament rupture: A retrospective case series. Medicine (Baltimore). 2019 Nov;98(48):e17956.
- Biau S, Burgaud I. Application of kinesiology taping to equine abdominal musculature in a tension frame for muscle facilitation increases longitudinal activity at the trot. Equine Vet J. 2022 Sep;54(5):973-978.
- King MR, Pavsek H, Ellis KL, et al. J Equine Rehabilitation. Volume 2, 2024, 100007. Accessed on 11-12-25 at https://www.sciencedirect.com/science/article/pii/S2949905424000021?via%3Dihub .
- Altinkaya N, Pekyavas NO, and Gungor GC. Immediate effects of kinesiotape on gait and static weight-bearing in dog. J Hellenic Vet Med Soc. 2025;76 (1):8829-8836.
- Christa Veinotte is the founder of Hestaband, a Canadian-based kinesiology tape brand developed specifically for use in veterinary medicine. Veinotte continues to educate and collaborate with professionals worldwide to expand the understanding and practical application of taping in veterinary care.
- Craighead DH, Shank SW, Volz KM, Alexander LM. Kinesiology tape modestly increases skin blood flow regardless of tape application technique. J Perform Health Res. 2017.
- Riemann BL and Lephart SM. The sensorimotor system, Part I: The physiologic basis of functional joint stability. J Athl Train. 2002;37(1):71-79.
- S Selmic LE, Ryan SD, Ruple A, Pass WE, Withrow SJ. Association of tibial plateau leveling osteotomy with proximal tibial osteosarcoma in dogs. J Am Vet Med Assoc. 2018 Sep 15;253(6):752-756. doi: 10.2460/javma.253.6.752
_____________________________________________________________________________
Have You Heard of the Chevron Lucency?
February 3, 2026
By John R. Lewis, VMD, DAVDC, Fellow, AVDC OMFS
Interpreting dental radiographs in dogs and cats can be challenging, particularly when assessing teeth for possible endodontic disease. The classic radiographic signs of endodontic disease include: 1) a wider than normal root canal, 2) periapical resorption and/or internal resorption of the root and 3) lucency of the bone surrounding the root apex, a.k.a. “periapical lucency.”
Let’s discuss each of these radiographic signs individually:
1) A wide canal
A wider than normal root canal is a reliable and specific sign of an endodontically diseased tooth, but the sensitivity of this finding is low, because it takes a long time for an endodontically diseased tooth to show evidence of this radiographic sign.
Think of a tooth as a tree, except the rings of the tree are laid down internally. The diameter of the tooth does not change, but the width of the canal gets narrower over time as a vital, living tooth continues to age. The hard tissue, dentin, is produced throughout the life of the tooth by cells lining the pulp called odontoblasts. Odontoblasts produce primary dentin when the tooth is developing. Secondary dentin is produced after eruption and throughout the life of the tooth. Odontoblasts can even produce a reparative dentin (tertiary dentin) in response to wear of the tooth. If the cells within the pulp die due to endodontic disease, the lack of odontoblasts results in a cessation of maturation of the tooth and a wider canal than the contralateral tooth. It takes months for this discrepancy in canal width to be radiographically evident.
2) Tooth resorption
Resorption of the hard tissue of the root can occur in the area of the apical delta or anywhere within the canal as a result of infection and inflammation. When it occurs within the canal, internal resorption manifests radiographically as a focal widening of the canal, often in the mid-root area. When resorption occurs around the apex of the root, it results in an irregular surface of the normally smooth, dense hard tissue of the tooth root.
3) Periapical lucency
Periapical lucency can be evidence of periapical infection/inflammation. Normally, the apex of the root is surrounded by a narrow periodontal ligament space, which is radiolucent on radiographs. Adjacent to the periodontal ligament space, there is a thin layer of radiodense bone called lamina dura. When infection becomes established in the apical portion of the root, the bone of the lamina dura is lost, resulting in a periapical lucency that is often wider than the diameter of the tip of the root. Though this appearance is often referred to as a “tooth root abscess,” in reality, abscess is only one of the many possible reasons for a periapical lucency. Perhaps the most common cause of periapical lucency is a granuloma rather than a true abscess. Granulomas can sometimes be seen as a large ball of soft tissue attached to the root when extracting an endodontically diseased tooth. Periapical cysts can also cause a periapical lucency, but unlike abscesses or granulomas, a cyst often has a corticated border around the periapical lucency.
One of the most commonly mistaken normal variants that can mimic a pathologic periapical lucency is the chevron radiolucency. Frequently seen at the apices of canine teeth, molars and maxillary incisors of dogs, the chevron lucency represents trabecular bone and vascular channels rather than true periapical lysis.1
Other normal jaw structures may appear on radiographs and mimic pathology. In the maxillary canine and premolar region in dogs, the junction of the vertical body of the maxilla with its palatine process and the maxillary conchal crest—can mimic a cystic structure and interfere with evaluating apical structures. In the mandible, the canal and various mental foramina are normal radiolucent structures that may superimpose over a root, depending on the angle and exposure.
Distinguishing normal variants from true endodontic disease
How can we determine if a lucency is pathologic or anatomic? Sometimes, even for board-certified dentists who interpret dental radiographs daily, it can be difficult to determine.
Chevron lucencies usually extend apically at the same width of the apex instead of expanding beyond the dimensions of the apex, and are surrounded by a distinct periodontal ligament and lamina dura.
Normal anatomical variations often present with regular, well-defined margins. Pathologic changes, by contrast, tend to be mottled, irregular, or “moth-eaten” in character. Normal variants are typically bilaterally symmetrical or follow predictable patterns related to jaw or tooth anatomy. Disease processes usually involve asymmetric destruction or expansion of bone or tooth structure.
Clinical clues matter. When trying to determine if radiographic findings are truly significant, evaluate for signs such as tooth abrasion, discoloration (intrinsic staining), fracture, pulp exposure, swelling over the roots, and draining tracts. Normal anatomical variants often occur in teeth that have no other signs of trauma. If there are no clinical signs of disease, consider monitoring the tooth with serial radiographs to look for changes over time, which will distinguish stable anatomical variants from progressive pathologies.
I hope this helps you with some of your future radiographic challenges. In next month’s column, we will continue to discuss diagnostic imaging modalities of the mouth, head, and neck.
John R. Lewis, VMD, DAVDC, FF-OMFS practices at Veterinary Dentistry Specialists and teaches at Silo Academy Education Center, both in Chadds Ford, Pa.
Reference
- DuPont GA, DeBowes LJ. Atlas of Dental Radiography in Dogs and Cats. Saunders Elsevier; St. Louis (2009): p. 150.
_____________________________________________________________________________
Veterinary Practice News I Geriatric pain: A look at common considerations for pain management in senior pets
Let’s break down pharmaceutical and non-pharmaceutical options when dealing with pain in apparently healthy geriatric patients.
By Michael C. Petty, DVM
January 8, 2026
In general terms, geriatric patients are defined as those that have completed 75-80 percent of their anticipated life span.1 In humans, geriatric comorbidities are the main cause of mortality during anesthesia.2 There is no reason to think it is any different in our dogs and cats. The second largest reason is that in most geriatric patients, there is a decline in the normal “reserve capacity” which can impede their response to situations of stress, such as hospitalization and surgery.3
Not all dogs and cats have issues with major organ systems, but those of high consideration are the cardiovascular system, major organs, such as the liver and kidneys, and the respiratory system. Additionally, for unknown reasons, older patients require less injectable and inhalant anesthetic drugs to produce general anesthesia.4
The following is a list of common considerations when dealing with apparently healthy geriatric patients. It is not intended to serve as a guide for geriatric patients with significant or profound comorbidities. Additionally, this writing concentrates on pain management, which also overlaps with anesthetic protocols. It is not meant to be a guide for induction or maintenance of anesthesia.
Pharmaceutical considerations: Acute pain Opioids are amazing pain drugs when used in acute pain settings, such as pre- and post-surgery, and in the case of trauma. Opioids are generally the safest and most effective agents available for the treatment of acute pain. Although many of us were taught to fear the serious side effects of opioids, these concerns were inappropriately extrapolated from experiences in human medicine.5 Opioids work on several levels, including the inhibition of pain at peripheral nociceptors, modulation of the pain signal in the spinal pathway, and reduced perception of pain in the brain. Modification of opioid doses for geriatric animals is not needed.
Opioids can be classified into three types:
- Full agonists. These opioids produce dose-dependent analgesia—that is, the more administered (within safety limits), the greater the effect. They bind and fully activate opioid receptors, particularly the μ receptor. Clinical application: Used for procedures that have the potential for causing severe pain, including perioperative and trauma pain.
- Partial agonists. Partial agonists bind to opioid receptors with high affinity but produce a submaximal effect, even at higher doses. This ceiling effect limits both their analgesic potential and their potential side effects. Clinical application: Appropriate for mild to moderate pain.
- Agonist-antagonists. These drugs act as agonists at some opioid receptors (typically κ) and antagonists at others (typically μ). This class of opioids rarely causes dysphoria. They may displace full agonists from the μ receptor, potentially reversing their effects. Clinical application: Used for mild to moderate pain. They are useful for opioid reversal when dysphoria occurs with full agonists but still allow partial maintenance of analgesia.
Morphine is the prototypical opioid against which other opioids are measured. It is extremely safe, with minimal cardiovascular and respiratory effects. As a full agonist, it is reversible. Morphine can cause a decrease in GI motility, which should not be of clinical importance when used short-term, such as for surgery. It also causes vomiting, especially in those patients that receive it prior to surgery.
Morphine can be given subcutaneously, intramuscularly, or very slowly intravenously to avoid histamine release. It has a rapid onset of action, occurring within three to five minutes, and provides analgesia for two to four hours.
Note: Morphine is potentially less effective in cats than it is in dogs, as cats only benefit from the parent drug but cannot make the intermediate metabolite.
Hydromorphone It is very similar to morphine in terms of onset and duration. All opioids can cause hyperthermia; however, it has been implicated, maybe wrongly, to be more severe in cats. One study showed all opioids tested caused a mild increase in body temperature.6 This is usually a self-limiting issue and may also be more closely associated with post-emergent dysphoria following sedation or general anesthesia.
Methadone Very similar to morphine but also has some N-methyl-D-aspartate (NMDA) antagonist activity. Methadone causes little nausea and is less likely to cause dysphoria compared to other full agonists.
Fentanyl A very potent opioid given intravenously as either a constant rate infusion or as a bolus. It has a rapid onset of action, typically occurring within two to five minutes. It is less likely to cause GI effects or dysphoria compared to the other full agonists.
Buprenorphine This has moderate potency compared to the full agonists, so consideration must be given for the surgical procedure to be performed. It has a long onset of action, as much as 30 minutes, but also has a long duration of action, typically in the six- to eight-hour range. Given to cats as the concentrated form of Simbadol, it can provide analgesia for 24 hours.
Butorphanol This agonist-antagonist is moderately potent, with an onset of action of one to five minutes when administered IV or IM. However, it has a short duration of action, ranging from 45 minutes. It has an excellent sedative effect, which can last several hours. This sedative effect is often confused with the much shorter analgesic effect.
Alpha-2 Agonists Alpha-2 agonists—primarily dexmedetomidine, medetomidine, and xylazine—are sedative-analgesic agents commonly used in veterinary practice. Their mechanism of action involves stimulation of central α₂-adrenoceptors, resulting in sedation, analgesia, and muscle relaxation, as well as dose-sparing effects on other anesthetic agents.
Dexmedetomidine is the most commonly used alpha-2 agonist due to its higher receptor selectivity and favorable pharmacodynamic profile.
Clinical use of alpha-2 agonists:
As premedication in balanced anesthesia protocols For sedation when combined with other agents (e.g. opioids, ketamine) Occasionally, as part of multimodal analgesia during painful procedures or in recovery. Note: Atipamezole is a specific antagonist and should always be available when using this class of drugs When combined with opioids, the recommended dose of dexmedetomidine is 0.5–2 µg/kg IM. Always dose based on lean body mass, not total weight. Special considerations when using alpha-2 in geriatric or compromised patients: Due to age-related reductions in cardiac output, vascular compliance, and organ perfusion, alpha-2 agonists must be used with extreme caution in geriatric animals. Avoid their use as a stand-alone sedative in older patients. Instead, consider combining with opioids for pre-anesthesia or combining with ketamine and opioids for procedural sedation.
The disadvantages of using alpha-2 agonists include emesis, muscle twitching and tremors, reflexive bradycardia due to increased central blood pressure, and reduced cardiac output.
Ketamine Ketamine is a dissociative anesthetic widely used in veterinary medicine for induction, sedation, and analgesia. It functions primarily as an NMDA receptor antagonist, disrupting nociceptive signal transmission and preventing central sensitization. Ketamine is most effective when combined with other agents, such as opioids and alpha-2 agonists, particularly in multimodal analgesia protocols.
Ketamine provides excellent somatic analgesia. However, it has poor visceral analgesia, making it a poor choice for intra-abdominal procedures. It also has the advantage of a rapid onset and a long duration of action. Its ability to maintain cardiovascular and respiratory function makes it a potential option for geriatric patients that do not have significant cardiovascular or respiratory disease.
There are some disadvantages with ketamine, including hypersalivation, increased muscle tone resulting in a rigid appearance, lack of a blink response requiring ophthalmic lubrication, and rough recoveries when used as a sole agent. Patients may experience vocalization, tremors, or dysphoria.
It is advisable not to administer NSAIDs preoperatively unless there are provisions for closely monitoring for a drop in blood pressure, and the attending veterinary staff have the necessary pharmaceuticals and skill set to respond appropriately to a blood pressure drop. Many veterinarians prefer to start NSAIDs during the immediate post-anesthetic recovery.
Local anesthetics Local anesthetics can and should be used whenever possible. As the only true analgesic, it can have a profound effect on patient comfort, as well as safety, as the use of local anesthetics will reduce the need and dosage of other analgesics. There are no contraindications specific to geriatric patients. The usual precautions of dose and route of administration are the same across all age groups.
Non-pharmaceutical considerations Geriatric pets may become disoriented in the unfamiliar surroundings of a veterinary hospital. This can interfere with both eating and sleeping, which have been shown to impact recovery in all age groups, but is decidedly a more important consideration in geriatrics that may have comorbidities. It is essential that we consider the factors that impact sleep7 and nutrition.8
Michael C. Petty, DVM, is a veterinary school graduate from Michigan State University. Dr. Petty has devoted his professional life to the care and well-being of animals, especially in pain management. Petty is the past president of the International Veterinary Academy of Pain Management and current World Small Animal Association Pain Council member. A frequent speaker and consultant, he has published articles in veterinary journals and serves in an advisory capacity to several pharmaceutical companies on pain management topics. Petty has been the investigator/veterinarian in 12 FDA pilot and pivotal studies for pain management products. He has lectured both nationally and internationally on pain management topics.
References
- Dodman NH, Seeler DC, Court MH. Aging changes in the geriatric dog and their impact on anesthesia. Compendium on Continuing Education for the Practicing Veterinarian. 1984;6:1106–1113.
- Muravchick S. The aging process: anesthetic implications. Acta anaesthesiologica Belgica. 1998;49:85–90.
- Conzen P, Peter K. Inhalation anaesthesia at the extremes of age: geriatric anaesthesia. Anaesthesia. 1995;50(Suppl):29–33. doi: 10.1111/j.1365-2044.1995.tb06187.x.
- Hughes J. Anaesthesia for the geriatric dog and cat. Ir Vet J. 2008 Jun 1;61(6):380-7
- Hansen B. Acute Pain Management. Veterinary Clinics of NA, Small Animal. K Mathews, ed. 2000;4:899‐916.
- Posner LP, et al. Vet Anaesth Analg. 2010;37(1):35-43.
- Emma A. Devereux, Alana V. Ejezie, Alex M. Lynch, Margaret E. Gruen, Stefanie J. LaJuett, James B. Robertson, Valery F. Scharf Factors Affecting Sleep Among Dogs and Cats in a Veterinary Intensive Care Unit, Journal of Veterinary Emergency and Critical Care: 2025;Volume 35, Issue 3:252-232
- Collins, S. (2016). The importance of nutrition in the post-operative recovery of cats and dogs. Veterinary Nursing Journal, 31(8), 233–236. https://doi.org/10.1080/17415349.2016.1194637
_____________________________________________________________________________
DVM 360 News | Key takeaways from AAHA’s 2026 oncology guidelines
Author(s)Bob Alaburda, Associate Editorial Director
January 7, 2026
AAHA’s 2026 oncology guidelines aim to update diagnostics, staging, treatment priorities, and team workflows to help primary care clinics detect, stage, and manage cancer while preserving patient quality of life.
Cancer is a leading cause of illness and death in older dogs and cats, affecting roughly 50% of dogs and about 30% of cats older than 10 years. The American Animal Hospital Association’s (AAHA) 2026 Oncology Guidelines synthesize current evidence and expert consensus into actionable recommendations for the primary care team from diagnosis and staging to treatment options, supportive care, referral triggers, and follow-up.1,2
Here are a few key takeaways. A link to the full guidelines can be found in the reference list below this article.
Make a definitive diagnosis before planning therapy
AAHA emphasizes that a cytologic or histopathologic diagnosis is necessary to determine prognosis and guide therapy.1-3 When fine-needle aspirate is inconclusive or additional information is required (for example, tumor grade), pursue biopsy and histopathology rather than assuming tumor behavior.3
Use staging to guide treatment intent and timing
Staging is more than local vs metastatic classification. It clarifies treatment intent (curative vs palliative) and identifies appropriate imaging and nodal assessment. AAHA provides tumor-specific staging checklists and recommends thoracic imaging, abdominal ultrasound, and regional lymph node cytology or biopsy as indicated.3
Frame chemotherapy around quality of life
The primary goal of systemic therapy in veterinary oncology is maintaining the best possible quality of life while controlling disease. Most veterinary chemotherapy protocols are generally well tolerated. Adverse events are usually manageable when clinicians and owners anticipate them and monitor appropriately.1,4
Educate owners proactively about the likelihood of appetite changes during chemotherapy and provide concrete management strategies. Addressing appetite loss early helps pets remain on recommended treatment plans and improves overall well-being during therapy.1
Evaluate new drugs and biologics carefully
The guidelines stress understanding the risks, benefits, cost, and regulatory status of novel therapeutics (such as small-molecule inhibitors, monoclonal antibodies, and other biologics). Clinicians should not adopt new agents without reviewing available evidence, registration status, and monitoring requirements.4
Integrate supportive and symptomatic care from diagnosis onward
Supportive care—nutrition, pain management, antiemetics, wound and infection management, and psychosocial support for owners—is fundamental and begins at diagnosis. AAHA provides screening and treatment checklists aimed at maintaining function and comfort throughout therapy and survivorship.7,8 When initiating symptomatic care, thoroughly evaluate for comorbidities that may alter drug choice, dosing, or prognosis. This ensures supportive measures truly improve quality of life rather than introducing harm.1
Optimize the veterinary team’s role
Technicians and other team members play a central role in oncology workflows, including intake screening, weight and appetite tracking, pain scoring, client education, medication administration training, and documentation. Standardized technician checkpoints improve early detection of complications and support better quality-of-life decisions.8
Know when to refer and how to collaborate
AAHA outlines clear referral triggers: complex staging that requires advanced imaging, specialized surgery, administration of advanced systemic protocols, or when weighing curative versus palliative intent. Good referral relationships and succinct transfer-of-care notes streamline transitions and may improve outcomes.5,6
Adopt structured post-treatment monitoring
Tumor-specific follow-up schedules and standardized monitoring (physical exam, imaging, and laboratory testing) are recommended to detect recurrence or late treatment effects early. Integrating these schedules into practice management software reduces the risk of patients being lost to follow-up.9
References
- American Animal Hospital Association. 2026 AAHA oncology guidelines for dogs and cats. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/
- American Animal Hospital Association. Section 1: overview of common cancers. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/section-1-overview-of-common-cancers/
- American Animal Hospital Association. Section 3: tumor diagnostics and staging. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/section-3-tumor-diagnostics-staging/
- American Animal Hospital Association. Section 5: therapeutic interventions. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/section-5-therapeutic-interventions/
- American Animal Hospital Association. Section 6: consultations and referrals. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/section-6-consultations-and-referrals/
- American Animal Hospital Association. Section 2: what’s new in veterinary oncology. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/section-2-whats-new-in-veterinary-oncology/
- American Animal Hospital Association. Section 7: supportive and symptomatic care. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/section-7-supportive-and-symptomatic-care/
- American Animal Hospital Association. Section 8: technician and team optimization. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/section-8-technician-and-team-optimization/
- American Animal Hospital Association. Section 9: post-treatment monitoring and follow-up care. American Animal Hospital Association. Published January 1, 2026. Accessed January 7, 2026. https://www.aaha.org/resources/2026-aaha-oncology-guidelines-for-dogs-and-cats/section-9-post-treatment-monitoring-and-follow-up-care/
https://www.dvm360.com/view/key-takeaways-from-aaha-s-2026-oncology-guidelines
_____________________________________________________________________________
DVM360 | USDA approves new Anti-IL31 monoclonal antibody injection
Author(s)Caitlin McCafferty, Editor
January 2, 2026
On December 31, 2025, Elanco Animal Health received approval from the USDA for tirnovetmab (Befrena; Elanco Animal Health), an anti-IL31 monoclonal antibody injection that targets canine allergic and atopic dermatitis. The new product Befrena is recommended in dosing intervals of 6-8 weeks post treatment compared to its competitor that is currently on the market that recommends 4-8 weeks post treatment.
“The animal health sector is increasingly focused on antibody-based therapies, and today’s approval is an important step forward in delivering high-impact innovation that enhances the quality of life for pets,” said Ellen de Brabander, PhD, executive vice president of research & development at Elanco, in an organizational release.1 “We are pleased to offer veterinarians and pet owners Befrena, which delivers long lasting relief to treat dogs with allergic itch.”
According to the release, Befrena is the second dermatology product from Elanco to receive approval in less than 18 months and now joins Zenrelia in its dermatology portfolio. Befrena will launch in the first half of 2026.1
In 2025, Elanco released its first report highlighting the impact itching has on canine patients, clients, and veterinary teams in its America’s Itchy Dogs Report. The report, made up of findings from multiple surveys of pet owners and veterinarians, revealed that 9 out of 10 dogs in the US are ‘itchy dogs’ and have experienced symptoms of itchiness at some point in the year according to their owners, which is nearly 79 million dogs.2 The study also found that clients spend around $400 on over-the-counter remedies and wait 6 weeks before bringing their pet to their veterinarian because they typically try to find solutions at home before seeking veterinary care. Because of this, pets whose owners waited too long to bring them in resulted in raw and infected skin as well as owners who are irritated because they want their pet to have some relief.3,4
Research coming out of Elanco, according to their release, stated that nearly 70% of veterinarians would be willing to stock another dermatology product in their clinic to help more patients find itch relief.
“Veterinarians need more options for itch relief so they can offer targeted therapy that focuses on mode of action to meet the unique needs of the individual patient to deliver fast, effective and valuable itch relief,” said Joya Griffin, DVM, DACVD, veterinary dermatologist. “USDA approval of Befrena brings veterinarians a step closer to having another beneficial and safe treatment option. I’m excited to collaborate with Elanco and plan to offer this product in my practice when it becomes available.”
References
- Elanco Receives USDA Approval for Befrena (tirnovetmab), a New Anti-IL31 Monoclonal Antibody Injection Targeting Canine Allergic and Atopic Dermatitis. News release. Elanco Animal Health. December 31, 2025. Accessed January 2, 2026. https://www.elanco.com/us/newsroom/press-releases/befrena-usda-approval
- Elanco Animal Health. America’s Itchy Dogs. Accessed January 2, 2026. https://assets.elanco.com/0cec44ed-3eaa-0009-2029-666567e7e4de/33474a48-bac3-4fc4-b7fc-3589b88560fb/ElancoItchyDogsReportFINAL.pdf
- Elanco Animal Health. Data on File.
- Elanco Animal Health. Data on File.
https://www.dvm360.com/view/usda-approves-new-anti-il31-monoclonal-antibody-injection
_____________________________________________________________________________
AVMA News I Wisconsin dog breeder, facing allegations of animal abuse, will no longer supply dogs for research
Published on December 31, 2025
Ridglan Farms, the nation’s second-largest research dog breeder, will stop supplying animals to laboratories under a settlement to avoid criminal prosecution, according to media reports.
This comes as the Beagle breeder, based in Blue Mounds, Wisconsin, faces allegations of animal abuse.
The Dane County Board of Supervisors in October adopted Resolution 119 calling on the Wisconsin Department of Agriculture, Trade, and Consumer Protection (DATCP) to revoke Ridglan’s state-issued commercial dog breeder license.
A judge has also upheld the Wisconsin Veterinary Examining Board’s emergency suspension of Dr. Rick Van Domelen, the lead veterinarian and facility manager at Ridglan Farms.
Ridglan Farms was charged with 311 violations of Wisconsin Administrative Code, including failing to handle dogs “in a humane manner that does not cause physical harm or unnecessary injury.”
It will surrender its DATCP license by July 1, 2026, under the settlement with the state.
Ridglan Farms was under criminal investigation for potential violations of state statutes banning mistreatment of animals and mandating proper shelter, after the appointment of a special prosecutor by the Circuit Court to conduct an independent inquiry into animal cruelty allegations.
The surrender of Ridglan’s license will leave only two major research dog breeders in the U.S.: Marshall Farms in North Rose, New York, and Oak Hill Genetics in Ewing, Illinois.
______________________________________________________________________________
AVMA News I The Canine Cognitive Dysfunction Syndrome Working Group guidelines for diagnosis and monitoring of canine cognitive dysfunction syndrome
Natasha J. Olby,VetMB, PhD, DACVIM, Joseph A. Araujo, BSc, Margaret E. Gruen, DVM, PhD, DACVB, Phillipa Johnson, BVSc, MSc, DECVDI, Eniko Kubinyi,PhD
December 24, 2025
Abstract
Canine cognitive dysfunction syndrome (CCDS) is diagnosed with increasing frequency, yet standardized diagnostic guidelines are lacking. The CCDS Working Group, an international group combining experts in the field and primary care veterinarians, proposes a definition of the syndrome and practical diagnostic criteria designed to aid clinicians and researchers alike. Canine cognitive dysfunction syndrome is defined as a chronic, progressive, age-associated neurodegenerative syndrome, characterized by cognitive and behavioral changes that affect daily life to varying degrees. These changes affect the behavioral domains of disorientation, social interaction, sleep disruption, house soiling, learning and memory, activity changes, and anxiety (DISHAA). We propose 3 severity stages. In mild CCDS, signs are subtle and of low frequency or severity, with preserved function. With progression, behavioral changes become more apparent and impactful, requiring management adjustments. In severe CCDS, debilitating deficits are overt, significantly impairing basic functions and necessitating comprehensive support. Two diagnostic levels are proposed. Level 1 is based on consistent history of progressive DISHAA signs, identification of alternate causes through physical, orthopedic, and neurologic examination and laboratory work; either normal neurologic examination or evidence of symmetrical, diffuse forebrain dysfunction; and persistence of signs following management of relevant comorbidities. Level 2 includes a brain MRI showing cortical atrophy with CSF cell counts within normal limits. Definitive postmortem histopathological confirmation rests on cortical atrophy, amyloid deposition, myelin loss, neuroinflammation, and amyloid angiopathy. Future priorities include the development of blood biomarkers and cognitive testing batteries for routine clinical settings, both of which will refine diagnostic accuracy and therapeutic monitoring.
Introduction
With increasing canine lifespan, age-related conditions like canine cognitive dysfunction syndrome (CCDS) have become more clinically important.1,2 However, diagnosis is challenging: signs are insidious and nonspecific, structural MRI changes emerge late, normal versus pathological decline is difficult to distinguish, and comorbidities further complicate interpretation. Recognizing CCDS early, when intervention may be most impactful, requires understanding of its spectrum and staging.1,2
Canine cognitive dysfunction syndrome has many parallels with Alzheimer disease (AD). Diagnostic guidelines for AD were first introduced in 1984 and have evolved with the understanding of the disease. Components of an antemortem diagnosis encompass history, core clinical criteria including cognitive or behavioral impairment in at least 2 defined domains, as well as blood, CSF, and imaging biomarkers. A final histopathological diagnosis requires certain criteria to be met (eg, scoring amyloid and neuritic plaques and staging neurofibrillary tangles).3–5 As AD diagnostic guidelines have evolved, the antemortem diagnostic accuracy has improved, facilitating timely care and supporting clinical trials. Similarly, unified diagnostic standards for CCDS are needed, with the intent to revise these standards as new data become available.
Problem Statement
Currently, there are no consensus-based diagnostic guidelines for CCDS. This absence contributes to diagnostic inaccuracy, impedes appropriate therapeutic decision-making and monitoring, and hinders the advancement of research efforts.
We are relatively early in our understanding of the clinical evolution of CCDS, and while biomarkers and cognitive testing are used in research settings, their application in routine clinical settings remains limited.1 There is a substantial body of data on cognitive aging in laboratory-bred Beagles,6 but the clinical picture in companion dogs is less well defined. Even standardized parameters to make a clear imaging or histopathological diagnosis are lacking. Caregiver questionnaires provide a practical way to capture behavioral changes and are important for monitoring and grading severity, given the difficulty in establishing cognitive status during a routine veterinary appointment and the expense and risks of a comprehensive diagnostic workup. However, responder bias and the influence of comorbid conditions limit their specificity, and current questionnaires should not be considered stand-alone diagnostic tools.
Proposed Solutions
The goal of this working group was to bring experts with diverse experience in CCDS together to accomplish the following:
Evaluate the published information on age-related canine cognitive decline and CCDS.
Provide a definition of CCDS and suggest practical criteria for establishing a diagnosis with 2 levels of certainty. These criteria are based on current knowledge and will be revised as understanding of the syndrome progresses.
Develop a severity grading of CCDS.
Definition of CCDS
Canine cognitive decline syndrome is a chronic, progressive age-associated neurodegenerative syndrome characterized by changes in cognitive function that are severe enough to affect daily life to varying degrees.
This syndrome manifests as changes in the following behavioral domains captured by the acronym DISHAA:
- Disorientation
- Impaired social interactions
- Sleep disturbances
- House soiling, learning, and memory deficits
- Activity changes (increased or decreased)
- Anxiety and fear (increased)
We propose that the term canine cognitive dysfunction syndrome should be considered an umbrella term for canine dementia. While amyloid and tau-associated pathology are recognized, other forms of neurodegenerative processes have not yet been explored and it is likely that different etiological processes exist.
Grading Severity
The following definitions of severity are proposed:
Mild CCDS: behavioral changes are subtle and at low frequency or severity; the dog is generally able to perform normal functions; changes are frequently attributed to old age by caregivers and may be missed unless specifically screened.
Moderate CCDS: behavioral changes are more frequent and severe, interfering with normal daily activities and requiring management adjustments.
Severe CCDS: behavioral changes are overt and debilitating; the dog needs support even for basic functions, and comprehensive management is needed.
These definitions reflect owners’ perception of their dog’s degree of disability rather than quantifying the pathology but provide a practical, relevant means of establishing syndrome stage.
Clinical Presentation
The caregiver history should describe slowly progressive behavioral changes that reflect deteriorating cognitive function. These signs are often described with the acronym DISHAA as listed in the definition.7,8 Dogs with CCDS will present with changes in one (early disease) or more of these behavioral domains, with recent studies suggesting that nighttime restlessness, decreased daytime activity, and changes in social interaction occur early in the disease, while house soiling and spatial disorientation are seen as disease severity increases.9–11 The characteristics of these signs might also vary by individual. For example, disorientation might present as going to the wrong side of doors, seeming to stare into space, or not recognizing caregivers (among others), while changes in activity might present as being less playful or active or showing increased repetitive or aimless activity. Similarly, changes in social interaction might include both increased (displaying attention-seeking behaviors) or decreased social behaviors. While many of these clinical signs affect quality of life for both dogs and their caregivers, changes in the sleep-wake cycle are often associated with increased caregiver burden.12 Caregivers also report sensory impairments (hearing, vision, and smell) and postural instability.13
It is critical to remember that CCDS remains a diagnosis of exclusion. Advancing age is associated with changes in all body systems, and many disease states can contribute to the behavioral changes within the DISHAA domains14 (Supplementary Table S1). A detailed physical examination that includes orthopedic, ophthalmic, and neurologic examinations is indicated, and a minimum database of CBC, serum chemistry panel, urinalysis, and blood pressure measurement should be offered. Full evaluation of each differential for the signs shown may require diagnostics beyond this testing. If, on neurologic examination, focal or lateralized (asymmetrical) signs of brain dysfunction, such as head tilt or unilateral circling, or cranial nerve deficits are identified or there are new-onset epileptic seizures, an alternate or additional cause of the behaviors is likely. In these cases, an MRI and CSF analysis should be strongly considered and offered to clients prior to establishing a presumptive diagnosis of CCDS. Neurologic examination in dogs with mild CCDS is likely normal, but in moderate to severe CCDS, findings might include bidirectional pacing, difficulty negotiating obstacles in the room (eg, getting stuck in corners), and difficulty engaging with the examiner or others in the room. Cranial nerve examination and postural reaction testing should be normal, but exaggerated startle responses might occur.
Importantly, identification of an alternate medical condition does not exclude CCDS as a diagnosis; however, medical conditions must be managed to evaluate their contribution to the clinical signs observed.
Caregiver Surveys
Caregiver-completed questionnaires have been developed with the goal of detecting and monitoring the progression of clinical signs of CCDS.15 While some questionnaires have designated scoring criteria to identify stages of CCDS, these are not stand-alone diagnostic tools, and the monitoring of an individual’s score over time is likely more revealing than the absolute score. Further, all questions must be evaluated for change from a dog’s normal behavior; for example, house soiling as a clinical sign of CCDS is only useful if the dog was previously fully housetrained. When used consistently and completed by the same caregiver over time, questionnaires allow identification of changes in one or more of the behavioral domains indicative of CCDS.
While several questionnaires have been developed, recent studies indicate that the DISHAA questionnaire is used most commonly in primary practice.16,17 Other scales used in clinical trials and research settings include the Canine Dementia Scale (CADES) and Canine Cognitive Dysfunction Rating scale (CCDR).10,18 These scales ask caregivers to rate the frequency or severity of their dog’s behavior across multiple domains commonly affected by CCDS; scoring of the questions generates total scores that can be tracked over time. Studies comparing CCDS questionnaires report that CADES is more sensitive to mild changes, while CCDR is less susceptible to placebo effect and remains more stable over time.11,19,20 While CADES and CCDR have been included in several research studies, the wide use of the DISHAA questionnaire by primary veterinarians makes it our recommended scale for clinical use (Supplementary Material S1). Furthermore, we believe that assessing and monitoring all 6 behavioral domains associated with CCDS as encompassed by the DISHAA tool is clinically valuable (anxiety, sleep, and/or activity levels are not fully addressed in CADES or CCDR). The DISHAA questionnaire may be completed by the caregiver prior to or during their appointment, with total scores tracked over time. When possible, a single caregiver should be designated to complete the questionnaire each time for consistency in response.
Advanced Imaging With MRI
Brain imaging is a pivotal component of establishing a diagnosis in brain disease. Disorders such as cerebral infarction, brain tumor, and encephalitis cause alterations in anatomy and tissue chemistry that are detectable by MRI. In contrast, neurodegenerative diseases are associated with progressive loss of brain parenchyma, which is more difficult to detect and differentiate from normal age-related changes. As such, the role of MRI in dogs displaying cognitive decline is as much to rule out other conditions as to attempt to establish a diagnosis of CCDS.
The MRI changes observed in CCDS overlap with those observed in the healthy aging brain, with both reflecting variable degrees of neuronal loss, dendritic degeneration, demyelination, small vessel disease, and white matter pathology. Studies to date suggest these changes are more severe in dogs with CCDS.21–23
Brain atrophy
Brain atrophy is the most significant alteration observed on MRI of dogs with CCDS.21 This condition manifests as reduction in cortical volume and enlargement of the CSF-filled space, which can be documented objectively through measurement of the interthalamic adhesion (ITA) and hippocampus.22–24 Although cerebral atrophy occurs with healthy aging as well as other neurodegenerative conditions,25 its detection on MRI combined with a presenting history and clinical signs consistent with CCDS increases the confidence of an antemortem diagnosis of CCDS.
In the clinical setting, ITA and hippocampal height can be measured from high-quality sequences acquired in a standard brain MRI protocol.22,23,26,27 However, inclusion of high-resolution T1-weighted isovolumetric 3-D sequences (magnetization-prepared rapid gradient echo [MPRAGE] or brain volume [BRAVO]), which permit standardized multiplanar reconstructions, optimizing hippocampal visualization and measurement accuracy, is recommended for aging dogs.
Three-dimensional isovolumetric T1-weighted brain volume sequence of a clinically and neurologically normal 4-year-old Beagle, with sagittal (A), transverse (B), and dorsal views (C) displayed. A 3-plane multiplanar reconstruction allows for an optimized and standardized transection of the interthalamic adhesion in sagittal plane. The interthalamic adhesion height measurement is demonstrated in yellow, and the cranial height extending from the floor to the ceiling of the calvarium is demonstrated in red.
Citation: Journal of the American Veterinary Medical Association 2026; 10.2460/javma.25.10.0668
Interthalamic adhesion height is affected by age, body size, breed, measurement plane, and imaging sequence; expressing it as a ratio to cranial height controls for body size and breed.26 Several studies have published findings for canine ITA height and report that ITA heights of < 5.0 mm are consistent with cerebral atrophy and are associated with clinical signs of cognitive decline.22,23 However, methodology was inconsistent and established normal ranges for dogs of different ages and breeds are lacking. The sensitivity, specificity, and accuracy of this measurement remain undetermined, particularly at different ages and disease stages, and normalizing to body size is likely necessary.
We suggest that measurements should be performed on the midsagittal plane slice where ITA margins are visible, surrounded by the third ventricle. This measurement can be performed with the use of a T2-weighted sequence, but an MPRAGE or BRAVO sequence should be used, when available, to optimize measurement at the maximum ITA diameter (Figure 1). Cranial height should be measured on the axial image at the same level to normalize to body size.
Hippocampal height decreases with age, and hippocampal volume is reduced in dogs with CCDS when compared with aged controls24,26,28; however, normal ranges need to be established. Although challenging on 2-D sequences, accurate and reproducible linear measurements of hippocampal height are described.26
Cerebral microbleeds
Vascular disease is well described in the aging canine brain and associated with cerebral amyloid angiopathy.29 On MRI, vascular disease can be observed as discrete microbleeds created by small collections of blood or blood products within the brain parenchyma and associated with small vessel pathology.30,31 These blood products create a focal disruption of the magnetic susceptibility within the tissue that is detectable on susceptibility-weighted and T2* gradient echo MRI sequences; one of these sequences should be included when imaging senior dogs. Lesions are observed as small, well-defined, rounded intraparenchymal signal voids. They are primarily located within the telencephalon, although they can be observed in the deeper gray and white matter structures of the brain.30 An increase in the number of cerebral microbleeds within the brain is described in geriatric dogs, both with and without signs of CCDS, and the intersection of cerebrovascular disease and CCDS is poorly understood.31
White matter pathology
Leukoaraiosis is described on MRIs of the geriatric canine population. It is observed as bilaterally symmetrical T2-weighted and FLAIR hyperintense white matter lesions. The etiology of these lesions is unclear although hypothesized to be due to cerebral small vessel disease. Their relationship to the diagnosis of CCDS has yet to be explored.32 The presence of more extensive age-associated white matter changes throughout the major white matter tracts of the canine cerebrum has been confirmed with the use of diffusion tensor weighted imaging, but currently these sequences are not routinely acquired in clinical canine brain MRI studies.33
Cerebrospinal fluid
Cerebrospinal fluid sampling is performed under anesthesia following an MRI with the goal of eliminating alternate diagnoses. A CSF sample is typically obtained from the cerebellomedullary cistern, and analysis includes measurement of protein concentration, a red and nucleated cell count, and cytological description. In dogs with CCDS, CSF analysis should be unremarkable. Measurement of biomarkers such as neurofilament and β-amyloid (Aβ) 42 can be performed on a research basis,34,35 but standard testing methods and normal ranges have not been established. Evidence of inflammation (elevated WBC count and protein concentration) or the presence of neoplastic cells or infectious organisms would support an alternate diagnosis.
Histopathology
The histopathological criteria to establish a diagnosis of CCDS have not been described and will require the establishment of the spectrum of normal age-related changes in cognitively successful agers, as well as an array of special staining and immunohistochemistry to highlight the full extent of the pathology.5 The neuropathological alterations in CCDS encompass a spectrum of structural, cellular, and molecular changes, reflecting a multifactorial disease process that disrupts neuronal function, network connectivity, and cognitive integrity. One of the most prominent neuropathological features in CCDS is cortical atrophy, particularly in the prefrontal and temporal lobes, which is accompanied by ventricular enlargement.21 In parallel, hippocampal neuron loss is frequently observed. However, these features are nonspecific with respect to the underlying pathological etiology.
Examination of postmortem brain tissue from aged dogs with and without CCDS has revealed Aβ plaques in the brain parenchyma, notably within the cortex and hippocampus, similar to those observed in human AD brains.36,37 Current literature suggests that Aβ accumulates progressively with age in dogs, both as extracellular plaques and soluble oligomers, mirroring AD-like pathology with changes in amyloid precursor protein processing that favors amyloidogenic cleavage.38 In dogs, these plaques are predominantly diffuse and less fibrillar compared to the dense-core neuritic plaques typical of advanced human AD. While Aβ deposition is a well-documented hallmark of canine brain aging, its association with cognitive impairment is debated.37
In AD, neurofibrillary tangles, consisting of hyperphosphorylated tau (pTau), are a defining pathological hallmark and strongly correlate with disease progression and neuronal dysfunction. The role of pTau and neurofibrillary tangles in CCDS remains unclear due to conflicting findings ranging from minimal to widespread pathology. This discrepancy may be explained by technical limitations, early-stage disease processes, or alternative mechanisms underlying neurodegeneration in dogs.21,39,40 Other important histopathological changes include myelin loss, particularly in the corpus callosum and frontal lobes, neuroinflammation with activated microglia and astrocytes, evidence of oxidative damage, and cerebral amyloid angiopathy.21,29,37
Future Direction
Cognitive testing
Although hands-on cognitive testing in dogs is challenging, it remains a critical diagnostic modality, highlighting the need for simple, robust protocols that can be implemented in routine clinical settings. Laboratory paradigms to assess learning, executive function, selective attention, and working memory are informative and sensitive to changes in early aging, but require weeks of training and specialized settings, making them impractical for companion dogs.6 Batteries of cognitive tests have been developed for use in companion dogs in both home41 and controlled clinical or laboratory settings.2,42–44 Despite these advances in test paradigms, cognitive testing is not yet feasible in everyday practice. Current methods are inconsistent, time intensive, confounded by practice or floor or ceiling effects, and lack benchmarks for healthy aging. Establishing large normative datasets across age groups, breeds, and health statuses and validating test performance both cross-sectionally and longitudinally against biomarkers and pathology will be essential for making CCDS assessment clinically practical and impactful.
Blood and CSF biomarkers
Development of blood, urine, or CSF tests that can be used to diagnose and monitor neurodegenerative diseases has long been a priority in human medicine. The ideal test should be sensitive and specific, noninvasive, readily available, and affordable, but a more realistic expectation is that biomarkers will provide additional evidence to support or refute a diagnosis and monitor disease progression. In recent years, blood and CSF tests have been developed to detect Aβ and pTau, increasing the certainty of AD diagnosis and staging.4
Measurement of canine CSF and plasma concentrations of neurofilament light chain (NfL) with the use of ultrasensitive techniques has shown increased levels with age and disease status.34,45,46 While NfL is a nonspecific marker of neuronal injury, not a diagnostic test, monitoring its plasma concentrations could be a practical means of longitudinal measurement. However, more data are needed on the influence of body weight and renal and nervous system disease burden before it is clinically useful. Measurement of CSF or plasma Aβ 40 and 42 concentrations and ratios potentially offer greater specificity, although concentrations are elevated in dogs with epilepsy and reference ranges have not been established.35,47,48 Reliable tests for canine pTau are still needed. Emerging studies of aging dogs report data on diverse biomarkers for CCDS, but further validation of their accuracy and reliability is needed.49,50 Currently, while measurement of NfL and Aβ 40 and 42 is feasible, there are not enough data to support their utility as clinical biomarkers for the development of clinical guidelines.
Recommendations
When a patient presents with age-associated behavioral changes, there is a progression of diagnostic steps and criteria that need to be met. These are presented in Figure 2 and summarized herein.
Flow chart depicting diagnostic approach to dogs exhibiting changes in 6 behavioral domains (disorientation; impaired social interactions; sleep disturbances; house soiling, learning, and memory deficits; activity changes; and increased anxiety and fear [DISHAA]), identified either through routine screening or as a presenting problem. Examples of canine cognitive dysfunction syndrome (CCDS) surveys include DISHAA, the Canine Cognitive Dysfunction Rating scale (CCDR), and the Canine Dementia Scale (CADES). Routine screening should be performed every 6 to 12 months from the age of 7 years onward to establish a behavioral baseline and ensure early recognition of changes across the 6 behavioral domains. BP = Blood pressure. Chem = Serum chemistry panel. NE = Neurologic examination. PE = Physical examination. UA = Urinalysis.
Citation: Journal of the American Veterinary Medical Association 2026; 10.2460/javma.25.10.0668
Core clinical monitoring criteria
Annual screening for behavioral signs of cognitive decline should start at 7 years of age. To increase compliance, this screening can be achieved by having caregivers complete a short senior health form (Supplementary Material S2). If behavioral abnormalities are reported by the caregiver, follow-up with a CCDS scale is recommended, with ongoing monitoring every 6 months. Beginning at 10 years of age, use of a CCDS scale every 6 months is recommended for all dogs, as changes in score for an individual inform on CCDS trajectory. The DISHAA scale is recommended for ease of clinical use, but the Canine Cognitive Assessment Scale, CADES, and CCDR are alternatives.
Core clinical diagnostic criteria to establish a diagnosis with level 1 certainty
History—Canine cognitive dysfunction syndrome is associated with slowly progressive behavioral signs reflecting cognitive impairment in an older dog, confirmed by an abnormal CCDS scale score. The history should confirm that the signs reported represent a deterioration from the dog’s previous level of function and should capture relevant environmental factors and medications. Questioning to identify possible comorbidities is critical.
Physical examination—A physical examination should be performed that includes orthopedic, neurologic, and ophthalmic examinations as well as blood pressure measurement. The goal is to identify and manage comorbidities that are contributory.
Neurologic examination—A neurologic examination should be performed to identify and localize neurologic signs. Canine cognitive dysfunction syndrome is not associated with lateralizing, focal signs, but rather diffuse signs of forebrain dysfunction. The cranial nerve examination, postural reactions, and spinal reflexes are unaffected by CCDS. Signs of CCDS identified on neurologic examination might include pacing, spatial disorientation, difficulty attracting or maintaining the dog’s attention, and exaggerated responses to tactile or threatening stimuli (eg, menace response, palpation), but the examination is likely to be normal with mild CCDS.
Minimum database—A CBC, serum chemistry panel, and urinalysis are recommended with the goal of identifying comorbidities that might result in similar behavioral changes and that could be treatable. Additional blood work and other diagnostics should be pursued as appropriate.
Core clinical diagnostic criteria to establish a diagnosis with level 2 certainty
The goal of MRI in dogs with suspected CCDS is to rule out alternate diagnoses and identify cerebral atrophy (ITA height of < 5 mm). Due to heightened clinical suspicion for an alternate diagnosis, brain MRI should be pursued whenever neurologic signs are lateralized or localized to a focal region of the brain or when there is presentation of new epileptic seizures. Establishing normal ITA-to-brain height ratios across breeds and ages is needed to determine pathological thresholds.Cerebrospinal fluid analysis parameters should be within normal limits.
https://avmajournals.avma.org/view/journals/javma/aop/javma.25.10.0668/javma.25.10.0668.xml
______________________________________________________________________________
AVMA News I Monthly chewable tablet for screwworm in dogs receives conditional approval
Published on December 24, 2025
The Food and Drug Administration (FDA) continues to build up veterinarians’ arsenal against New World screwworm (NWS), should it cross the U.S.-Mexico border.
On December 17, the FDA conditionally approved Credelio Quattro-CA1 (lotilaner, moxidectin, praziquantel, and pyrantel) monthly chewable tablets for the treatment of infestations of NWS (Cochliomyia hominivorax) larvae in dogs and puppies at least 8 weeks of age and weighing at least 3.3 pounds.
Credelio Quattro-CA1 received expedited review through the priority zoonotic animal drug designation, as provided under the Coronavirus Aid, Relief, and Economic Security (CARES) Act. (Courtesy of Elanco Animal Health)
This comes after recent emergency use authorizations for two animal drugs for NWS: Credelio and Credelio Cat for dogs and cats, respectively, and another conditional approval for Exzolt Cattle-CA1 (fluralaner) topical solution in cattle.
Screwworm’s northward migration from Central America has prompted a massive response from animal health officials, who have called for heightened vigilance from small and large animal veterinarians to ensure the parasite, declared as eradicated in 1966, does not regain a foothold in the U.S.
Credelio Quattro CA-1, available only by prescription from a licensed veterinarian, comes in five strengths of flavored chewable tablets with active ingredients by weight.
The minimum dosage is 9 mg per lb (20 mg/kg) lotilaner, 0.009 mg per lb (0.02 mg/kg) moxidectin, 2.28 mg per lb (5 mg/kg) praziquantel, and 2.28 mg per lb (5 mg/kg) pyrantel (as pamoate salt). Dogs over 100 lb should be given the appropriate combination of chewable tablets, according to the FDA.
Credelio Quattro-CA1 must be orally administered with food. The FDA says care should be taken to ensure that the dog consumes the complete dose and that if vomiting occurs within an hour after administration, readminister a new dose.
Credelio Quattro and Credelio Quattro-CA1 contain the same active ingredients at identical doses: lotilaner, moxidectin, praziquantel, and pyrantel.
Credelio Quattro, sponsored by Elanco Animal Health, is already approved for various flea-, tick-, and worm-related indications in dogs and puppies.
The conditional approval was based on a study evaluating the efficacy of lotilaner against New World screwworm, according to the company, which said a peer-reviewed study showed oral administration of the antiparasitic drug at the minimum recommended dosage demonstrated 100% efficacy against NWS larvae within 24 hours of treatment in naturally infested dogs.
A single dose of Credelio Quattro-CA1 is “reasonably expected” to treat NWS larvae present at the time of treatment, though reinfestation may occur sooner than one month, according to Elanco’s application for conditional approval. “Treatment should be used in conjunction with the mechanical removal of larvae (live and dead) remaining in the wound after treatment.”
While livestock are most commonly affected, NWS can infest all warm-blooded animals, including wildlife, pets, and even humans. Still, most dogs in the U.S. remain at low risk of contracting NWS due to their geographic location, though animals near the U.S.-Mexico border and those that travel to countries with active cases are at higher risk of exposure.
“As a veterinarian, I’d encourage pet owners to monitor for any wounds on their dogs as even a minor scratch could serve as an entry point for New World screwworm,” said Dr. Casey Locklear, parasitology lead at Elanco, in the company announcement. “I also recommend pet owners keep their dogs on year-round parasite protection like Credelio Quattro to help minimize self-inflicted scratching that could create vulnerable sites.”
https://www.avma.org/news/monthly-chewable-tablet-screwworm-dogs-receives-conditional-approval
_______________________________________________________________________________________
DVM 360 I USDA grants full approval for novel canine parvovirus therapy
Elanco’s monoclonal antibody therapy, Trutect, received a conditional license from the federal agency in 2023.
Kristen Coppock Crossley, MA
December 16, 2025
The USDA has granted full approval for a novel therapy to treat canine parvovirus (CPV), according to Elanco Animal Health. The Canine Parvovirus Monoclonal Antibody (CPMA)—now Trutect— from Elanco received a conditional license from the USDA in 2023, and the agency extended its use for passive immunity in puppies exposed to the virus, in June 2025 .
CPV is a common infectious disease in dogs around the world. Left untreated, it results in death in approximately 90% of cases. Survival rates are estimated to be 80% to 90% in cases with aggressive treatment.2
According to data compiled by Elanco, 93% of puppies with CPV that received CPMA treatment survived. Furthermore, patients infected with the virus spent an average of 1.87 fewer days in the hospital.1
“Receiving full USDA approval of our lifesaving [parvovirus] treatment reinforces our promise to go beyond for veterinarians, pet owners, and, most importantly, the puppies across the country who need it most,” Bobby Modi, executive vice president, US pet health and global digital transformation for Elanco, said in a news release.1 “Under the brand name Trutect, our mission remains the same, to save puppies from parvovirus.”
As a nonenveloped, single-stranded DNA virus, CPV can survive for more than a year in the environment, which serves as a means of transmission. Transmission occurs through a fecal-oral route such as exposure to CPV in feces or vomit as well as fomites.3
Puppies ages 6 weeks to 6 months are more likely to develop severe illness with CPV, especially those younger than 12 weeks. CPV disease can also develop in unvaccinated or improperly vaccinated adult dogs. In some regions, distribution of CPV disease has been seasonal, while socioeconomic disadvantage was found to play a role in at least one surveillance study.3
Tannetje Crocker, DVM, an emergency veterinarian at Veterinary Emergency Group in Dallas, Texas, and owner of Alta Vista Animal Hospital in Fort Worth, said she has seen firsthand the “devastating” effect of CPV throughout her career. “Since 2023, I have proudly used CPMA and have reunited countless puppies with their families. I’ve also used the product on exposed puppies, helping to keep them protected from this highly contagious virus. I believe every [veterinary] clinic should keep [CPMA] on hand because you never know when a parvo puppy will need this targeted treatment,” Crocker said in the release.1
According to Elanco data on file, there is a 90% satisfaction rate from clinics that incorporated CPMA into their treatment protocols. Additionally, 92% of veterinarians reported a reduction in overall clinic stress while using CPMA in the treatment process, and 83% expressed they would recommend it to their peers.
“The real-world data collected over the first year of CPMA availability shows that as a result of these efforts and more, survival rates for puppies battling the deadly virus improved. We also saw enhanced operational efficiency and high satisfaction within veterinary clinics–demonstrating that Trutect has already started rewriting the protocol for parvovirus treatment,” Jill Pattee, DVM, a veterinarian for Elanco Animal Health, said in the release.1
In a 2024 interview with dvm360, Crocker also recommended vaccination against CPV infection for reducing disease exposure and transmission, as well as client education. “I see a ton of dogs come in that are unvaccinated. Puppies are not fully vaccinated…So the recommendation is simple, and it’s something that I feel like people should know. But then the more I talk to pet owners, and I talk to them about what [CPV] is, they just don’t know. So we want to make sure we get the word out about how important vaccination is and knowing the early signs of [CPV],” she said in the interview.4
Common clinical signs of CPV include fever, lethargy, inappetence, vomiting, severe diarrhea, dehydration and depression. In rare cases, sudden death or tachypnea caused by myocarditis may also occur.2,3
Elanco launched a multi-year campaign in 2024 with a mission to help prevent and defeat CPV in puppies. As part of this campaign, the company introduced the first National Parvo Awareness Day to provide education about CPV. Additionally, Elanco has expanded its manufacturing facility in Elwood, Kansas, and is continuing to invest in the growth of the company’s monoclonal antibody platform.1
For pet owners with dogs exposed to or infected with CPV, Elanco is increasing availability of CPMA and offers a $200 rebate with purchase. The company has also donated more than $3 million of CPMA product across 2300 clinics and shelters throughout the US.1
References
- Elanco announces full USDA approval of CPMA (now Trutect) the first-and-only treatment for deadly canine parvovirus. News release. Elanco. December 15, 2025. Accessed December 16, 2025. https://www.elanco.com/us/newsroom/press-releases/trutect-USDA-approval
- What every pet owner should know about parvo-virus. Jefferson Animal Hospital Regional Emergency & Trauma Center. Accessed December 16, 2025. https://jeffersonanimalhospitals.com/what-every-pet-owner-should-know-about-parvo-virus
- Sykes JE. Chapter 14: Canine parvovirus infections and other viral enteritides. In: Sykes JE, ed. Canine and Feline Infectious Diseases. 2013;141-151. doi:10.1016/B978-1-4377-0795-3.00014-4
- Crossley KC. Generating awareness about parvovirus vaccination. dvm360. June 25, 2024. Accessed December 16, 2025. https://www.dvm360.com/view/generating-awareness-about-parvovirus-vaccination
https://www.dvm360.com/view/usda-grants-full-approval-for-novel-canine-parvovirus-therapy
_________________________________________________________________________
AVMA I Personality traits and workplace factors predict professional quality of life among companion-animal veterinary professionals
Lindsay Nakonechny, MSc, Alissa Cisneros, MSc, Carly M. Moody, PhD, and Anastasia Chiara Stellato, PhD
December 15, 2025
Objective
To assess the prevalence of burnout (BO), secondary traumatic stress (STS), and compassion satisfaction (CS) and identify associated individual, clinic, and dog-handling factors among veterinary professionals.
Methods
A cross-sectional online questionnaire was distributed to veterinary professionals in Canada and US (2023 to 2024). The questionnaire collected individual, clinic, and dog-handling information and measured ProQOL (BO, STS, CS). Logistic regression models examined associations between these factors and ProQOL.
Results
Participants (n = 691) had moderate BO (71.2%), STS (71.8%), and CS (74.3%); 2.4% reported high STS, and none had high BO. Veterinarians had lower odds of moderate/high BO (OR, 0.50; 95% CI, 0.32 to 0.78) and CS (OR, 0.12; 95% CI, 0.021 to 0.64) compared with nonveterinarians. Below-normal personality traits were associated with moderate/high BO and/or STS: extraversion (BO: OR, 2.25; 95% CI, 1.47 to 3.46), agreeableness (BO: OR, 2.02; 95% CI, 1.29 to 3.18; STS: OR, 1.60; 95% CI, 1.07 to 2.39), conscientiousness (BO: OR, 3.91; 95% CI, 2.41 to 6.34; STS: OR, 3.81; 95% CI, 2.47 to 5.88), emotional stability (BO: OR, 1.96; 95% CI, 1.24 to 3.11), and openness (BO: OR, 1.64; 95% CI, 1.05 to 2.56; STS: OR, 1.88; 95% CI, 1.26 to 2.81). Stress-reducing certification was associated with moderate/high BO (OR, 2.04; 95% CI, 1.14 to 3.64).
Conclusions
Personality traits and individual factors were associated with ProQOL, whereas handling techniques were not.
Clinical Relevance
Findings provide exploratory evidence for workplace strategies to reduce BO and STS and enhance CS while generating hypotheses for future intervention research.
Veterinary professionals work under emotionally and physically demanding conditions that can adversely affect their professional quality of life (ProQOL) or how individuals feel in relation to their work as a helper.1 There are both positive and negative aspects of helping others that affect an individual’s ProQOL, including compassion fatigue (CF) and compassion satisfaction (CS).1 Compassion fatigue refers to the adverse consequences of helping others and is further broken down into burnout (BO) and secondary traumatic stress (STS).1 Burnout arises from cumulative high workloads and stressors and includes feelings of overwhelm, inefficacy, and physical and mental exhaustion.1 Secondary traumatic stress results from direct or indirect exposure to work-related trauma (eg, animal suffering, distressed clients) and presents with symptoms such as fear, sleep disruptions, and avoiding traumatic triggers.1 In contrast, CS includes pleasurable and positive feelings derived from helping others; it is independent from CF but can help to mitigate its negative effects.1 In studies2–4 of veterinary populations, ProQOL is commonly assessed using the ProQOL-5 scale, which is a validated, 30-item self-report measure.1 High levels of BO and STS have been reported among veterinarians of all practice types in the US and Canada, with 41% to 50% and 59% to 69% of respondents classified as having high BO and STS, respectively.2–4 Globally, studies report that 40% to 74% of veterinary populations have moderate scores of CS, including samples of nonveterinarians and veterinarians5–6 and veterinarians only.3,4,7
Common occupational stressors in the veterinary industry include high job demands (eg, long work hours, work pressure, cognitive burden) and exposure to ethical conflicts, moral distress (eg, animal neglect), animal suffering, and client distress, such as performing euthanasia and/or making euthanasia-related decisions.8–10 Such stressors have been identified as contributors to BO and STS in individuals who work with animals, including veterinarians and technicians.5,11–13 In veterinarians, BO has been associated with poor physical health, depression, anxiety, suicidal ideation, and suicide.10,14 Compounded by the dual responsibility to both patients and their human caregivers and client expectations, these stressors are uniquely characteristic and of pressing concern in veterinary medicine.8,9,13
Several risk factors for BO, STS, and CS have been identified in veterinary populations, including staff role, practice type, and individual characteristics. Differences in role (ie, veterinarians vs nonveterinarians) have been linked to variations in ProQOL. For example, technicians have been found to have higher BO compared to veterinarians.13,15,16 However, other research has not identified differences in BO or STS between these roles.11 Practice type may also impact ProQOL as emergency veterinary medicine is associated with unique stressors, such as shift work, critical cases, and increased exposure to patient death and distressed clients,17,18 potentially increasing risks of BO and STS compared to general practice. Individual characteristics may further shape ProQOL as younger veterinary staff, females, and recent graduates have been reported to be at a higher risk of BO and STS.13,19–21 Further, personality may influence how individuals experience and respond to occupational stress and risk of BO.22 Personality refers to consistent patterns of feelings and behaviors among individuals and is commonly assessed using the 5-factor model of personality traits, or the “Big Five,” which is comprised of 5 personality traits: extraversion, agreeableness, conscientiousness, emotional stability, and openness.23 Research in human healthcare has linked certain Big Five personality traits, particularly higher neuroticism and lower emotional stability, to an increased risk of BO in physicians.24
Beyond these individual and clinic-level influences on ProQOL, interactions with patients may also shape veterinary professionals’ ProQOL. Handling animals that are fearful and/or show aggression can be especially stressful for handlers as staff must manage the immediate risk of injury, often in fast-paced or unpredictable situations. Dog bite and scratch injuries are consistently among the most frequently reported occupational hazards for veterinary staff globally and across practice types.25 Using passive or minimal restraint (ie, holding the dog’s shoulders, allowing some movement of the body and limbs) is recommended26 as it has been associated with fewer signs of fear in dogs during routine examinations in some experimental studies.27,28 Reduced dog fear may enhance the safety of veterinary staff as a North American survey study29 reported lower patient-related injury rates among small-animal clinics where all staff were certified in stress-reducing practices compared to clinics with some or no certified staff. The use of minimal restraint may be associated with lower BO and STS and/or higher CS in veterinary staff due to its potential to reduce dog fear and aggression during examinations,27,28 support positive dog-human interactions,26 and reduce patient-related injuries.26,29
To date, research in the US and Canada has primarily focused on risk factors for ProQOL among veterinarians of all practice types, with comparatively less attention given to the broader veterinary team, including technicians and other support staff. Furthermore, there has been limited exploration of the factors that contribute to CS. The aim of this study was to estimate the prevalence of BO, STS, and CS and to identify predictors of ProQOL among veterinary professionals in Canada and the US who routinely handle dog patients. We hypothesized that ProQOL scores would be associated with (1) individual factors (eg, demographics, personality traits), (2) clinic-level factors (eg, staff role, practice type), and (3) restraint techniques used (minimal vs full-body restraint).
Methods
This cross-sectional study was reviewed and approved by the Texas Tech University Research Ethics Board (No. IRB2023-462) for research involving human participants.
Data collection
An online cross-sectional questionnaire was distributed to veterinary professionals who handle dogs routinely across Canada and the US. The questionnaire was originally developed for a previous study30 using the same dataset; full methodological details are available in Nakonechny et al.30 Briefly, eligible participants were 18 years of age or older, resided in Canada or the US, and worked as a licensed veterinarian, veterinary technician, or assistant (either licensed or unlicensed) who handles dogs during routine health examinations. Participants were recruited using convenience snowball sampling via advertisements on social media platforms, including Facebook and Instagram. Email invitations were also sent to veterinary organizations in both eligible countries (eg, Canadian Veterinary Medical Associations and AVMA) to distribute to their members for further recruitment. Data collection occurred from October 2023 through February 2024. Participation was voluntary and anonymous, with informed consent obtained prior to survey access. The completion time averaged approximately 15 minutes. To minimize social desirability bias, no identifying information was collected or linked to participant responses, and participants were informed of such anonymity. We estimated the minimum sample size as 550 responses based on a conservative event-per-variable value of 30 with approximately 15 explanatory variables, and after inflating the sample size by approximately 30% to account for a low response rate,31 we estimated a sample of 700 responses. To verify external validity, we also considered the size of the target population. Using the target population of companion-animal veterinarians (US, approx 71,693; Canada, approx 11,885) and veterinary technicians and assistants in US and Canada (US, approx 122,000; Canada, approx 15,300), the required sample at 95% confidence and a 5% margin of error was estimated to be approximately 400.32–34
Questionnaire
The online questionnaire was created using Qualtrics survey software (Qualtrics LLC). The questionnaire was comprised of 26 questions, categorized into 5 sections: (1) participant and clinic information, (2) participant ProQOL (using the ProQOL-5 scale), (3) general examination practices, (4) perceived factors affecting dog handling during examinations, and (5) frequency of using 14 dog-handling techniques and tools. This analysis focused exclusively on data from sections 1 and 2. Variables from section 5 were included only as explanatory factors in the current models, whereas data from sections 3 through 5 are examined in detail in a separate analysis in Nakonechny et al,30 where the full questionnaire is also provided.
Statistical analysis
All analyses were performed with Stata statistical software, version 15.1 (StataCorp).
Data management
Professional quality of life was assessed using the ProQOL-5 scale, which provides 3 subscale scores: BO, STS, and CS. This validated self-report tool is widely used in research to assess the beneficial and detrimental impacts of helping professions, including veterinary professionals.2–7,11,12 Each subscale contains 10 items rated on a 5-point Likert scale (1 = never to 5 = very often), generating total scores from 10 to 50. Scores were categorized using established thresholds into low (< 23), moderate (23 to 41), and high (> 41) ranges.1 Personality traits were evaluated with the Ten Item Personality Inventory (TIPI), a brief 10-item measure of the Big Five domains: extraversion, agreeableness, conscientiousness, emotional stability, and openness.23 Each trait is represented by 2 items, with responses rated on a 7-point scale, ranging from 1 (strongly disagree) to 7 (strongly agree).23 Handling practices were examined by asking participants to report on a Likert scale how often they used 14 specific dog-handling techniques with small (< 35 lb) and large (≥ 35 lb) dogs under 3 behavioral states (calm, fearful, aggressive). The list of techniques, accompanied by written descriptions and photographs, was adapted from Carroll et al35 and is provided in the questionnaire documented in Nakonechny et al.30
To appropriately conduct logistic regression models, our outcome variables (BO, STS, and CS) were consolidated to create a binary outcome with 2 levels, “high” (moderate, high; ≥ 23) and “low” (low; < 23). Given the distribution of ProQOL scores, in which the majority of participants reported moderate scores and relatively few to no participants reported scores at the high or low cutoffs, all subscale outcome variables were dichotomized to “low” (range cut point, < 23) and “moderate/high” (range cut point, moderate: 23 to 50) categories based on established cut points (range cut point).1 This approach was chosen to address the non-normal distribution of the data and enable the use of logistic regression, which does not assume normality of the outcome variable. Further, it helped to consolidate categories with sparse count responses (eg, high BO and STS) to improve model stability and interpretability of results while maintaining alignment with established ProQOL cut points. Given the distribution of the data, binary categorization was deemed most appropriate given the data distribution.
A total of 19 explanatory variables were used for analysis, encompassing 13 variables related to participant demographics and clinic characteristics and 6 variables describing handling practices of calm, fearful, and aggressive dogs. To reduce the total variables tested for analysis, conceptually similar items were combined into composite variables. For example, the category “nonveterinarian” was generated by grouping licensed veterinary technicians, nonlicensed veterinary technicians, and veterinary assistants as these roles originated from the same survey item on staff position and were closely related in meaning. Reference categories for categorical predictors were selected either based on the most common response or biological plausibility. For example, in analyses of staff role, nonveterinarians were used as the referent group since they represented the larger proportion of respondents. Data cleaning included review of responses to “other” options, which were crosschecked against existing categories to reduce misclassification bias. Categories with limited conceptual relevance or < 5 responses were collapsed with similar categories or excluded to prevent overfitting. For instance, participants who selected “other” for practice type but whose responses did not align with established categories (small animal practice, mixed animal practice, or emergency clinic) were removed from analyses. Suspicious responses that suggested automated submissions, such as unusually rapid completion times (< 5 minutes) or nonsensical open-text answers, were also excluded.36,37
Risk factor analysis
Logistic regression models were developed to assess the impact of the explanatory variables on the following outcomes: (1) CS score, (2) BO score, and (3) STS score. Explanatory variables were related to individual and clinic factors: country, gender, age, graduation year, TIPI of the Big Five personality traits (extraversion, agreeableness, conscientiousness, emotional stability, openness), staff role (veterinarian, nonveterinarian), practice type (small animal, mixed animal, emergency clinic), dog bite history, and stress-reducing certification (eg, Fear Free Certified Veterinary Professional). Additionally, 3 separate logistic regression models were conducted to assess whether the frequency of using minimal or full-body restraint when handling calm, fearful, and aggressive dogs influenced the outcome variables (BO, STS, CS scores). Two restraint techniques were selected for analysis: minimal restraint, representing a low level of physical restraint, and full-body restraint, reflecting a high degree of physical control and recognized as aversive for dogs.26,27 These techniques were chosen because they reflect contrasting levels of physical restraint. To facilitate interpretation, responses regarding frequency of use were recoded into a binary variable with 2 categories: “used” (always, often, and sometimes) and “not used” (rarely and never). This variable was based on handling practices with large dogs across all behavioral demeanors, as in a previous study30 using the same questionnaire and participant data, there were no meaningful differences in restraint frequency observed between small and large dogs.
Relationships between continuous predictors (TIPI scores) and outcomes (CS, BO, STS) were examined using locally weighted regression curves and by testing quadratic terms.31 These associations were neither linear nor quadratic, and log transformations did not improve model fit. As a result, continuous variables were categorized. Following Gosling et al,23 TIPI scores were classified as below or above population norms (extraversion, 4.44; agreeableness, 3.23; conscientiousness, 5.40; emotional stability, 4.83; openness, 5.38). Correlation testing among explanatory variables showed that none exceeded a coefficient of 0.70, reducing multicollinearity concerns.
Univariate analyses were conducted to evaluate each independent variable’s association with the 3 ProQOL outcomes (CS, BO, STS). Variables with a liberal significance threshold of P ≤ .20 were retained for multivariable modeling to ensure that potentially meaningful factors were retained.31 Multivariable models were then constructed using a manual stepwise approach, with only predictors meeting P < .05 kept in the final models. Potential confounders were identified using biological and clinical plausibility based on prior research.31 A covariate was considered a confounder if its inclusion altered another variable’s coefficient by more than 30%,31 a conservative cutoff to identify influential covariates. To improve model parsimony, only interactions that were both biologically relevant and reduced the Bayesian information criterion were retained. Model fit was evaluated with the Bayesian information criterion, with a lower value preferred. Results were expressed as ORs with 95% CIs. Odds ratio values above 1 indicated greater odds (potentially risk enhancing), whereas values below 1 indicated lower odds (potentially protective). Given the cross-sectional design, risk factors generated from these models are reported as statistical associations with BO, STS, and CS without implying causality or temporal direction.
Results
Participants
In total, 920 responses were submitted. After excluding 229 suspected automated responses (identified using recommended screening methods)37 and individuals who did not meet eligibility requirements, 691 responses remained for analysis. As several questionnaire fields were optional, the number of respondents differs across variables. The majority of participants were located in the US (541 of 691 [78.3%]), identified as female (429 of 690 [62.2%]), and were between 25 and 44 years of age (531 of 686 [77.4%]). With respect to staff role, 454 of 671 (67.7%) were nonveterinarians (technicians and assistants), and 217 of 671 (32.3%) were veterinarians. Participants worked primarily in general small-animal practice (361 of 689 [52.4%]), followed by mixed-animal practice (209 of 689 [30.3%]), emergency clinics (105 of 689 [15.2%]), and other settings, such as shelter medicine (14 of 689 [2.0%]). A large proportion (544 of 626 [86.9%]) reported holding at least 1 stress-reducing certification, including Fear Free Certified Veterinary Professional, Sophia Yin Low Stress Handling Silver Certification, or Karen Pryor Better Veterinary Visits. Detailed characteristics of participants are provided in Supplementary Table S1 in Nakonechny et al.30
Risk factors for BO, STS, and CS
All 5 personality traits (extraversion, agreeableness, conscientiousness, emotional stability, and openness) were associated with BO scores; specifically, participants with below-normal scores had higher odds of moderate/high BO compared to those with above-normal scores (Table 2). Participants with a stress-reducing certification had higher odds of moderate/high BO compared to participants with no certification. Further, veterinarians had lower odds of moderate/high BO compared to nonveterinarians.
Several personality traits (agreeableness, conscientiousness, and openness) were associated with STS scores. Similar to BO, participants with below-normal scores had higher odds of moderate/high STS compared to those with above-normal scores (Table 2).
Regarding CS, veterinarians had lower odds of moderate/high CS compared to nonveterinarians (Table 2). Further, participants who were unsure of their dog bite history had higher odds of moderate/high CS compared to those with no bite history.
Several risk factors were not detected to influence ProQOL outcomes. Specifically, the frequency of using minimal or full-body restraint on calm, fearful, and aggressive dogs did not influence participants’ BO, STS, or CS scores. Participants’ country of residence was not associated with ProQOL nor were factors reported in previous literature, including gender, age, and graduation year.9,21–23
Discussion
The majority of participating veterinary professionals in this study scored within the moderate range for BO, STS, and CS. This finding is consistent with other studies5–7,11,13 assessing the prevalence of low, moderate, and high BO, STS, and CS among mixed samples of veterinarians and support staff internationally. No participants in our sample were classified as having high BO, and only a small proportion were categorized as having high STS (2.4%). In contrast, prior studies2–4 of veterinarians in Canada and the US, focusing exclusively on homogeneous samples of practicing veterinarians, have reported much higher proportions of individuals whose ProQOL scores fall within the high range for BO and STS. Thus, differences in the prevalence of low, moderate, and high ProQOL scores among various studies may be attributed to sample composition. Although most participants had moderate scores for all ProQOL subscales, the risk of moderate/high BO and CS varied between roles.
Results from regression models found that those who identified as nonveterinarians had an increased risk of moderate/high BO and CS compared to veterinarians. Previous research has reported high BO among veterinary technicians, with contributing factors including unmanageable workloads, interpersonal relationships, low reward for effort, and feeling unsupported.15,16,38,39 Though day-to-day responsibilities and job requirements differ by staff role, veterinarians have reported experiencing similar occupational stressors as veterinary technicians and assistants.8,9 Technicians may engage in frequent difficult interactions with clients and coworkers, including incivility, and are often under time constraints with limited autonomy and decision-making authority.39,40 The cumulative demands of these responsibilities, coupled with lower compensation and reward and limited support, may contribute to the higher rates of BO observed among technicians.15,16,39
Differences in job duties between veterinarians and support staff may also explain the observed variation in CS. For example, veterinarians often move from patient to patient throughout the day, focusing on diagnoses, treatment plans, and documentation,6 whereas support staff may spend more time directly engaging with patients by providing supportive care (eg, petting, feeding, monitoring patients after treatment).6 There are limited studies on CS among veterinary populations; one mixed-methods study41 of Canadian veterinarians and technicians found that interacting with, helping, and developing relationships with patients and clients were highly satisfying aspects of their work and were positively associated with CS. Although negative interactions with clients may contribute to BO, positive interactions may also enhance CS for support staff.41 Given the limited research and observed variation in ProQOL across staff roles, further investigation is needed to inform the development of role-specific interventions and workplace practices that reduce BO and support CS.
Participants with a stress-reducing certification were more likely to have moderate/high BO. Other survey studies42,43 have found that veterinary professionals often encounter barriers to using stress-reducing practices, such as high caseloads and time pressures, unsupportive workplace cultures, and limited colleague understanding of these approaches. These individuals possess the knowledge about how to reduce dog fear during routine care but may be prevented from doing so due to various constraints, potentially resulting in moral conflicts leading to distress, as they cannot pursue actions they perceive to be ethically correct.44 One such constraint may be the clinic culture and environment not being conducive for stress-reducing practices. For example, we hypothesize that individuals who work at clinics where all staff involved with animal handling are certified and/or have positive attitudes toward stress-reducing practices may experience fewer barriers and, subsequently, less BO and/or higher CS. Future research is warranted to clarify the relationship between stress-reducing certification and staff ProQOL.
Personality traits were found to influence both BO and STS. Lower emotional stability and extraversion scores were associated with higher BO. This is consistent with prior research, where individuals with greater difficulty managing negative emotions (lower emotional stability) or less tendencies to seek out social interactions (lower extraversion) may be more vulnerable to emotional exhaustion.45 Prior research in veterinary and human healthcare also links neuroticism, conceptually similar to low emotional stability, with increased stress and BO.13,45 Below-normal scores of agreeableness, conscientiousness, and openness were associated with higher STS. Individuals with low agreeableness and/or openness may be less inclined to seek help from others, such as mental health professionals, or lack supportive relationships.1,46 Low levels of conscientiousness have been associated with decreased coping abilities and increased susceptibility to stress.47 Several personality traits associated with STS were also predictive of BO (agreeableness, conscientiousness, and openness), suggesting possible comorbidity between these constructs. Below-normal Big Five personality scores may also reflect a mismatch between staff characteristics and the traits that clients and colleagues tend to value, specifically higher agreeableness, conscientiousness, and extraversion in veterinary professionals.48 This mismatch could increase the likelihood of stress due to interpersonal conflicts between veterinary staff and clients, thereby exacerbating the risk of BO. However, this relationship is speculative and likely not straightforward. For instance, Perret et al49 found that poor veterinarian mental health measures (eg, high perceived stress) were associated with higher client satisfaction. Assessing personality traits may provide insights into individuals’ susceptibility to occupational stress and their ability to cope effectively. Future studies should clarify these associations, which could inform tailored preventive or intervention approaches to lessen BO and STS.
Several tested predictors of ProQOL were not statistically associated but remain important to consider. Notably, there were no detected associations between the use of minimal or full-body restraint and BO, STS, or CS, suggesting that handling practices alone may not influence ProQOL. It is possible that the use of full-body restraint is common enough in clinical settings that it is not perceived as stressful or problematic by participants or that handling techniques are associated with more transient effects (eg, acute stress, changes in mood) that are not captured by constructs like BO, STS, or CS. Other stress-reducing strategies (eg, providing treats, use of counterconditioning and/or desensitization) may have a greater impact on staff ProQOL than the handling techniques assessed. Data from a related analysis using the same survey and participant population revealed that individuals with low to moderate STS were more likely to apply full-body restraint on fearful dogs, compared to those with high STS.30 This suggests that STS may influence handling practices, but the routine use of these handling techniques does not influence STS or other ProQOL subscales in this context. It is possible that the effects of handling-related stress on ProQOL are moderated by other unexplored factors, such as perceived control, resilience, and training or previous experiences. Additionally, the approach in dichotomizing handling frequency may have limited our ability to detect subtle effects.
Furthermore, individual factors, such as gender, age, and graduation year, reported in previous studies13,19–21 as risk factors for poor veterinary ProQOL (eg, high BO and STS), were not detected as risk factors. This is possibly due to the sample composition (eg, low variability in ProQOL scores). These findings highlight the complexity of factors influencing ProQOL and suggest that predictors are likely context and sample dependent. Future research should examine individual, clinic, and handling-related factors using larger, more diverse samples and observational designs to better capture their potential impact on staff ProQOL.
This study has several limitations, primarily related to potential biases. The use of convenience and snowball sampling through email and social media may have introduced voluntary response bias, likely favoring younger individuals, those comfortable with online platforms, and participants with a stronger interest in stress-reducing practices. Nonresponse bias or a healthy worker effect is also possible as most respondents reported moderate ProQOL scores, whereas national surveys estimate that approximately 18% of US veterinarians experience high or very high BO.13 This suggests that professionals with high BO and/or STS may have been underrepresented in our sample. Nonetheless, the demographic composition of our respondents, predominantly female, younger or recent graduates, and working in small-animal practice, reflects the general profile of veterinary professionals in North America.32–34 Further, given the broad inclusion criteria, it is possible that veterinary staff outside of clinical veterinary practice, such as animal laboratory staff, may have participated in the study; however, no participants self-identified as laboratory animal staff (ie, in the “other” category).
Several methodological and analytic limitations should be considered when interpreting these findings. Veterinary technicians and assistants were grouped together (into nonveterinarians) to allow ease of comparison to veterinarians. Despite some overlap in responsibilities between technicians and assistants (eg, performing dog handling/restraint during examinations), these roles have different education requirements, skill sets, and scopes of duties. Future studies should examine these roles separately to identify role-specific risk factors.
Certain individual predictors identified in other research (eg, gender, age, early career status) were not detected in our models, potentially due to limited variability within our sample. Further, dichotomizing ProQOL scores simplified interpretation and improved model stability given the sparse data in high BO and STS categories; however, this likely reduced sensitivity to subtle differences in subscale scores within the moderate to high range. Wide CIs for some associations also suggest imprecision, likely attributable to low event counts for some variables. Further, testing multiple predictors across 3 regression models may increase the chance of a type I error; thus, findings should be considered exploratory and hypothesis generating. There were also some findings that were difficult to interpret and warrant further investigation. For example, participants who reported an unknown bite history had higher odds of moderate/high CS (compared to reporting no bite history); the mechanism underlying this association is unclear. Further, the cross-sectional design of this study also limits the ability to infer causality between explanatory variables and ProQOL.
Despite these limitations, this study contributes novel insights into the ProQOL of veterinary professionals who routinely handle dogs. Most participants reported moderate levels of BO, STS, and CS. Individual factors, including staff role, stress-reducing certification, and certain personality traits, were associated with ProQOL, but handling techniques were not. Notably, Big Five traits were linked to BO and STS but not CS, suggesting that distinct factors may underlie protective versus risk outcomes. Understanding predictors of both positive and negative aspects of ProQOL is essential for guiding targeted, evidence-based interventions. Future research should incorporate broader measures, such as empathy and resilience; use mixed-methods approaches to capture complexity at individual and clinic levels; and validate these findings across more diverse veterinary populations.
https://avmajournals.avma.org/view/journals/ajvr/aop/ajvr.25.10.0355/ajvr.25.10.0355.xml
__________________________________________________________________________
FDA Approves Renewal of Canalevia-CA1, Jaguar Health’s Drug for Chemotherapy-Induced Diarrhea (CID) in Dogs
December 10, 2025
Topic: Company Update
Conditional approval extended through December 2026 for the treatment of CID in dogs
CID confirmatory effectiveness trial expected to conclude in February 2026, ahead of FDA’s June deadline – 51 dogs enrolled to date; ~49 more expected
SAN FRANCISCO, CA / ACCESS Newswire / December 10, 2025 / Jaguar Health, Inc. (NASDAQ:JAGX) today announced that the U.S. Food and Drug Administration (FDA) has granted renewal of the conditional approval for Canalevia-CA1 (crofelemer delayed-release tablets). Canalevia-CA1, the company’s prescription drug for the treatment of chemotherapy-induced diarrhea (CID) in dogs, is available from multiple leading veterinary distributors in the U.S., including Chewy. The renewal of conditional approval is in effect until December 21, 2026.
“Canalevia-CA1 is an important prescription drug for the veterinary community and the thousands of dogs experiencing CID. We’re very pleased about this conditional approval renewal, which, per FDA regulations, is for the fifth and final allowable year of conditional approval for Canalevia-CA1 for the CID indication in dogs. When an animal drug receives conditional approval, the FDA requires that a confirmatory trial take place within 5 years to provide the substantial evidence of effectiveness required for full approval of the drug for the indication,” said Dr. Michael Guy, D.V.M., M.S., Ph.D., Jaguar’s Vice President of Preclinical and Nonclinical Studies. “As announced, our full effectiveness study of Canalevia-CA1 for the treatment of CID in dogs is underway, and this recent conditional approval renewal was granted because we were able to demonstrate active progress toward generating this required data.”
“Diarrhea is a highly neglected and unmet medical need in dogs and people undergoing cancer treatment,” said Lisa Conte, Jaguar’s president and CEO. “Jaguar is deeply committed to supporting the quality of life of people and animals undergoing cancer treatment.”
About Conditional Approval and Full Approval
Canalevia-CA1 initially received conditional approval in December 2021 from the FDA for the treatment of CID in dogs. FDA’s conditional approval allows a drug company to legally promote, advertise and sell the animal drug for the labeled uses before proving it meets the “substantial evidence” standard of effectiveness for full approval. The conditional approval is valid for one year, with up to four annual renewals, for a total of five years of conditional approval. To receive a renewal from the FDA, the company must show active progress toward proving “substantial evidence of effectiveness” for full approval. After collecting the remaining effectiveness data, the company then applies to the FDA for full approval. The FDA reviews the application and, if appropriate, fully approves the drug.
About Canalevia®-CA1
Canalevia-CA1 contains crofelemer, Jaguar’s novel, oral plant-based drug sustainably harvested from the Croton lechleri tree, that modulates chloride channels in the gastrointestinal tract to reduce diarrhea. Importantly, Canalevia is not an antibiotic drug. The overuse and misuse of antibiotics, both in humans and animals, contribute to the development of bacteria that are resistant to antibiotics. Canalevia-CA1, currently conditionally approved by the FDA under application number 141-552, is a tablet that can be given orally twice a day for up to three days and can be used for home treatment of CID in dogs.
About Chemotherapy-induced Diarrhea (CID) in Dogs
According to the American Veterinary Medical Association, approximately 1 in 4 dogs will at some stage in their life develop cancer. Nearly half of dogs over 10 will develop cancer.1 According to the National Cancer Institute at the National Institutes of Health, roughly 6 million new cancer diagnoses are made in dogs yearly in the US.
Due to the increasing number of chemotherapeutic agents, chemotherapy is fast becoming the most widely used cancer treatment in veterinary medicine. Studies have found the incidence of CID to be one of the three most prevalent side effects in dogs undergoing cancer treatment,2 and managing side-effects such as diarrhea can be important to maintain successful cancer treatment. More than half of the US veterinarians who responded to a Jaguar-sponsored survey reported that CID interferes with their patients’ chemotherapy treatment plans, indicating an unmet need for an effective product for the treatment of CID.
Important Safety Information About Canalevia®-CA1
For oral use in dogs only. Not for use in humans. Keep Canalevia-CA1 (crofelemer delayed-release tablets) in a secure location out of reach of children and other animals. Consult a physician in case of accidental ingestion by humans. Do not use in dogs that have a known hypersensitivity to crofelemer. Prior to using Canalevia-CA1, rule out infectious etiologies of diarrhea. Canalevia-CA1 is a conditionally approved drug indicated for the treatment of chemotherapy-induced diarrhea in dogs. The most common adverse reactions included decreased appetite, decreased activity, dehydration, abdominal pain, and vomiting.
Caution: Federal law restricts this drug to use by or on the order of a licensed veterinarian. Use only as directed. It is a violation of Federal law to use this product other than as directed in the labeling. Conditionally approved by FDA pending a full demonstration of effectiveness under application number 141-552.
About the Jaguar Health Family of Companies
Jaguar Health, Inc. (Jaguar) is a commercial stage pharmaceuticals company focused on developing novel proprietary prescription medicines sustainably derived from plants from rainforest areas for people and animals with gastrointestinal distress, specifically associated with overactive bowel, which includes symptoms such as chronic debilitating diarrhea, urgency, bowel incontinence, and cramping pain. Jaguar family company Napo Pharmaceuticals (Napo) focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states. Jaguar family company Napo Therapeutics is an Italian corporation Jaguar established in Milan, Italy in 2021 focused on expanding crofelemer access in Europe and specifically for orphan diseases. Jaguar Animal Health is a Jaguar tradename. Magdalena Biosciences, a joint venture formed by Jaguar and Filament Health Corp. that emerged from Jaguar’s Entheogen Therapeutics Initiative (ETI), is focused on developing novel prescription medicines derived from plants for mental health indications.
For more information about:
Jaguar Health, visit https://jaguar.health
Napo Pharmaceuticals, visit napopharma.com
Napo Therapeutics, visit napotherapeutics.com
Magdalena Biosciences, visit magdalenabiosciences.com
Canalevia-CA1, visit canalevia.com
Forward-Looking Statements
Certain statements in this press release constitute “forward-looking statements.” These include statements regarding Jaguar’s expectation that the company’s ongoing trial will provide the substantial evidence of effectiveness required for full approval of the drug for treatment of CID in dogs and will conclude in February 2026 with an approximate total of 100 dogs having participated. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “aim,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions. The forward-looking statements in this release are only predictions. Jaguar has based these forward-looking statements largely on its current expectations and projections about future events. These forward-looking statements speak only as of the date of this release and are subject to a number of risks, uncertainties and assumptions, some of which cannot be predicted or quantified and some of which are beyond Jaguar’s control. Except as required by applicable law, Jaguar does not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
1 “Cancer in Pets.” American Veterinary Medical Association, 2021, https://www.avma.org/resources/pet-owners/petcare/cancer-pets
2 Mason SL, Grant IA, Elliott J, Cripps P, Blackwood L. Gastrointestinal toxicity after vincristine or cyclophosphamide administered with or without maropitant in dogs: a prospective randomised controlled study. J Small Anim Pract. 2014;55:391-398
__________________________________________________________________________
AVMA News I Computed tomographic features of renal neoplasms in dogs: introduction of the collateral vessel sign in veterinary medicine
Michele Occhiuzzi, DVM , Swan Specchi DVM, PhD, DACVR , Roberto Rabozzi DVM , Simone Teodori DVM , Caterina Puccinelli DVM, PhD , Erica Visconti DVM , Samuele Berti DVM, Laura Brizzi DVM, and Simonetta Citi DVM, PhD
December 5, 2025
Abstract
Objective
To assess the role of CT in detecting canine renal neoplasms and describe their imaging features.
Methods
32 dogs with histologically and cytologically confirmed renal tumors were included. Computed tomography scans were reviewed for qualitative parameters: lesions number, size, margins, enhancement pattern, mineralization, laterality, intrarenal location, hemorrhage, lymphadenopathy, pulmonary metastases and collateral vessels. Quantitative parameters included lesion diameter and pre- and postcontrast attenuation. Data distribution was tested with the Shapiro-Wilk test, and intergroup differences were evaluated using the Fisher exact test.
Results
Diagnoses comprised primary renal tumors—renal cell carcinoma (RCC; 12 of 32), sarcoma (2 of 32), hemangiosarcoma (1 of 32), primary lymphoma (1 of 32), and cystic adenoma (1 of 32)—as well as multicentric lymphoma (ML; 5 of 32) and metastatic lesions (10 of 32). Primary tumors were mainly unilateral masses, whereas ML and metastases appeared as bilateral nodules. Collateral vessels were significantly more frequent in RCC than in ML or metastases and were also associated with mass lesions rather than nodular lesions. Multicentric lymphoma showed more homogeneous postcontrast enhancement than primary tumors.
Conclusions
CT provides key information to differentiate canine renal tumor types. Collateral vessels may indicate RCC and are more prevalent in mass lesions; therefore, they should be considered in surgical planning. Canine lymphoma showed CT features consistent with human reports, although overlap among tumor subtypes reduced specificity.
Clinical Relevance
This study describes novel tomographic features of canine renal neoplasia and comprehensively summarizes their CT characteristics, aiding diagnosis and treatment planning.
Keywords: neoplasia; computed tomography; dog; collateral vessels; kidney
Renal neoplasms in dogs are considered uncommon, accounting for less than 1% of all neoplastic diseases and approximatively 2% of all malignant tumors.1,2 Primary renal neoplasms are extremely rare, reported in only 0.3% of oncologic canine patients.1,2 Among these, epithelial neoplasms, such as renal cell carcinoma (RCC, all acronyms are listed in Supplementary Table S1), transitional cell carcinoma, adenoma, and papilloma, have been described.2 Various types of sarcomas can also affect the kidneys, including hemangiosarcoma (HSA), renal sarcoma, leiomyosarcoma, malignant fibrous histiocytoma, spindle cell sarcoma, fibroleiomyosarcoma, lipoma/liposarcoma, and teratoma.3,4 Other primary renal tumors include nephroblastoma,2 renal interstitial cell tumors,5 renal cystadenocarcinoma—described as a unique hereditary syndrome in German Shepherd Dogs6—and primary renal lymphoma.1,2
Metastatic involvement of the kidneys is more common than primary neoplasms, particularly in cases of disseminated metastatic disease.7 Secondary renal involvement has been reported in epithelial neoplasms arising from the lungs,3 thyroid gland,7 mammary gland,3,7 and ovaries3 as well as in lymphosarcomas,3 mast cell tumors,3 and osteosarcomas.3,7,8
Ultrasonography (US) is widely used for the evaluation of renal lesions in dogs; however, it presents several limitations in discrimination and identification of such lesions. Specifically, US lacks distinctive features that allow reliable differentiation among various massive renal lesions, and a recent retrospective study7 reported that US failed to detect nodules that were isoechoic to the renal cortex and did not protrude from the renal profile.9 Moreover, the quality of US images decreases in large dogs due to the lower-frequency probes required.7
Computed tomography is less influenced by patient size, allows multiplanar image reconstruction, and is less operator dependent.10 Its use has been described in diagnostic protocols for renal neoplasia, offering a comprehensive evaluation that combines staging with high diagnostic accuracy.11 Compared to abdominal US, CT has been shown to be superior in detecting renal nodules.7
Currently, in veterinary medicine, no CT features have been identified that can reliably differentiate between benign and malignant renal neoplasms or among different primary tumor types.12 Therefore, fine-needle aspiration and biopsy remain essential for a definitive diagnosis.9 Fine-needle aspiration is generally considered a safe procedure, whereas renal biopsy is associated with complications, such as arteriovenous fistula formation, hemorrhage, hydronephrosis, infarction, thrombosis, and, in rare cases, death, with complication rates reported between 1% and 18%.13,14 Thus, the ability to differentiate renal lesions through diagnostic imaging could represent a safer and valuable approach.12
In human medicine, CT is considered useful in most urological oncology diagnostic algorithms.15 Several CT features, including lesion size, distribution, enhancement patterns, and the presence of collateral vessels (CVs), assist in lesion characterization.15,16
The aim of this study was to (1) describe the CT findings in dogs affected by renal neoplasia and (2) assess the potential utility of CT in differentiating among the various types of renal neoplastic lesions in dogs.
Methods
Selection and description of subjects
Inclusion criteria were dogs with a cytological or histopathological diagnosis of primary or metastatic renal neoplasia that underwent pre- and postcontrast CT examinations between 2017 and 2024. To minimize potential bias, only patients that were not receiving chemotherapy at the time of the CT examination were included. Data were retrieved from the medical archives of 4 veterinary institutions: Anicura Roma Sud Veterinary Hospital, the “Mario Modenato” Veterinary Teaching Hospital of the University of Pisa, Cetego Veterinary Center in Rome, and Anicura “I Portoni Rossi” Veterinary Hospital in Zola Predosa. Data collection was performed by 3 investigators (OM, VE, and BL) using the keywords “renal neoplasia,” “renal lesion,” and “kidney tumor.” Investigators planned to categorize the patients identified in the initial search according to their final cytological or histopathological diagnosis. Data use was approved by each institution; approval from an IACUC or ethical review board was not required for this retrospective study.
Data recording and analysis
Patient data—For each dog, age, sex, breed, and body weight were recorded. Additionally, the method used to obtain the cytological or histopathological diagnosis of both primary and metastatic lesions was reviewed. For patients undergoing nephrectomy, surgical data were also collected.
CT acquisition protocol—Computed tomography image acquisition was performed using 4 CT scanners: a 16-slice BrightSpeed (GE HealthCare), a 16-slice Revolution Act (GE HealthCare), a 32-slice Somatom Go Up (Siemens Healthineers), and a 128-slice Somatom Perspective (Siemens Healthineers).
All examinations were performed under general anesthesia with patients positioned in sternal recumbency. Acquisition parameters varied slightly depending on the equipment but included helical acquisition mode, 100 to 120 kVp, 150 to 250 mAs, 0.625- to 2.5-mm slice thickness, 0.3- to 2.5-mm reconstruction interval, 512 X 512 matrix, field of view tailored to patient size, and pitch values between 0.5 and 1.3.
Following precontrast image acquisition, iodinated contrast medium (iohexol; Omnipaque; 350 mg/mL) was administered using power injectors at a flow rate of 2 mL/s, with a maximum pressure of 100 lb/square inch and dosage of 600 mg/kg, and then postcontrast imaging was performed.
Images evaluation—Computed tomography images were reviewed using dedicated open-source DICOM software (Horos, version 4.0.0; Horos Project) with both transverse and multiplanar reconstructions. Image datasets were evaluated with soft tissue kernels using window settings of 500:50 (window width to window length).
Two radiologists, each with 7 years of experience (MO and EV), independently reviewed all CT scans. A third radiologist with over 20 years of experience (SC) subsequently verified the collected data. Discrepancies were resolved by consensus. Reviewers were blinded to the final diagnosis, and image sets were analyzed in random order to reduce potential bias.
For each kidney, both qualitative and quantitative CT characteristics were assessed as follows.
Qualitative features included lesion margins (well defined or ill defined) and location (cortical, medullary, or both). Quantitative features included maximum lesion diameter and lesion density. Lesion diameter was measured in transverse, sagittal, and coronal planes, with the largest value recorded. Based on these measurements, lesions were classified by size as nodules (< 3 cm) or masses (≥ 3 cm in maximum diameter). Lesion density was calculated by manually drawing 3 regions of interest (ROIs) within the lesion on both pre- and postcontrast images, avoiding cystic or necrotic areas. The median attenuation value (HU) and SD were recorded. The same procedure was applied to adjacent normal renal parenchyma for comparison. The number of renal lesions was recorded for each patient, and dogs were subsequently classified as having either a single lesion or multifocal lesions (defined as ≥ 2 lesions). The presence of intralesional mineralization and postcontrast enhancement patterns was also recorded. Enhancement was classified as homogeneous or heterogeneous. Enhancement heterogeneity was evaluated as described in the statistical analysis section. Other types of enhancement were evaluated for each case by the operators, and a consensus was subsequently reached after discussion. The same process was applied when assessing adjacent organs, retroperitoneal space, and lungs.
Collateral vessels were defined as vascular structures directly connected to the tumor regardless of diameter.16
As previously stated, patients were divided into groups—primary neoplasia, metastatic neoplasia, multicentric lymphoma (ML), RCC, mass-size lesions, and nodular-size lesions—and statistical analyses were performed.
Statistical analysis
Continuous data were tested for normality using the Shapiro-Wilk test and visual inspection. If distribution of the data was normal, then the mean and SD were calculated. Conversely, if the distribution of data was not normal, median and range were computed.
Categorical variables were reported as frequencies and percentages, and differences between groups were analyzed using the Fisher exact test. Continuous variables were assessed for normal distribution through visual inspection of bar graphs and histograms as well as using the Shapiro-Wilk test. Normally distributed variables are reported as the mean ± SD, whereas non–normally distributed variables are expressed as the median (range).
Enhancement heterogeneity was evaluated as follows. Two investigators (MO and CP) manually delineated a single, maximally sized ROI on postcontrast CT images of renal lesions by use of dedicated open-source DICOM software (Horos, version 4.0.0; Horos Project). Cystic or necrotic areas were carefully excluded. The SD of attenuation values within each ROI were recorded.
Enhancement pattern was subjectively classified by the same investigators as heterogeneous or homogeneous based on direct inspection of the postcontrast CT images. In instances of disagreement, a consensus classification was achieved.
The normality of SD values was assessed with the Shapiro-Wilk test. The Mann-Whitney U test was then applied to compare SD values between lesions with heterogeneous and homogeneous enhancement, testing the hypothesis that heterogeneously enhancing lesions would exhibit higher SD values.
A significance level of 5% was established for all statistical methods. Statistical analyses were performed by RR, MO, and CP using a commercially available statistical program (MedCalc Statistical Software, version 23.2.8; MedCalc Software Ltd).
Results
General
A total of 68 dogs were initially identified through database screening. Thirty-six dogs were excluded due to the absence of either a cytological or histopathological diagnosis or lack of available CT images.
Ultimately, 32 dogs met the inclusion criteria. Of these, 9 were female, and 23 were male. Among the females, 4 were neutered, whereas 14 of 23 males were castrated. The median age of the study population was 9.8 years (SD, 2.8).
The most frequently represented breeds were German Shepherd Dog (4 of 32) and Golden Retriever (3 of 32). Other breeds included Dachshund, Flat-Coated Retriever, Rottweiler, Chihuahua, Dobermann, Rhodesian Ridgeback, Cocker Spaniel, Shih Tzu, Lagotto Romagnolo, Maltese, Maremmano-Abruzzese Sheepdog, Labrador Retriever, Bernese Mountain Dog, Italian Mastiff, Siberian Husky, and Miniature Poodle. Additionally, 9 dogs were mixed breed.
Seventeen of 32 dogs were diagnosed with primary renal neoplasia, 5 with ML, and 10 with metastatic renal lesions. Within the primary renal neoplasia group, 12 had RCC, 2 had sarcomas, 1 had a primary renal HSA, 1 had a primary renal lymphoma, and 1 had an adenoma
In the metastatic group, renal lesions were observed in 3 cases of carcinoma (including 2 mammary gland carcinomas and 1 anal sac gland adenocarcinoma [AGASAC]), 3 cases of metastatic HSA, 1 adrenal pheochromocytoma, 1 axillary liposarcoma, 1 osteosarcoma, and 1 histiocytic sarcoma.
Ultrasound-guided fine-needle aspiration for cytological diagnosis was performed in 18 dogs, whereas 14 dogs underwent nephrectomy followed by histopathological examination of the excised lesions.
Regarding CT examinations, although minor differences in acquisition protocols were present, all scans included both baseline images and postcontrast-phase images.
Qualitative CT features
Out of the 32 dogs included in the study, 23 presented unilateral renal lesions, whereas 9 showed involvement of both kidneys. The left kidney was more frequently affected, with 13 lesions located in the left kidney and 10 in the right. All primary renal neoplasms were unilateral, whereas 4 of 5 lymphomas and 5 of 10 metastatic lesions involved both kidneys.
Seventeen of 32 dogs showed large masses, and 15 presented nodular lesions. Within the primary neoplasia group, most patients (15 of 17) had mass lesions, whereas dogs with metastatic renal neoplasia (P = .003) and lymphoma (P = .003) more commonly presented with nodular lesions (9 of 10 and 4 of 5, respectively; This observation was statistically significant.
Lesions were well marginated in most cases (28 of 32). Renal masses were often located near 1 kidney pole, compressing and distorting the adjacent normal renal parenchyma, affecting both cortex and medulla. In larger lesions, the affected kidney was almost entirely replaced by neoplastic tissue, making normal renal structures difficult to identify.
Nine of 32 lesions were located within the renal cortex; all were nodular lesions. Four of these were located at the corticomedullary junction. The renal capsule was deformed in all mass lesions and in the majority of nodular lesions (9 of 15).
Enhancement heterogeneity was significantly associated with higher SD values (P < .001). The median SD for heterogeneously enhancing lesions was 17.73 HU (range, 7.3 to 31.37 HU), whereas the median SD for homogeneously enhancing lesions was 10.57 HU (range, 5.97 to 15.07 HU). The visual assessments performed by the 2 operators were in complete agreement.
In addition to homogenous and heterogenous enhancement, different types of postcontrast enhancement were identified: ring enhancement and spotty postcontrast linear or amorphous strong hyperdensity (SPLASH sign).17
Renal cell carcinoma showed significantly more frequent heterogeneous postcontrast enhancement compared to ML (P = .001) and metastatic lesions (P = .03). The same observation has been noted when comparing RCC and metastatic lesions in terms of intralesional mineralization. However, no significant difference in intralesional mineralization was found when comparing RCC with other primary renal neoplasms or ML.
Moreover, 90% of RCC cases showed the presence of CVs, often consisting of multiple venous and arterial vessels surrounding the renal mass, with variable origin and size. Collateral vessels were significantly more frequent in RCC compared to metastatic neoplasia (P = .001) and ML (P = .001). Collateral vessels were also observed in 1 case of primary renal HSA, 1 case of ML, and 1 case of metastatic pheochromocytoma. Unfortunately, the Fisher exact test showed no statistically significant difference between RCC and other mass lesions regarding the presence of CV. Moreover, the occurrence of CV was significantly higher in mass-size lesions compared to nodular-size lesions (P < .001). Cases diagnosed as renal sarcoma and primary renal HSA showed mass lesions similar to RCC, without evidence of hemorrhage, pulmonary metastases, or renal lymphadenopathy. Primary renal lymphoma presented as a single, unilateral, well-marginated mass with homogeneous postcontrast enhancement. In the case of cystic adenoma, a single cyst-like mass was observed in the left kidney, with a thin, enhancing wall. The inner portion of the lesion showed a hyperdense, heterogeneous, nonenhancing structure, consistent with a sentinel clot sign, later confirmed as intralesional hemorrhage. Moderate retroperitoneal effusion was also present. No CT features were useful in distinguishing among different primary renal neoplasms, as the Fisher exact test revealed no statistically significant differences between these groups.
The ML group showed a significantly higher frequency of nodular (P = .003), multifocal (P < .001), and bilateral (P < .001) lesions, with homogeneous enhancement patterns (P < .001), compared to the primary renal neoplasia group. No statistically significant CT features were identified for differentiating ML from metastatic renal neoplasia.
Metastatic renal lesions were more frequently nodular (P < .001) and bilateral (P = .003) compared to primary neoplasia. Specifically, a single renal nodule was observed in 1 case of AGASAC. In cases diagnosed with metastatic HSA, all dogs exhibited the SPLASH sign, associated with a sentinel clot sign and irregular lesion margins, consistent with hemorrhagic rupture. In the metastatic pheochromocytoma case, a strongly hyperenhancing renal nodule was observed, with extensive invasion of the caudal vena cava and left renal vein originating from an adrenal mass.
In the case of metastatic liposarcoma, a heterogeneous axillary mass with negative attenuation values was detected, along with multifocal renal nodules in both kidneys, lungs, muscles, and axillary lymph nodes. These renal lesions also exhibited negative attenuation values on precontrast images. In the metastatic osteosarcoma case, bilateral, heterogeneously enhancing renal masses were identified. Multifocal renal nodules were observed in a Bernese Mountain Dog diagnosed with histiocytic sarcoma.
Analyses of adjacent organs, retroperitoneal space, and lungs revealed renal lymphadenopathy (lymph nodes > 5 mm in diameter)18 in 11 of 32 cases, retroperitoneal effusion in 7 of 32 cases, sentinel clot sign (hyperattenuating hematoma adjacent to a bleeding organ)19 in 7 of 32 cases, and suspected pulmonary metastasis (multifocal nodular lung lesions) in 6 of 32 cases.
Quantitative CT features
Continuous variables were lesion maximum diameter, CT attenuation, and patient age. The mean diameter was 8.1 cm (SD, 2.7) in the primary neoplasia group, 1.7 cm (SD, 0.7) in the metastatic renal lesions group, and 2.2 cm (SD, 1.6) in the ML group. Regarding CT density (HU), the median values in basal scan were 37.8 (range, 16.0 to 60.4) in primary neoplasia, 57.4 (range, 28.8 to 91.0) in ML, and 36.1 (range, −1.3 to 51.4) in metastatic neoplasia. The values in the nephrographic phase were 74.7 (range, 35.7 to 85.0) in primary neoplasia, 103.6 (range, 60.1 to 108.7) in ML, and 75.73 (range, 22.8 to 182.9) in metastatic neoplasia.
Discussion
Renal cell carcinoma was the most frequent primary renal tumor in our study, consistent with the incidence rates reported in the veterinary literature.20 In dogs diagnosed with RCC, the lesions were typically large, encapsulated masses, frequently located near one of the renal poles, in agreement with these well-documented characteristics of RCC.3,4,20,21 Histologically, RCC can present with different subtypes, including clear cell (accounting for approx 9% of cases and associated with a poorer prognosis), chromophobe, papillary, and multilocular cystic variants.21 These subtypes may coexist within the same lesion.3 In human medicine, postcontrast CT enhancement patterns have been correlated with different RCC subtypes.22 Unfortunately, in our study, information regarding RCC subtypes was unavailable.
Our results align with those of previous studies, indicating that RCC exhibits variable metastatic potential, ranging from moderate (16% to 34%) to high (70% to 75%) depending on the timing of diagnosis. Reported metastatic sites include the contralateral kidney (with approx 5% of cases presenting bilateral lesions), abdominal organs, omentum/peritoneum, lungs, bones, and renal lymph nodes.2,21 Pulmonary metastases have been documented in 18% to 48% of affected dogs, with an overall metastatic rate at the time of death reaching 69%.4,12,21
Regarding CT images, none of the evaluated findings in this study proved useful in distinguishing RCC from other primary renal tumors. Previous studies have attempted to establish a correlation between CT features and histopathological diagnosis of primary renal neoplasms. However, no statistically significant associations have been identified between imaging findings and histological diagnoses for the 3 most common primary renal tumors (RCC, HSA, and lymphoma).12
Heterogeneous enhancement in RCC, due to intralesional hemorrhage and necrosis, has been consistently reported, and our observations confirm these findings.12
Intralesional mineralization has been described in canine soft tissue tumors, such as extraskeletal osteosarcoma,23 dedifferentiated chondrosarcoma,24 liposarcoma,25 and isolated case reports.26,27 To the best of our knowledge, mineralization within RCC has not been previously documented in canine CT studies and overall remains an uncommon finding.12,23 Consequently, this observation may represent a novel discriminating factor in the diagnosis of RCC.
In human medicine, the presence of CV is considered a useful CT sign to differentiate RCC from other renal masses, such as fat-poor angiomyolipomas. Collateral vessels are associated with increased tumor angiogenesis and are more frequently observed in aggressive RCC subtypes, such as clear cell carcinoma. Mechanisms underlying neoangiogenesis in clear cell RCC have been well documented. Neoplastic cells frequently harbor mutations in the von Hippel–Lindau tumor suppressor gene, leading to dysregulation of hypoxia-inducible factors 1α and 2α, which, in turn, promote neoangiogenesis.28 Collateral vessels have been reported to provide a sensitivity, specificity, positive predictive value, and negative predictive value of 48.5%, 45.5%, 100%, and 95%, respectively, for RCC diagnosis.16 Additionally, in humans, RCCs are often detected as small renal masses with a diameter of less than 4 cm.16 In contrast, RCCs in dogs tend to be larger at the time of diagnosis, likely due to their subclinical progression, allowing for significant tumor growth before detection.4 Consequently, the increased angiogenic stimulus associated with larger neoplastic lesions may reduce the diagnostic reliability of CV as a specific CT feature for RCC.29
Therefore, CV may be related to both tumor size and histopathological diagnosis in light of the neoangiogenic mechanisms in RCC that are well characterized in human medicine. Our observations of reduced pulmonary metastasis and renal lymphadenopathy in dogs with primary renal sarcoma compared to those with RCC suggest that sarcomas may exhibit lower malignancy. However, due to the small number of sarcoma cases, statistical comparison with the RCC group was not feasible.
Primary renal HSA is considered a rare neoplasm in dogs compared to visceral or retroperitoneal forms. According to the available literature, dogs with renal HSA may have a more favorable prognosis than those with other forms, although prognosis worsens in the presence of retroperitoneal hemorrhage at diagnosis.30 Hemorrhage is infrequently reported, likely due to the anatomical location and specific histological characteristics of renal HSA.30 Our findings are consistent with these observations. On CT, renal HSA may demonstrate contrast enhancement in the corticomedullary phase, with gradual contrast expansion around vessels. This pattern likely reflects the histologic architecture of HSA, characterized by multiple cavernous, anastomosing vascular spaces.12 Additionally, renal HSA lesions may appear hypoattenuating on CT, likely due to inadequate vascular supply and extensive cystic areas containing hemorrhage and necrosis.31 Enhancement patterns are variable, ranging from marked vascular enhancement in early phases to heterogeneous or poor enhancement in delayed phases.31,32 In our study, the absence of large cavitary areas and marked perivascular enhancement differed from previous reports, although statistical evaluation was not possible due to the limited number of cases.
Renal adenomas are rarely described in dogs and may be difficult to distinguish from carcinoma based on imaging alone. Macroscopically, they usually appear as single, expansively growing nodules.3,33 Cystic adenomas are even more infrequently reported, with only isolated case descriptions available.34 In our study, CT was useful for presumptive diagnosis and surgical planning. To the best of our knowledge, this represents the first CT description of a cystic renal adenoma with intralesional hemorrhage in dogs.
In human medicine, renal lymphoma may present as multiple lesions, solitary masses, contiguous perirenal extension, or diffuse perirenal involvement.35 Mass pattern could present as homogeneously enhancing renal masses, a characteristic attributed to the absence of intralesional necrosis.35 Most commonly, patients present with multiple lesions, similar to the renal nodules observed in dogs with ML in our study. To our knowledge, this is the first description of CT findings of renal involvement in canine ML. However, CT was not helpful in distinguishing renal lesions associated with ML from those due to metastatic disease. Primary renal lymphoma remains poorly described in the veterinary literature.12 Bilateral, multiple renal masses have been reported as typical CT findings in dogs.12 In our study, primary renal lymphoma lesions resembled those described in mass human pattern, differing from previous canine reports. However, the small sample size precluded statistical analysis.
In a human autopsy study,36 renal metastases have been detected in 7% to 12% of cases, whereas diagnostic imaging studies report a lower incidence of approximately 0.9%.37–40 Metastases most commonly originate from primary tumors of the lung, gastrointestinal tract, breast, soft tissue, and thyroid.39 Various imaging modalities, including CT, MRI, and contrast-enhanced ultrasound, have been used to detect these lesions, with comparable diagnostic performance.40
Computed tomography findings of renal metastases are often nonspecific.37 They typically appear as bilateral, multifocal nodules with reduced contrast enhancement relative to normal renal parenchyma and preservation of renal contours.41 While intralesional mineralization has been described in metastatic mucoid carcinoma, papillary carcinoma, osteosarcoma, and chondrosarcoma,42 such findings are rare,37 and definitive diagnosis often requires biopsy.43
In veterinary medicine, renal metastases are rarely reported; only isolated cases have been described, including metastases from nasal chondrosarcoma and from extramedullary plasmacytoma of the oral cavity.44,45 In small animals, renal metastases typically present as bilateral, nodular lesions.7 Our observations confirm these findings. No CT findings were able to differentiate between different metastatic lesions. Notably, unlike in human patients, renal metastatic lesions in our canine cases frequently distorted renal contours and did not exhibit intralesional mineralization, even in cases of osteosarcoma.
Carcinoma was the most common primary tumor associated with renal metastasis in our cohort. In 1 dog with AGASAC, a breed predisposition (Cocker Spaniel) was observed, consistent with the known biological behavior of this tumor type.46
Our findings regarding extrapulmonary metastasis of HSA align with previous reports describing heterogeneous enhancement with central or peripheral linear or amorphous areas of contrast uptake, known as the SPLASH sign. This CT feature corresponds to a distinct pattern of contrast enhancement that may assist in the diagnostic assessment of HSA.17 It was described in a recent retrospective observational study,17 in which the authors identified this feature in 75% of renal HSA metastases. Its presence has been associated with neoangiogenesis, leading to the formation of new vascular channels, or with fluid-filled hemorrhagic intralesional areas.17
In contrast to metastatic HSA, no evidence of hemorrhage was observed in primary renal HSA in our study despite histological difficulties in differentiating primary from metastatic lesions.30 Primary HSA typically appeared as large, space-occupying masses with significant necrosis and a high risk of rupture, suggesting potential histopathological differences between primary and metastatic renal HSA. However, the limited number of HSA cases precluded statistical confirmation, highlighting the need for further research.
Computed tomography characteristics of adrenal pheochromocytomas have been described, and our findings are consistent with those reports.47 Histologically, pheochromocytomas may exhibit telangiectatic areas with distended vascular spaces and hemorrhage, which likely explain the contrast enhancement pattern observed in our case.48 Statistical evaluation was not possible due to the single case.
The CT appearance of liposarcomas in dogs has been described, although reports of metastatic liposarcomas are rare, with only 1 documented case of hepatic metastasis from an axillary liposarcoma.49,50 To the best of our knowledge, this is the first report of renal metastasis from a liposarcoma in a dog.
Our findings related to metastatic osteosarcoma and histiocytic sarcoma were consistent with previously reported data in veterinary literature.8,51
The limitations of our study include the following:
(1)Due to the retrospective design, a standardized CT acquisition protocol could not be applied; therefore, the analysis was based on the available postcontrast images.
(2)There was an absence of histopathological confirmation for all cases as well as a lack of immunohistochemical characterization; this fact prevented us from linking CV to specific subtypes of RCC that rely on hypoxia-driven mechanisms to promote neoangiogenesis.
(3)Although the sample included 32 dogs, the number of cases in each subgroup was small, and no control group was available for comparison.
(4)Although the operators were blinded to the final diagnosis, they were aware that all renal lesions included in the study were neoplastic due to the predetermined inclusion criteria, which may have introduced a potential source of bias.
(5)As previously mentioned, a considerable number of cases from the initial database search did not meet the inclusion criteria; this limitation is likely related to the referral-based structure of the diagnostic imaging sections of the participating veterinary institutions.
Our study demonstrated that, in canine patients with renal neoplasia, certain CT features showed statistically significant differences between groups. Multicentric lymphoma and metastatic neoplasia more frequently presented as bilateral, nodular renal lesions, whereas primary tumors were more commonly characterized as unilateral masses. Among primary tumors, RCC was significantly more likely to exhibit CVs compared to lymphoma and metastatic neoplasia. Collateral vessels are a novel tomographic sign that has not previously been described in dogs. Although this finding reached statistical significance when comparing RCC with other neoplastic entities, a significant difference was likewise observed between mass-size and nodular-size lesions in terms of CV occurrence.
Further studies with larger sample sizes are warranted to determine whether the presence of CV is truly associated with the type of neoplasia or is simply a consequence of lesion size. Nonetheless, considering that nephrectomy remains an effective treatment option in metastatic-free patients,2,4,30 the detection of CV on CT could be a valuable tool for presurgical planning. Compared to metastatic neoplasia, RCC more frequently exhibited intralesional mineralization, a feature that should therefore be considered in the differential diagnosis of mineralized renal masses.
In patients with ML, postcontrast enhancement was more commonly homogenous compared to primary renal neoplasms. However, no CT features demonstrated statistical significance in differentiating ML from metastatic renal lesions. The description and characterization of secondary renal tumors provided by this study may serve as a foundation for future research aimed at improving diagnostic accuracy.
In conclusion, while CT can assist in guiding the differential diagnosis of certain renal neoplastic lesions in dogs, definitive diagnosis still requires fine-needle aspiration and histopathological evaluation. Our research introduces novel tomographic signs, such as CVs, and provides a detailed description of CT features in canine renal neoplasia.
Supplementary Materials
Supplementary materials are posted online at the journal website: avmajournals.avma.org.
Acknowledgments
The authors thank Fidelia Fasano, DVM, for her help with data acquisition at Anicura Roma Sud Veterinary Hospital.
Disclosures
The authors have nothing to disclose. No AI-assisted technologies were used in the composition of this manuscript.
https://avmajournals.avma.org/view/journals/ajvr/aop/ajvr.25.09.0342/ajvr.25.09.0342.xml
________________________________________________________________________________________________
DVM 360 | Understanding oncology diagnostics and therapeutics
Multiple approaches to cancer treatment can be initiated by primary care veterinarians.
December 3, 2025
Author(s)Craig A. Clifford DVM, MS, DACVIM (Oncology), Christine Mullin VMD, DACVIM (Oncology)
What can primary care veterinarians do to provide care to patients with cancer? These approaches can help guide treatment.
Staging is a critical step in cancer patient management, but it does not always require referral to a specialist to be performed effectively. Most staging procedures—such as complete blood count, serum chemistry panel, cytologic evaluation of regional lymph nodes, and 3-view thoracic radiographs—are standard diagnostic tests readily available in general practice.
These tools provide essential information about the extent of disease, metastatic potential, and overall patient health, guiding both treatment decisions and client expectations. For example, aspirating a regional lymph node in a dog with a mast cell tumor or obtaining thoracic radiographs in a soft tissue sarcoma or osteosarcoma case are impactful yet straightforward steps that can be performed in-house. When these results accompany a referral, they not only streamline case management but also reduce client costs and improve continuity of care. By recognizing that cancer staging is based upon the same diagnostic principles used in primary care, veterinarians can confidently contribute to accurate diagnosis, timely treatment planning, and collaborative oncology care.
Key tests for primary care
- Lymphoma: Complete blood count (CBC), serum chemistry, consider 3-view thoracic radiographs with radiologist review
- Mast cell tumor: CBC, serum chemistry, consider regional lymph node cytology
- Soft tissue sarcoma: CBC, serum chemistry, 3-view thoracic radiographs with radiologist review
- Osteosarcoma: CBC, serum chemistry, radiographs of affected limb, 3-view thoracic radiographs with radiologist review
- Melanoma: Mass measurement and photo, CBC, serum chemistry, 3-view thoracic radiographs with radiologist review, regional lymph node cytology
Clinical takeaways
Performing baseline staging in-house supports faster case turnover, more informed prognostication, and smoother communication with oncology referral centers. Most initial cancer staging can be efficiently and accurately performed by primary care veterinarians, expediting or even in some cases replacing oncology referral and reducing client cost.
Diagnosing lymphoma with a blood test
The IDEXX Cancer Dx is a new blood-based screening/diagnostic panel aimed at detecting canine lymphoma from serum or whole blood. It reports the presence or absence of lymphoma-associated circulating biomarkers and, when positive, can provide a B- vs T-cell phenotype classification in some cases.1
Key performance numbers2:
- Sensitivity: ~79% for dogs with lymphoma in IDEXX’s internal validation.
- Specificity: ~99% versus healthy controls and dogs with inflammatory disease/other cancers in the same internal dataset.
Practical details:
- Sample: single blood draw (serum or whole blood). Interference from mild–moderate hemolysis, lipemia, or icterus is reportedly minimal.
- Intended use cases highlighted by IDEXX:
- Dogs suspected of having lymphoma
- Cancer screening for apparently healthy “at-risk” dogs (IDEXX suggests integration within wellness panels for older dogs).
- Cost/rollout: IDEXX promotes the test as low-cost (advertised as starting around US$15 as an add-on) and has been available in the US and Canada as of March 2025.1
How to use it in primary care:
- If lymphoma is suspected clinically (lymphadenopathy, constitutional signs, cytology suspicious): Cancer Dx can be run on the blood sample as a noninvasive adjunct, but positive results still require confirmation with cytology/biopsy/flow/PCR for antigen receptor rearrangement (PARR) as clinically indicated. A negative result does not rule out lymphoma because sensitivity is <100%.
- If used for screening (as an add-on in older / at-risk dogs): discuss with owners that the test increases the chance of detecting lymphoma earlier, but is not a guarantee—explain the possibility of false negatives and what you’d do if the test is positive (confirmatory diagnostics, staging conversation).
Clinical takeaways
The IDEXX Cancer Dx is a practical, low-cost blood test that helps detect canine lymphoma earlier and in some cases supplies phenotyping information useful for initial case planning. It’s a reasonable adjunct for dogs with clinical suspicion and an attractive screening add-on for older/at-risk dogs — but it is not diagnostic by itself. Use positive results to prompt confirmatory diagnostics such as cytology/biopsy/flow cytometry/PARR, and use clinical judgment for negative results because sensitivity is not 100%. Consider the current evidence base (IDEXX internal validation data only) and watch for independent peer-reviewed studies as they emerge.
Immunophenotyping Canine Lymphoma
Immunophenotyping (determining B-cell vs. T-cell origin) is now considered a standard part of lymphoma diagnosis because it directly influences prognosis and treatment expectations. Multiple studies confirm that dogs with T-cell lymphoma have shorter survival times and lower response rates to standard CHOP chemotherapy compared to dogs with B-cell lymphoma.
- In one study, T-cell lymphoma had only a ~50% response rate to single-agent doxorubicin, versus nearly 100% for B-cell lymphoma.3
- T-cell protocols incorporating alkylating agents such as lomustine (CCNU) have shown partial responses in ~60% of cases and complete responses in 17%.4
- Modified multiagent protocols (eg, L-MOPP, CCNU-based CHOP variants) may offer benefit, but toxicity and practicality limit their use in general practice.5
- A prospective study comparing CHOP +/- the investigational agent AT-005 in T-cell lymphoma demonstrated similar response rates (~65% CR) and median progression-free survival (64–103 days), confirming that outcomes remain inferior to B-cell lymphoma.6
Methods for immunophenotyping
Flow cytometry (preferred):
- Most informative test of all those available. Confirms B-cell vs. T-cell, but also provides additional prognostic markers that can indicate grade and expected behavior
- Can be performed on tissue aspirate (ie, lymph node, organ) or fluid (ie, blood, effusions, CSF)
- Requires live cells in media and overnight shipping
- Available at multiple academic and reference labs
PARR:
- Can be performed on both cytology and biopsy samples
- Only confirms B-cell vs. T-cell status, does not provide grade
- Available at most academic and reference labs
Immunocytochemistry (ICC):
- Can be performed on cytology slides
- Somewhat subjective
- Available at some academic and reference labs
Clinical takeaways
Primary care veterinarians can play a crucial role by determining lymphoma immunophenotype before referral and treatment initiation, allowing the care team and pet owners to have a more informed and directed conversation about treatment options and expected prognosis.
B-cell: CHOP-based therapy remains standard, potentially good long-term prognosis.
T-cell: Expect shorter remission; consider referral or alternate protocols.
Cytologic Grading of Mast Cell Tumors (MCTs)
Traditional histologic grading remains the gold standard, but cytologic grading is validated as a noninvasive, predictive tool when tissue biopsy isn’t feasible or clinically indicated. Studies have shown that specific cytologic features—including mitotic figures, multinucleation, nuclear atypia, and karyomegaly—strongly correlate with histologic grade and outcome.
Cytologic grading predicted histologic grade in 94% of cases.7
Cytologic grading carries an 88% sensitivity and 94% specificity relative to histopathology.8
Compared to low-grade tumors , cytologically high-grade MCTs are 25–39 times more likely to result in death within 2 years.7
Clinical takeaways
For general practitioners, cytologic grading provides a rapid, low-cost prognostic tool that can guide surgical planning and inform prognosis. Communicate with reference laboratory to ensure that cytologic grading can be provided for MCT submissions.
Noninvasive diagnosis of Transitional Cell Carcinoma (TCC)
The BRAF mutation test (CADET BRAF Mutation Detection Assay) represents one of the most impactful molecular diagnostics in veterinary oncology. Independent studieshave identified a single-point mutation (V595E) in the canine BRAF gene that is present in ~85% of TCC/urothelial carcinoma (UC) cases, but absent in benign or inflammatory bladder conditions. 9,10
This assay detects the mutant DNA in urine sediment, allowing:
- Noninvasive diagnosis without aspirate, cystoscopy, or surgical biopsy
- Early detection—often months before clinical signs
- Monitoring for response to treatment and early detection of recurrence
A secondary test now detects an additional ~10% of previously negative cases. The test is offered through Antech Diagnostics (BRAF Plus; Sentinel Biomedical platform).
Clinical takeaways
- Consider this test for any dog with persistent hematuria, pollakiuria, or suspicion of a urinary tumor.
- Submit a free-catch or catheterized urine sample for CADET BRAF testing before pursuing more invasive diagnostics such as prostatic wash for cytology, cystoscopy, or biopsy.
- This test eliminates the need for needle aspiration of urinary tumors, a known risk for tumor seeding.
- This test is 100% specific, so can be performed in dogs with known or suspected urinary tract infection (UTI) without concern for a false positive result.
Oncologic therapies available in primary care practice
Use of verdinexor (Laverdia-CA1; Dechra)
Verdinexor is an oral anticancer medication conditionally approved by the FDA for the treatment of lymphoma in dogs. It belongs to a novel class of compounds known as Selective Inhibitors of Nuclear Export (SINE), which work by trapping tumor suppressor proteins inside the nucleus, thereby restoring normal control of cell growth and promoting cancer cell death.
For the primary care veterinarian, verdinexor offers an important at-home treatment option for canine lymphoma—particularly when referral for multiagent chemotherapy is not possible, or when a client prefers palliative care that still provides measurable clinical benefit.
Efficacy and clinical use
In a multicenter phase II study of dogs with both B-cell and T-cell lymphoma, verdinexor achieved a clinical benefit rate of 55% (stable disease or better), with a median duration of benefit of 71 days (range 21–273 days). Notably, T-cell lymphoma cases, which are often less responsive to traditional chemotherapy, still showed a 71% clinical benefit rate.11 Commonly reported adverse effects are mild and include transient loss of appetite, lethargy, or gastrointestinal upset, which can often be managed symptomatically.
This makes verdinexor a reasonable monotherapy option for:
- Dogs that cannot undergo intravenous chemotherapy.
- Patients that have relapsed after CHOP-based treatment.
- Owners seeking palliative oral therapy that maintains quality of life.
How SINE drugs work with other chemotherapy agents
In addition to its single-agent activity, the SINE drug class has shown synergistic and sensitizing effects when combined with other chemotherapeutics such as doxorubicin and platinum-based agents. These effects may help:
- Sensitize lymphoma cells to respond better to chemotherapy.
- Reverse or delay drug resistance that develops after cytotoxic treatment.
Verdinexor vs prednisone: Impact on drug resistance
Historically, prednisone has been used for palliative treatment of lymphoma, but glucocorticoids can induce multi-drug resistance (MDR) by increasing P-glycoprotein expression in lymphoma cells—reducing the efficacy of subsequent chemotherapy and worsening prognosis.12
In contrast, verdinexor is not a substrate for P-glycoprotein, meaning it should not promote MDR or interfere with future chemotherapy response. In clinical studies, dogs receiving prednisone alone survived an average of 4–7 weeks, whereas verdinexor therapy offers a longer median benefit with potentially improved quality of life.11
Potential novel uses
Cutaneous (epitheliotropic) lymphoma is a challenging disease to treat, with lomustine & prednisone considered the current standard of care. New data suggests that verdinexor has efficacy vs. this form and may serve as a novel second line agent.13,14
Clinical takeaways
- Verdinexor oral tablet can be prescribed and monitored in a general practice setting.
- It provides a targeted, noncytotoxic option for dogs with newly diagnosed or relapsed lymphoma.
- It can be considered a bridge between comfort care and referral for chemotherapy.
- Technicians play a key role in client education—reviewing safe handling, side-effect monitoring, and adherence.
Tigilanol tiglate (Stelfonta; Virbac)—A localized therapy for canine mast cell tumors (MCT)
Tigilanol tiglate is a novel anticancer protein kinase C activator that is FDA-approved for the treatment of certain canine MCTs. Tigilanol tiglate was originally isolated from the seed of the Australian tree Fontainea picrosperma and is now formulated for intratumoral injection.15 Tigilanol tiglate is approved for dogs with all grades of non-metastatic MCTs, including:
- Any cutaneous MCT
- Subcutaneous MCTs located at or distal to the elbow or the hock
- Note: Tumors must be £10 cm3 in volume and accessible for intratumoral injection
How it Works
- Tigilanol tiglate (1 mg/ml) is injected via a single puncture site into the tumor, then the dose is distributed by fanning the needle throughout the tumor mass.
- Dose to be injected is calculated based upon tumor volume (Modified Ellipsoid Method, in cm3): https://stelfonta.com/how-to-calculate-correct-stelfonta-dose/
- Once injected, tigilanol tiglate induces a rapid and localized inflammatory response and destruction of local blood supply to the tumor primarily via ischemic necrosis.
- Healing of the affected area occurs with minimal scar formation.
- Most wounds heal within 4-6 weeks; full healing was observed within 3 months in 98.2% of cases16
Efficacy
In a prospective study, a single tigilanol tiglate treatment resulted in 75% complete response (CR) by 28 days, with that CR maintained in 94% of dogs by 84 days and 89% of dogs by 12 months.17 Dogs not achieving a complete response with the first treatment can receive a second treatment, which results in an improved overall CR rate of 88%. Another more recent study (showed similar results—75% CR rate after a single treatment, with that CR lasting at least 1 year in 64% of dogs.18
Adverse Effects
Adverse effects of tigilanol tiglate are typically low grade, transient, and directly associated with the mode of action. These include:
- Wound formation (possibly extensive) following tumor necrosis and slough
- Localized pain, swelling, erythema, and bruising at the tumor site
- Lameness in a treated limb
- Regional lymph node enlargement
Suggested visit schedule:
- Day 0 – injection
- Day 2 or 3 – check site to confirm necrosis occurring
- Day 7 – assess wound
- Day 28 – reassess wound
- Further visits will be dependent upon patient response and wound healing
Tricks of the trade
- Perform cytologic grading since histopathology will not be obtained
- If cytologically high grade, expect lower response rate
- Systemic therapy is warranted for high grade MCTs
- Perform regional lymph node cytology to confirm nonmetastatic prior to administering tigilanol tiglate.
- Bandaging and Elizabethan collars are not recommended, as the dog’s licking helps remove necrotic tumor tissue from the site.
- The treatment site is considered a “clean” wound, so antibiotics are likewise not commonly needed.
- Consider previsit anti-anxiety and analgesic drugs and/or injectable sedation for treatment of very active or agitated patients.
- Wear personal protective equipment including gloves, protective eye wear, and a lab coat or gown when handling and administering this product.
- Caution is required during treatment to avoid accidental self-injection, which may cause severe wound formation.
Clinical takeaways
Tigilanol tiglate is not for every case, but can be a viable nonsurgical option for the following cases:
- Dogs with multiple low grade MCT
- Dog whose owner whoelectsagainst surgery
- Dogs with a tumor in a challenging location to obtain a complete surgical removal
References
- McCafferty C. Affordable early detection test for canine lymphoma to hit the market. dvm360. January 27, 2025. Accessed December 3, 2025. https://www.dvm360.com/view/affordable-early-detection-test-for-canine-lymphoma-to-hit-the-market
- Connell D, Drake C, Michael H, Nascimento, Stuart S, Lyons H. Performance of IDEXX Cancer Dx testing for detection of lymphoma and corresponding phenotype in dogs. IDEXX. 2025. Accessed December 3, 2025. www.idexx.com/files/cancer-dx-white-paper-en-na.pdf
- Beaver LM, Strottner G, Klein MK. Response rate after administration of a single dose of doxorubicin in dogs with B-cell or T-cell lymphoma: 41 cases (2006-2008). J Am Vet Med Assoc. 2010;237(9):1052–1055. doi:10.2460/javma.237.9.1052
- Brodsky EM, Maudlin GN, Lachowicz JL, Post GS. Asparaginase and MOPP treatment of dogs with lymphoma. J Vet Intern Med. 2009;23(3):578–584. doi:10.1111/j.1939-1676.2009.0289.x
- Rebhun RB, Kent MS, Borrofka SAEB, Frazier S, Skorupski K, Rodriguez CO. CHOP chemotherapy for the treatment of canine multicentric T-cell lymphoma. Vet Comp Oncol. 2011;9(1):38–44. doi: 10.1111/j.1476-5829.2010.00230.x
- Musser ML, Clifford CA, Bergman PJ, et al. Randomised trial evaluating chemotherapy alone or chemotherapy and a novel monoclonal antibody for canine T-cell lymphoma: a multicentre US study. Vet Rec Open. 2022;9(1):e49. doi:10.1002/vro2.49
- Camus MS, Priest HL, Koehler JW, et al. Cytologic criteria for mast cell tumor grading in dogs with evaluation of clinical outcome. Vet Pathol. 2016;53(6):1117–1123. doi:10.1177/0300985816638721
- Scarpa F, Sabattini S, Bettini G. Cytological grading of canine cutaneous mast cell tumours. Vet Comp Oncol. 2016;14(3):245–251. doi:10.1111/vco.12090.
- Hanazono K, Fukumoto K, Endo Y, et al. Ultrasonographic findings related to prognosis in canine transitional cell carcinoma. Veterinary Radiology and Ultrasound. 2013;55(1):79–84. doi:10.1111/vru.12085
- Mochizuki H, Kennedy K, Shapiro SG, Breen M. BRAF mutations in canine cancers. PLoS One. 2015;10(6):e0129534.doi: 10.1371/journal.pone.0129534.
- Sadowski AR, Gardner HL, Borgatti, A, et al. Phase II study of the oral selective inhibitor of nuclear export (SINE) KPT-335 (verdinexor) in dogs with lymphoma. BMC Vet Res. 2018;14(1):250. doi:10.1186/s12917-018-1587-9
- Bergman PJ, Ogilvie GK, Powers BE. Monoclonal Antibody C219 Immunohistochemistry Against P-Glycoprotein: Sequential Analysis and Predictive Ability in Dogs With Lymphoma. J Vet Intern Med. 1996;10(6):354-359. doi:10.1111/j.1939-1676.1996.tb02080.x
- Vlodaver EM, Keating MK, Bidot WA, Bruyette DS, Rosenkrantz WS, et al. Open-label pilot study: Efficacy of verdinexor for naïve canine epitheliotropic cutaneous T-cell lymphoma. Vet Dermatol. 2024;35(1):536-546. doi:10.1111/vde.13280
- Grady JL, Gencher J, Adrianowycz S, Martinez-Romero G. Clinical remission of cutaneous lymphoma in a dog treated with verdinexor. J Am Anim Hosp Assoc. 2024;60(5):223-226. doi:10.5326/JAAHA-MS-7443
- Miller J, et al. Dose characterization of the investigational anticancer drug tigilanol tiglate (EBC-46) in the local treatment of canine mast cell tumors. Front Vet Sci. 2019;6:106. doi:10.3389/fvets.2019.00106
- Reddell P, De Ridder TR, Morton JM, et al. Wound formation, wound size, and progression of wound healing after intratumoral treatment of mast cell tumors in dogs with tigilanol tiglate. J Vet Intern Med. 2021;35(1):430-441. doi:10.1111/jvim.16009
- Jones PD, Campbell JE, Brown G, Johannes CM, Reddell P. Recurrence-free interval 12 months after local treatment of mast cell tumors in dogs using intratumoral injection of tigilanol tiglate. J Vet Intern Med. 2021;35(1):451–455. doi:10.1111/jvim.16018
- Musser ML,Jones PD,Goodson TL,Roof E,Johannes CM.Response to tigilanol tiglate in dogs with mast cell tumors. J Vet Intern Med. 2024; 38(6):3162-3169. doi:10.1111/jvim.17211
https://www.dvm360.com/view/understanding-oncology-diagnostics-and-therapeutics
_________________________________________________________________________________________________
American Humane Society I Cold-Weather Pet Tips
People who live in areas experiencing cold weather and winter storms need to take extra precautions to keep their animals safe. Pets left to fend for themselves in cold weather are susceptible to injury and death. Here are some simple tips from American Humane:
Be Prepared
- Plan ahead and pay attention to cold-weather warnings.
- Unless significant power outages are experienced, most cold-weather episodes and winter storms are “shelter in place” events, so pet care needs should be planned for in the home. Keep your pet preparedness kit well-stocked and ready — in a winter storm, you may not be able to leave your home for several days.
- Leave your pets’ coats a little longer in the winter to provide more warmth. That summer “short cut” from your groomer should be avoided during cold weather. If you have short-haired breeds, consider getting them a coat or sweater that covers them from neck to tail and around the abdomen.
Winter Pet Care
- When you bathe your dogs in cold weather, make sure they are completely dry before taking them outside for a romp or walk.
- When walking your dogs during bad weather, keep them on leash. It’s easier for a dog to become lost in winter storm conditions — more dogs are lost during the winter than during any other season. (And don’t forget to microchip and put ID tags on your dogs and cats!)
- Leash your pets if you have frozen ponds, lakes or rivers nearby, as loose pets can break through ice and quickly succumb to hypothermia before trained ice-rescue personnel can arrive. Never try an ice rescue of a pet yourself — leave that to trained professionals.
- When you are working on housebreaking your new puppy, remember that puppies are more susceptible to cold than are adult dogs. In cold conditions or bad weather, you may need to opt for paper training your new pet rather than taking the pup outside.
- Keep your pets inside, both during the day and night. Just because they have fur doesn’t mean they can withstand cold temperatures.
- If dogs are left outside, they should have a draft-free shelter large enough to stand and turn around in, yet small enough to retain body heat. Use a layer of straw or other bedding material to help insulate them against the cold. Make sure the entrance to the shelter faces away from the direction of incoming wind and snow.
- Keep your cats indoors. Cats can freeze in cold weather without shelter. Sometimes cats left outdoors in cold weather seek shelter and heat under the hoods of automobiles and are injured or killed when the ignition is turned on. Banging loudly on the hood of your car a few times before starting the engine will help avoid a tragic situation. (This is true for wild animals in cold weather as well).
- When taking your pets out for a bathroom break, stay with them. If it’s too cold for you to stand outside, it is probably also too cold for your pets.
- Level Up Your Pet Parenting Skills With Our Free Guide!
- Unleash the ultimate pet parenting guide! Sign up now to receive our exclusive guide filled with expert advice from American Humane Society’s animal care and veterinary specialists. Discover essential training techniques, vital pet care tips, and much more.
Precautions for Outdoor Pets
- If your pet is outside during cold weather:
- Remember that staying warm requires extra calories. Outdoor animals typically need more calories in the winter, so feed them accordingly when the temperature drops. Talk to your veterinarian for advice on proper diet.
- Watch your pet’s outside fresh-water bowl. If it is not heated, you may need to refresh it more often as it freezes in cold weather.
- Salt and de-icers: Many pets like to go outside to romp and stomp in the snow, but many people use powerful salt and chemicals on their sidewalks to combat ice buildup. Thoroughly clean your pets’ paws, legs and abdomen after they have been outside, to prevent ingestion of toxic substances and to prevent their pads from becoming dry and irritated. Signs of toxic ingestion include excessive drooling, vomiting and depression.
- Ice and snow: When you let your pets in from a walk or a romp outside, make sure to wipe their paws and undersides — get those ice balls off as soon as possible, as they can cause frostbite. After being outside, check your pets’ paws, ears and tail for frostbite. Frostbitten skin usually appears pale or gray and can be treated by wrapping the area in a dry towel to gradually warm the area. Check with your veterinarian if you suspect frostbite.
- Use nontoxic antifreeze. Antifreeze is great-tasting to pets, but even a very small amount ingested can be deadly. Look for “safe” nontoxic antifreeze, consider using products that contain propylene glycol rather than ethylene glycol, and make sure all spills are cleaned up immediately and thoroughly. Contact your veterinarian immediately if you suspect your pets have ingested any antifreeze!
________________________________________________________________________________________________
AVMA Publications I Effectiveness of Multiwavelength Locked System laser therapy for calcinosis cutis in 3 dogs: a case series
Federica Gesuete, DVM, Filippo Ferri DVM, DECVIM, Sara Muñoz Declara DVM, DECVD, Luca Luciani DVM, Federica Tirrito DVM, DECVN, and Giordana Zanna DVM, PhD, DECVD
Abstract
Objective
To describe the clinical effects of photobiomodulation (PBM) therapy using a dual-wavelength near-infrared laser (Multiwave Locked System; Asa Srl) as adjunctive treatment in dogs with calcinosis cutis (CC).
Animals
3 client-owned dogs presented to 2 referral veterinary hospitals from 2022 through 2023 were included. Cases were identified retrospectively via medical record review. Inclusion criteria were clinical and cytological features consistent with CC and histopathological examination when available, diagnosis of hyperadrenocorticism (HAC) based on clinical signs and diagnostic workup, availability of sufficient clinical records and photographic documentation, and owner consent for PBM therapy. Photobiomodulation was applied following a standardized protocol in scanning mode with a 2-cm collimated handpiece (approx 3-cm2 spot): 3 sessions during week 1, every 72 hours in week 2, and then weekly with 4 J/cm2 at the first session and 2.03 J/cm2 thereafter.
Clinical Presentation
Included dogs were a 5-year-old male Labrador Retriever and an 8-year-old male English Bulldog with naturally occurring HAC and a 3-year-old female German Shepherd with iatrogenic HAC. All presented with dermatologic lesions compatible with CC (erythematous-crusted plaques, erosive-ulcerative lesions, and/or papules).
Results
2 dogs achieved complete lesion resolution with hair regrowth within 7 to 12 weeks; the third showed partial improvement. No adverse events occurred.
Clinical Relevance
Findings, although based on a limited number of cases, suggest a potential supportive role for PBM in the management of CC. Controlled studies are warranted to establish standardized treatment parameters and clarify its role in CC.
Calcinosis cutis (CC) is an uncommon condition characterized by the deposition of inorganic, insoluble minerals in the dermis, subcutis, or, rarely, epidermis. It generally involves the accumulation of cations (ie, calcium) and anions (ie, phosphate or carbonate), which are typically in equilibrium in the extracellular fluid within the collagen fibers and elastin of the dermis.1,2
Based on their etiology, 4 subtypes of CC have been described: dystrophic, metastatic, idiopathic, and iatrogenic. Dystrophic is the most common form of CC, often associated with hyperadrenocorticism (HAC), either naturally occurring or iatrogenic.1–3 Inflammation and hypoxia are believed to contribute to tissue damage, acting as a focal point for dystrophic calcification.4 Despite this, there is limited evidence of effective treatments for CC in veterinary medicine.5 Recently, fluorescent light energy biomodulation, a form of photobiomodulation (PBM) consisting of a topical component containing specific chromophores that require activation by a light-emitting diode lamp, has been demonstrated to be useful in managing pyoderma associated with CC in a dog.6
In general, experimental and clinical studies suggest that PBM may beneficially influence biological tissues by reducing inflammation and improving tissue microcirculation.7–8 Furthermore, some in vitro and in vivo studies9–10 have reported potential effects on bacterial growth, although evidence in veterinary clinical settings remains limited.
This case series describes the effectiveness of PBM using a high-power, dual-wavelength near-infrared (NIR) laser source (Multiwavelength Locked System [MLS] laser; ASA Srl) as adjunctive therapy in producing a regression of skin lesions associated with CC in 3 dogs.
Methods
Case selection
This descriptive case series included 3 client-owned dogs evaluated for dermatological lesions consistent with CC from 2022 through 2023. Cases were identified retrospectively by reviewing the medical records of 2 veterinary facilities in Italy: AniCura Istituto Veterinario di Novara (Granozzo con Monticello, Novara; cases 1 and 2) and AniCura Portoni Rossi (Zola Predosa, Bologna; case 3).
Inclusion criteria were (1) clinical presentation compatible with CC supported by cytological or histopathological findings when available, (2) diagnosis of HAC, (3) availability of sufficient clinical records and photographic documentation, and (4) owner consent for adjunctive PBM therapy. Exclusion criteria included insufficient medical documentation or use of a nonstandardized PBM protocol.
All procedures were performed with owner informed consent and in compliance with patient welfare. No sedation was required for diagnostic or therapeutic procedures.
Photobiomodulation therapy was standardized across cases using a high-power, dual-wavelength NIR laser with 2 different wavelengths within the NIR spectrum: 808 nm in continuous or frequency-modulated mode and 905 nm in pulsed mode. Treatments were applied in scanning mode with a 2-cm collimated handpiece (approx 3-cm2 spot size). Each affected area was divided into multiple sections to ensure uniform dosing. The first session delivered 4 J/cm2 (“infected wound” mode), whereas subsequent sessions delivered 2.03 J/cm2 (“wound” mode). Sessions were performed 3 times during week 1, every 72 hours during week 2, and then weekly thereafter. Cases 1 and 3 received PBM for approximately 12 weeks and case 2 for 7 weeks.
Concomitant therapies
Dogs received standard management for HAC. Other therapies included topical antiseptics and systemic or topical antimicrobials when cytology confirmed secondary superficial pyoderma. Routes, dosages, and administration frequencies are reported in the case descriptions.
Safety considerations
Protective goggles were worn by the operator and handlers throughout all PBM sessions. Treatments were performed without sedation, respecting patient behavior and welfare. No adverse events were observed during therapy.
Follow-up
Monitoring consisted of serial clinical examinations and photographic documentation. Follow-up duration was determined by lesion resolution or until PBM treatment was discontinued.
Statistical analysis
No formal statistical analyses were performed due to the descriptive nature and limited number of cases. Data were compiled and summarized using Excel 365 (Excel 365; Microsoft Corp, Version 16.89) to organize clinical records, images, and follow-up information.
Results
A total of 3 dogs were identified during the study period, and none were excluded after eligibility screening. The diagnostic approach varied among cases, reflecting real-world clinical practice.
Case 1
A 5-year-old intact male Labrador Retriever was referred with a 1-month history of obtunded mental status and decreased appetite. The owners reported polyuria and polydipsia as well as episodes of epistaxis over the previous few months. On physical examination, the patient showed marked lethargy, diffuse cutaneous lesions, and an increased body condition score of 7/9. Dermatological examination revealed the presence of 2 symmetrical erythematous, crusted-plaque lesions on the temporal region; a large, crusted plaque on the neck; and alopecia associated with 2 erythematous, crusted plaques across the dorsal thorax and dorso-lumbar region. Microscopic examination of plucked hairs showed hair shafts in the telogen phase. Multiple skin scrapings were negative for ectoparasites. Cytology of the plaques revealed a variable number of macrophages and neutrophils as well as the presence of extracellular and intracellular cocci and the presence of amorphous crystalline deposits.
Complete bloodwork and serum biochemistry showed mild lymphopenia (0.99 K/μL; reference range, 1.06 to 4.95 K/μL), marked increases in ALP (1,800 U/L; reference range, 14 to 147 U/L), and moderately elevated ALT (327 U/L; reference range, 25 to 122 U/L,), GGT (31 U/L; reference range, 2 to 13 U/L), and glutamate dehydrogenase (56 U/L; reference range, 1 to 18 U/L). Urine specific gravity was 1,022, with a urinary protein-to-creatinine ratio of 2.42 (reference range, < 0.5). Abdominal ultrasound revealed bilateral adrenomegaly and hepatomegaly, with the liver being diffusely hyperechoic.
Based on these results, naturally occurring HAC with secondary CC was suspected. To confirm the diagnosis, the following tests were recommended: the low-dose dexamethasone suppression test and skin biopsy under local anesthesia for dermatopathological examination. Additionally, to rule out infectious diseases, bacteriological and mycological examinations from deep skin tissue were also performed. The low-dose dexamethasone suppression test showed a lack of suppression at any time points (before: 3.7 µg/dL, range 1.0 to 6.0 µg/dL; 4 hours after: 3.5 µg/dL, range 0 to 1.4 µg/dL; 8 hours after: 2.7, range 0 to 1.4 µg/dL).
Skin biopsy specimens were collected from the plaque lesions on the neck and on the dorso-lumbar region. Histological examination showed severe and diffuse orthokeratotic hyperkeratosis of the epidermis, with mild-to-moderate irregular hyperplasia. Multifocal deposits of amorphous, crystalline, and deeply basophilic material were observed in the mid- and deep dermis, consistent with dermal mineralization. These mineralized structures were surrounded by a marked accumulation of activated fibroblasts and macrophages and occasionally accompanied by multinucleated giant cells. Both bacteriological and mycological examinations from deep skin tissue were negative.
To investigate the abnormal mental status, and considering the results of the previous examinations, an MRI scan (1.5 Tesla; SIGNA Creator; General Electric) of the head was performed. The scan revealed a large space-occupying lesion (measuring 26 X 26 X 24 mm) in the sellar region, which extended dorsally and exerted clinically important compression on the surrounding brain structures, including the optic chiasm, hypothalamus, and thalamus. The pituitary-to-brain ratio was > 0.31. The MRI scan findings were primarily compatible with a pituitary tumor, and a pituitary macroadenoma was suspected. A final diagnosis of pituitary-dependent hypercortisolism, due to pituitary macroadenoma, with cutaneous CC and secondary superficial bacterial infection, was proposed.
Treatment with trilostane (Vetoryl) was administered orally at a dose of 0.7 mg/kg every 12 hours, resulting in a rapid improvement of clinical signs. Additional treatments, such as radiation therapy and cabergoline, were declined by the owners. Due to the extensive skin lesions, topical therapy with chlorhexidine and systemic antibiotic therapy with amoxicillin-clavulanic acid (Synulox) administered orally at a dose of 20 mg/kg twice daily were started.
Photobiomodulation therapy was proposed as an additional treatment to address the dermatological lesions caused by mineral deposition, following the protocol in the flowchart (Figure 1). Owner informed consent was obtained, and all procedures were based on respecting patient behavior and welfare. The operator wore protective goggles throughout the procedure to ensure safety. After 14 days of initiating PBM therapy, antibiotic treatment was discontinued as serial cytology evaluations confirmed the improvement of superficial pyoderma. From that point onward, the only treatments were PBM therapy and topical chlorhexidine.
Figure 1
A class IV laser (Multiwave Locked System; Asa Srl) was used in all 3 cases. Three sessions were performed during the first week, followed by treatments every 72 hours in the second week and weekly sessions thereafter. The treatment was conducted in scanning mode using a collimated-lens handpiece (2-cm diameter, 3-cm2 spot area). In cases 1 and 3, sessions continued for a total of 12 weeks, whereas treatments were conducted for a total of 6 weeks in case 2. The operator wore protective goggles during all sessions to ensure safety. The affected areas were divided into multiple sections, with each section receiving the same dosage of 4 J/cm2 (“infected wound” mode) during the first session, followed by a dosage of 2.03 J/cm2 (“wound” mode) in subsequent sessions. The flowchart illustrates the different settings using a reference area of 100 cm2.
Citation: American Journal of Veterinary Research 2025; 10.2460/ajvr.25.09.0314
Figure 2
Wound healing progression of crusted plaque-like lesions on the neck of case 1, a 5-year-old male Labrador Retriever, treated with adjunctive PBM therapy using a dual-wavelength near-infrared laser (808 and 905 nm; Multiwave Locked System; Asa Srl). Images were taken at time (T)-0 (baseline), T45, T60, and T90 days. At T0, lesions exhibit crusting and marked erythema. By T45 and T60, crusting and inflammation are visibly reduced. At T90, lesions are resolved and hair regrowth is evident. These images illustrate the progressive effect of PBM on lesion resolution and tissue repair. Study period: 2022 through 2023.
Citation: American Journal of Veterinary Research 2025; 10.2460/ajvr.25.09.0314
Figure 3
Wound healing progression in the right temporal region of case 1, a 5-year-old male Labrador Retriever, treated with adjunctive PBM therapy using a dual-wavelength near-infrared laser (808 and 905 nm; Multiwave Locked System; Asa Srl). Images were taken at different time points during treatment: T0, T45, T60, and T90 days. At T0, the temporal area shows crusts, marked erythema, and inflammation. By T45, crusts are reduced while erythema and inflammation persist. At T60, inflammation is markedly reduced, with initial hair regrowth visible. At T90, healing is complete, with full hair regrowth and a normal appearance of the treated area. The images illustrate the gradual and effective impact of PBM on tissue repair and regeneration in this case. Study period: 2022 through 2023.
Citation: American Journal of Veterinary Research 2025; 10.2460/ajvr.25.09.0314
Six months after diagnosis, the dog was reevaluated for anorexia and worsening of neurological signs, presenting disorientation, head pressing, circling, and pain upon palpation of the cervical column. A pain treatment with tramadol (3 mg/kg) and paracetamol (15 mg/kg) was started, but 2 weeks later the owner opted for euthanasia due to the poor evolution of the dog’s clinical conditions.
Case 2
A 3-year-old intact female German Shepherd was referred for a 2-week history of an erosive-ulcerative lesion on the neck. Topical treatment with streptomycin (q 12 h) was initially prescribed by the referring veterinarian for 10 days, but no improvement was observed. The dog had a previous history of primary immune-mediated nonerosive polyarthritis and meningomyelitis of unknown origin diagnosed through a comprehensive workup, including a total body CT scan, fine-needle aspiration cytology of multiple joints, and an MRI study of the vertebral column with cerebrospinal fluid analysis. Blood screening for vector-borne diseases (including Ehrlichia spp, Leishmania spp, Borrelia burgdorferi, Babesia canis, Anaplasma spp, Dirofilaria immitis, and Hepatozoon spp) ruled out infectious diseases.
After the initial diagnosis of immune-mediated disease, the dog had been receiving prednisolone therapy (1.5 mg/kg once daily) for 2 months.
At the time of presentation to the dermatology service, the immune-mediated condition appeared to be in remission. The owners reported that, following glucocorticoid therapy, the dog had developed severe polyuria, polydipsia, and polyphagia.
During physical examination, the dog exhibited mild lethargy, a distended abdomen, and generalized muscle atrophy. Dermatological evaluation revealed an 8 X 8-cm erosive-ulcerative lesion on the dorsal neck, along with 2 firm, white-coated lesions on the lateral tongue and perilabial region.
The collateral diagnostics performed included microscopic examination of plucked hairs, which showed follicles predominantly in the telogen phase, and multiple superficial and deep skin scrapings, which were negative for parasites. Cytology of the erosive-ulcerative lesions demonstrated abundant amorphous, basophilic material with a few crystal-like structures, neutrophils, macrophages, multinucleated giant cells, and extracellular cocci.
Blood analyses were performed and revealed marked increases in ALT (1,125 U/L), ALP (1,081 U/L), and glutamate dehydrogenase (110 U/L), with mild elevations in AST (117 U/L) and GGT (46 U/L). Further diagnostic tests, including dermatopathology, bacteriology, and mycology examination, were proposed but declined by the owner.
Based on the clinical history, cytological findings, and bloodwork, a diagnosis of CC secondary to iatrogenic HAC, complicated by a superficial bacterial infection, was made.
Glucocorticoid therapy was gradually tapered and discontinued over a 4-week period to minimize the relapse risk of the immune-mediated disease. Topical gentamicin (q 12 h) was initiated, and PBM therapy was applied exclusively to the dorsal neck lesion according to the protocol outlined in Figure 1.
Owner informed consent was obtained, and all procedures respected patient welfare. The operator wore protective goggles throughout treatment. Topical therapy was discontinued after 10 days, following serial cytological evaluations showing progressive improvement of superficial pyoderma. After 25 days of treatment, an > 80% reduction in lesion size was observed, and complete clinical resolution of the dorsal neck lesion was achieved 7 weeks after initiation of laser therapy.
Case 3
An 8-year-old intact male English Bulldog was referred for a 2-month history of progressive polyuria and polydipsia and dermatological lesions. On physical examination, the dog was panting, and a distended abdomen was noted. Dermatological examination revealed erythematous plaques with crusts on the dorsal neck region along with several erythematous papules on the left shoulder area, some of which were confluent and crusted.
Microscopic examination of plucked hairs revealed a predominantly telogen phase. Multiple superficial-to-deep skin scrapings were negative for ectoparasites. Cytological examination of the lesions revealed a mixture of neutrophils, macrophages, and a few multinucleated giant cells around some crystal-like structures. Additionally, cocci were observed, with both intracellular and extracellular distribution. The CBC revealed an increased platelet count (761 X 103/μL; reference range, 150 to 500 X 103/μL) and mild lymphopenia (800 K/μL; reference range, 900 to 4,000 K/μL). Serum biochemistry showed elevated ALP (551 U/L; reference range, 14 to 147 U/L), mildly increased ALT (113 U/L; reference range, 15 to 65 U/L) and GGT (16.7 U/L; reference range, 2 to 13 U/L). Urine specific gravity was 1,004, with a urinary protein-to-creatinine ratio of 12.22 (range, < 0.5).
The ACTH stimulation test (pre-ACTH cortisol: 8.4 µg/dL, range 1 to 4.8 µg/dL; post-ACTH cortisol: 30.80 µg/dL, range 6 to 18 µg/dL) was positive for hypercortisolism. A total body CT scan revealed a 7.3 X 13 X 8-mm pituitary mass in the sellar region, extending dorsally to the basisphenoid, with a pituitary-to-brain ratio > 0.31, hepatomegaly, and bilateral adrenomegaly. A pituitary macroadenoma was considered the most likely diagnosis. The owner declined other diagnostic investigations, such as dermatopathological, bacteriological, and/or mycological examinations of deep skin tissue.
Based on the patient’s history, the type and distribution of the lesions, and the results of the investigations, a diagnosis of macroadenoma with secondary CC and secondary bacterial infection was made.
Treatment with trilostane (Vetoryl; 0.6 mg/kg, q 12 h) was started; to treat the CC and the concurrent secondary bacterial infection, topical therapy with chlorhexidine and systemic therapy with amoxicillin-clavulanic acid (Synulox; 20 mg/kg twice daily) were started. Photobiomodulation therapy was also introduced following the protocol in the flowchart (Figure 1). After 10 days of combined therapy, the antibiotic treatment was discontinued as the pyoderma improved. Owner informed consent was obtained, and all procedures were based on respecting patient behavior and welfare. The operator wore protective goggles throughout the procedure to ensure safety. Clinical improvement of the dermatological clinical signs was observed from the eighth PBM session, with significant progress noted by the sixth week. However, some lesions remained partially improved and persisted at week 12, and complete resolution was not achieved.
Three months after the initial presentation, the dog developed severe dyspnea and 2 episodes of collapse; an acute pulmonary thromboembolism was suspected. The owners declined further diagnostic tests, and compassionate euthanasia was performed.
Discussion
To the best of the authors’ knowledge, this is the first report to describe the use of MLS for the treatment of CC in 2 cases with naturally occurring HAC and in 1 with iatrogenic HAC.
There are no broadly effective treatments for CC owing to the lack of a complete understanding of the mechanisms involved. In human medicine, a variety of treatment modalities are used based on the type, severity, and number of lesions. These include pharmacological therapies, such as antibiotics, immunosuppressants, bisphosphonates, colchicine, IV immunoglobulins, and other biologic therapies. Additionally, nonpharmacological treatments, such as surgical excision, shockwave therapy, and carbon dioxide laser therapy, are also employed.11 Similarly, in veterinary medicine there is no standardization of therapies for CC, and many treatments are supported by limited case reports or rely on the clinical expertise of practitioners. For example, there is one case report12 that describes a dog with CC locally treated with DMSO with a resolution of wounds in 3 months. Dimethyl sulfoxide is known for its high skin-penetrating and anti-inflammatory properties, but its efficacy remains anecdotal as do the mode and frequency of application. The effectiveness and safety of minocycline, a bacteriostatic antibiotic in the tetracycline family used for CC in human medicine, has also been assessed in 5 dogs, with a clinical resolution observed in 4 of 5 cases in 6 to 12 weeks. It is thought that minocycline reduces inflammation, chelates calcium and iron, and influences angiogenesis beyond its antibacterial properties.5 Finally, an accelerated improvement in lesions treated with fluorescent light energy compared to those not exposed and treated with DMSO and chlorhexidine was recently documented in a dog with CC.6
In general, PBM is a nonthermal process that utilizes endogenous chromophores to promote photophysical and photochemical reactions at various biological levels.13 This process involves light in the red or NIR spectrum, with specific wavelengths ranging from 600 to 1,100 nm. Lasers, light-emitting diodes, and various other light-emitting devices with class 3B and class 4 lasers are commonly employed as they emit monochromatic, coherent, and collimated light, enabling precise and targeted treatment of specific tissues.14 Although the exact mechanism of PBM is not fully understood, it could depend on the target and type of cell being modulated. It is highly likely that photons are absorbed by cytochrome c oxidase, which is in the inner membrane of mitochondria. Cytochrome c oxidase absorbs light in the range of 500 to 1,100 nm, becomes activated, and dissociates from nitric oxide, binding to oxygen. This results in improved electron transport and ATP production and the triggering of several pathways that lead to cell proliferation and tissue repair.15,16 On this basis, the use of PBM has been reported for the management of musculoskeletal disorders and neurological diseases, to relieve pain, and, especially, for treatment of wounds given its noninvasive nature and its minimal side effects in both human and veterinary medicine.17–25
However, the efficacy of PBM on target tissues also depends on treatment parameters, light source, wavelength, density, type of energy emission (continued or pulsed), type of lasers, and duration and frequency of laser application.26 Although there have been several in vivo studies,25 results in veterinary medicine are inconclusive due to the lack of standardization of parameters under different clinical conditions. In this case series, we introduced a more standardized approach by following a specific protocol.
The laser used was the MLS laser, a class 4 therapy system emitting NIR beams at 2 wavelengths within the NIR spectrum: 808 nm in continuous or frequency-modulated mode and 905 nm in pulsed mode. The beams are spatially overlapped and synchronized during treatment. The laser has an average maximum power of 3.5 W and a peak power of 270 W.
With the treatments detailed here, complete remission of CC was achieved in 2 of the 3 cases in 7 to 12 weeks. In the case associated with iatrogenic HAC, lesions resolved after approximately 7 weeks—shorter than the 12 weeks reported by Huang et al27 following corticosteroid withdrawal alone. In the dog affected by naturally occurring HAC, lesion resolution occurred after 12 weeks, consistent with previously reported cases treated with minocycline or DMSO. It is likely that additional treatment sessions in the third case would have resulted in complete clinical resolution.
It was hypothesized that these therapeutic effects could be due to multifactorial mechanisms. Based on previously reported effects of PBM, it is possible that laser therapy contributed to modulating inflammation7–29 and supporting tissue repair, enhancing microcirculation,30 reducing local edema,31 and promoting a more favorable environment for healing and resorption of calcium deposits.
Second, the superficial bacterial infections commonly associated with CC lesions also play a critical role in the disease progression, often complicating the healing process. The potential antibacterial effect of PBM,32 along with its role in impacting the skin microbiome as recently documented,33 may have contributed to reducing the bacterial load and shortening the duration of antibiotic use, thereby promoting tissue regeneration and helping to alleviate inflammation.
These observations suggest that PBM may have contributed to accelerated lesion healing. However, due to the small number of cases and the absence of controls, its specific contribution to the clinical outcome cannot be determined. While these findings are descriptive and cannot establish efficacy, PBM may represent a promising adjunctive approach for managing CC in dogs, warranting further investigation in larger, controlled studies.
https://avmajournals.avma.org/view/journals/ajvr/aop/ajvr.25.09.0314/ajvr.25.09.0314.xml
________________________________________________________________________________________
AVMA NEWS I American Bar Association highlight link between domestic violence, animal cruelty
Experts say the biggest red flag of potential animal abuse is inconsistent medical history
Published on November 19, 2025
The AVMA and the American Bar Association (ABA) have partnered to draw attention to the link between violence against people and animals.
Dr. Amanda Bisol, who represents District 1 on the AVMA Board of Directors and has a law degree, and Dr. Kendall Houlihan, associate director in the AVMA Animal Welfare Division, participated in a webinar, “Protecting People and Pets from Domestic Violence,” on October 22.
Spotting the signs
Dr. Bisol highlighted the importance of recognizing signs of animal maltreatment in clinical settings, such as unexplained injuries and a general lack of care or history that “just doesn’t add up.”
“The biggest red flag is an inconsistent [medical] history,” Dr. Bisol said. Another sign that is not as common are multiple injuries on the same animal that are different ages—for example, if an X-ray of a new fracture shows a healing fracture.
It’s important to be aware of how domestic violence and animal maltreatment can be interconnected, Dr. Bisol said, because sometimes, veterinarians may be the only ones to see the potential signs of an issue in a home. She also urged documenting a suspicious case with additional detail, including pictures, as such veterinary records may be valuable evidence if the case is investigated.
The sessions include an overview of how to identify signs of domestic violence and animal abuse or neglect and show how the two may be connected. Presenters also discuss how to access resources that support survivor decision-making and how to foster collaboration with local organizations and agencies that can help protect both survivors and their pets.
Collaborative effort
This collaboration between the AVMA and ABA comes after the law association adopted Resolution 504 earlier this year, urging all levels of government “to enact legislation and/or support judicial processes that protect individuals by protecting their pets in family law and civil restraining order proceedings.”
“The aim of this collaboration is to highlight the roles and responsibilities of different professionals and how we may more effectively interact with and support each other and our communities,” said Dr. Houlihan.
AVMA policy on “Animal Abuse and Animal Neglect” states that prompt disclosure of abuse is necessary to protect the health and welfare of animals and people.
While disclosure is important, “it is not the veterinarian’s task to determine if the maltreatment meets the enumerated elements of a crime; this is the duty of the investigating authority and the criminal justice system,” according to the AVMA’s “The Veterinarian’s Framework for Identification and Response to Suspected or Known Animal Maltreatment,” a free resource for veterinary professionals.
Veterinary social workers are uniquely dedicated to tending to the human needs that arise from relationships between humans and animals. Accordingly, Dr. Bethanie Poe, associate director of education and training for the University of Tennessee’s Center for Veterinary Social Work, also provided her expertise to the sessions.
Guiding future efforts
Animal cruelty has historically been considered an isolated issue that is not indicative of other violence. However, “recent research shows a well-documented link that it is a predictive or co-occurring crime with violence against humans (including intimate partners, children, and elders) and is associated with other types of violent offenses,” according to a 2021 Federal Bureau of Investigation Law Enforcement Bulletin. “Increased awareness of this linkage and a collaborative approach to these investigations strengthens the identification and reduction of such crimes.”
One result of the growing awareness of the link between animal and personal violence is greater consideration of what to do with a pet when the owner enters a domestic violence shelter.
For example, a paper published in the May 2024 issue of JAVMA described the creation of a safekeeping program for pets owned by domestic violence victims.
Dr. Hillary L. Pearce and others found that involving at least one veterinary practice was integral, “as most pets entering the program needed vaccination, testing for infectious disease, and/or parasiticide prevention before being placed into foster homes. The veterinary practice also served as a crucial temporary holding facility until pets could be matched into suitable foster homes.”
Recognizing that a variety of professionals engage with people and pets affected by domestic violence, future collaborations may add the perspectives of law enforcement and judges, who also work to support the human-animal bond in challenging circumstances.
________________________________________________________________________________________________________
AVMA I News Normalize pet nutrition conversations at every veterinary visit
Purina handbook explores pet nutrition across the lifespan with proactive and practical recommendations
Published on November 17, 2025
As pet obesity rates continue to climb nationwide, nutrition is top of mind for pet owners and veterinarians alike.
The Purina Institute Handbook of Canine and Feline Well-Pet Nutrition, launched in August, provides evidence-based recommendations for veterinary care teams to help them prioritize pet nutrition in practice. The handbook is available for free with a sign-up for scientific communications from the institute.
The handbook cites a May 2023 Purina Institute survey, which showed 96% of pet owners said they trust their veterinarian for pet nutrition advice, but only 22% of veterinary professionals have proactive conversations about nutrition at most visits.
“Nutrition also brings the pet pleasure, positively reinforces the human-animal bond, and helps owners express love to their pets,” according to the introduction. “As we continue to recognize the integral role that pets play in our lives, it is essential to acknowledge the importance of proper nutrition in ensuring their health and longevity.”
Pet nutrition
Developed with contributions from 54 experts across 20 countries, the 322-page handbook covers nutrient fundamentals in small animals, specialized nutrition guidance by life stage, proactive nutrition recommendations for various health benefits, and other practical tools.
For example, Dr. Yuki Okada, a board-certified veterinary nutritionist in San Francisco, in the guide calls protein “undoubtedly the most critical macronutrient in the nutrition of both dogs and cats, serving as the primary source of amino acids and nitrogen for various physiological functions.”
The recommended protein intake is approximately 4 g/kg of body weight for senior dogs and about 5.2 g/kg for senior cats, according to the handbook, unless kidney disease necessitates certain restrictions.
Cats, as obligate carnivores, have significantly higher protein needs than dogs to maintain a constant state of gluconeogenesis that meets energy demands.
And senior pets may need up to 50% more protein than younger adults to counteract age-related muscle loss.
Overall, geriatric pets experience physical, functional, and metabolic changes that are still amenable to nutritional intervention to improve health and quality of life. A cohesive framework of nutritional prioritization and planning can aid in managing comorbidities in geriatric pets, according to the guide.
The section on proactive strategies covers the impact nutrition has on everything from oral and gastrointestinal health to joint, behavioral, and urinary health.
In addition, a table of ingredients commonly found in dental diets, treats, and chews, such as folic acid or polyunsaturated fats, shows the respective modes of action.
Client conversations
The handbook calls for veterinary care teams to have “a well-stocked communication skill toolkit” on hand to equip conversations with clients about pet nutrition.
Talk model
“Clients want to be partners in their pet’s veterinary care,” the handbook states. “Clients that understand the beneficial outcome for their pet (the ‘why’ behind recommendations) are more likely to adhere to plans of care.”
Or, talking about pets’ skin and coat health, an overall health indicator, is also a great conversation starter.
The section also addresses some common communication challenges and how to navigate them, including client resistance, skepticism, and the perception that a discussion on pet foods is driven by financial motivations and wanting to “make a sale.”
Veterinary professionals need to be prepared to confront and correct misinformation, as well as disinformation, differentiated as “false or incorrect information that is shared with an underlying intent to mislead.”
Care teams can counter misinformation by proactively discussing and providing resources on common nutritional myths. This is known as inoculation or “prebunking” with “cognitive antibodies,” with the aim of making pet owners more resilient to nutritional misinformation in the future.
Practical tools
Beyond nutritional science, the handbook offers 25 “Practical Tools,” including a fecal scoring chart with pictures and descriptions of different stools.
It also includes guidance on feeding “finicky” felines, managing cat allergens through nutrition, and selecting products marketed as supplements, as well as scoring charts for assessing body and muscle condition.
“It has been reported that pet owners tend to underestimate the body condition of their pet, emphasizing the importance of equipping every team member to perform and teach body condition scoring (BCS) and muscle condition scoring (MCS) at each visit,” writes Ashley Self, assistant director of veterinary nutrition at Texas A&M University College of Veterinary Medicine and Biomedical Sciences. “By involving the owners in the process of obtaining a body weight and estimating BCS and MCS, veterinary teams can identify trends that support proactive management while also enhancing the pet owner’s understanding of what constitutes a healthy body weight, BCS, and MCS.”
Veterinarians should also educate pet owners on available tools like wearable devices, litter box monitoring systems, and mobile applications that support at-home monitoring for more proactive and preventive care.
“Nutritional science has evolved significantly, and there are many facets to consider in a well-pet diet—not just from the pet’s perspective, such as life stage, activity level, and body condition, but also the owner’s perspective—their lifestyle, budget, and preferences,” said Natalia Wagemans, MD, the institute’s global head, in an August 28 press release. “This resource simplifies the process of making informed recommendations and can be a vital tool for veterinary professionals to both facilitate nutrition conversations with their clients and provide informed responses to questions.”
________________________________________________________________________________________________________
AVMA News I FDA reminds pet food makers that raw ingredients pose risk of exposure to H5N1
Published on November 14, 2025
Federal agencies are tightening regulations on raw pet food ingredients due to the risk of highly pathogenic avian influenza (type A H5N1) transmission.
Recent investigations have pointed to raw meat, specifically poultry, and unpasteurized milk as a source of infection for cats, both domestic and wild, which can experience severe illness or death from infection with the virus.
Dogs can also contract H5N1, although they usually exhibit mild clinical signs and low mortality.
“During the fall migratory season, H5N1 detections typically increase throughout the United States in wild birds, with potential spread to commercial and backyard poultry flocks,” according to the Food and Drug Administration (FDA) Center for Veterinary Medicine.
Suspected or verified sources of highly pathogenic avian influenza type A H5N1 infecting cats include unpasteurized cow’s milk and raw poultry products.
With that in mind, on September 30, the agency issued a reminder to pet food manufacturers that H5N1 is a “known or reasonably foreseeable hazard” when using uncooked or unpasteurized ingredients.
Under the agency’s Food Safety Modernization Act Preventive Controls for Animal Food requirements, animal food businesses must conduct a reanalysis of their food safety plan when the FDA determines it is necessary to respond to new hazards and developments in scientific understanding.
Earlier this year, the FDA determined that food safety plans of manufacturers that specifically use uncooked or unpasteurized materials from poultry or cattle should include H5N1 as a potential hazard and implement preventive controls as needed. This could include seeking ingredients from healthy flocks or herds, taking processing steps such as heat treatment, or implementing a supply chain–applied control to provide assurance that ingredients used in animal food do not come from H5N1-infected animals.
“Manufacturers that implement a preventive control for the H5N1 hazard as a result of their reanalysis will be taking an important step toward protecting cat and dog health and helping to prevent spread of H5N1,” according to the FDA.
_______________________________________________________________________________________________________________
Taylor & Francis Online I Serum Onconeural Antibodies in Dogs and Cats for Early Diagnosis of Cancer
Marianna Agassandian & Khristofor Agassandian
Published online: November 12, 2025
Abstract
Cancer is one of the leading causes of death in dogs and cats worldwide, and effective early detection techniques and reliable therapies are still lacking. Given the high demand for early cancer detection and differentiation in veterinary diagnostics, we developed and validated a new diagnostic approach to assess onconeural antibodies, also known as high-risk antibodies, in dog and cat blood serum samples. We determined the presence of systemic onconeural/high-risk (ONHR) antibodies, their suitability for early cancer diagnostics, and the feasibility of differentiating various malignancies. Our results identified several ONHR antibodies in 0.1 mL of specimens by the immunoblot-based technique, which was confirmed by indirect immunofluorescence assay. The diagnostic performance for detecting identified antibodies has demonstrated >95% sensitivity in dogs, >93% sensitivity in cats, as well as >97% specificity in dogs, and >95% specificity in cats. Thus, our data provide the first proof-of-principle that onconeural antibodies can be detected in dogs and cats, and their identification in serum might serve as a new tool for early cancer diagnosis.
Introduction
Malignant tumors in companion animals are a significant problem affecting the lives and well-being of pets and their owners. According to the Veterinary Cancer Society, cancer is the leading cause of death in 47% of dogs, especially elderly dogs, and 32% of cats.Citation1 Dogs develop cancer at almost the same rate as humans.Citation2 Cancer detection at the earliest stage may significantly improve the efficacy of the treatment and survival. Sensitive and specific methods for cancer screening can reduce mortality and improve the quality of life of affected animals.
Recently, two advanced techniques adapted from human cancer diagnostics have been implemented into veterinary medicine. The OncoK9 “liquid biopsy”, based on next-generation sequencing, could only differentiate hematological malignancies, required large blood volumes, was costly, and was discontinued from commercial use.Citation3
The second, Nu.Q test, measures plasma nucleosome concentration to predict certain cancers but provides only positive/negative results and can be provoked by inflammation, trauma, or other diseases.Citation4
To address these diagnostic limitations, we focused on antibody biomarkers, widely used in human medicineCitation5 but still limited in veterinary diagnostics due to a lack of development and standardization.Citation6–11 Canine and human cancers share biological similarities,Citation12 making this approach promising. We investigated onconeural antibodies, produced against neuronal proteins aberrantly expressed in tumor cells. These antibodies are linked to paraneoplastic neurological syndromes (PNS), which may precede cancer diagnosis.Citation13 Although not always directly pathogenic, they are valuable diagnostic markers.Citation14
Onconeural antibodies target intracellular or extracellular (membrane-bound) neuronal or glial proteins. Antibodies against intracellular proteins are more specific for malignancy rather than neurological syndromes and considered “high-risk”,Citation14,Citation15 while extracellular-directed antibodies are more closely tied to neurological diseases.Citation16 Sometimes, multiple antibodies coexist and are associated with the same cancer or PNS.Citation17 Paraneoplastic disorders are diagnosed in human patients with cancer, but there is a lack of knowledge about these diseases in animals. Even though these syndromes have been reported in dogs and cats, these diseases are usually diagnosed too late after the identification of cancer.
Since ONHR antibody formation usually precedes cancer, its early detection can predict malignancy before clinical symptoms, aiding timely treatmentCitation18–21 and better outcomes for pets.Citation22–24 Thus, serological detection of ONHR antibodies represents a convenient diagnostic method for early cancer detection and differentiation.
Depending on the type of malignancy, tumor cells may express intracellular proteins/antigens such as amphiphysin, CV2/CRMP5, Ri/NOVA1, Yo/CDR2, Hu/ELAV, recoverin, SOX1, titin, ZIC4, GAD65, and Tr/DNER that are normally restricted to the nervous system.Citation25–35 These antigens can induce specific antibody formation before tumors are symptomatic. Altered expression, mutations, or posttranslational modifications can create neoantigens, provoking immune responses.Citation36
As illustrated in many studies, between 50 and 90% of tumor-associated proteins/antigens in the human body acquire a variety of modifications that alter immunologic processing and presentation.Citation37 Thus, the modified self-antigens and neoantigens can be recognized as non-self-antigens by the immune system and promote onconeural antibody formation in the blood of patients with different types of tumors and have the potential to be an early sign of cancer. According to the literature, ONHR antibodies were detected in more than 90% of cases with underlying cancer within a few years of the study.Citation38
Unfortunately, in veterinary medicine, only a few antibodies to neuronal proteins have been identified.Citation39–45 In our study, we validated ONHR antibodies in sera of cancer-bearing dogs and cats, confirming their stability and strong association with malignancies. These findings support their use as novel, noninvasive biomarkers for early cancer detection and malignancy-associated neurological diseases in pets.
Materials and Methods
Ethics Statement
Ethical approval was not required for this study, as it used de-identified residual serum samples submitted to Pet Preferred Diagnostics for research purposes and after routine diagnostics, with no additional collection or animal interventions.
Inclusion Criteria
The validation study was conducted on a total of 202 dogs and 50 cats, including 76 presumably healthy dogs and 20 presumably healthy cats with medical records from participating veterinary clinics and the National Institute of Health blood bank. The records included patient information such as age, sex, breed, neuter status, clinical history, and examination details.
Serum samples were collected from dogs and cats with confirmed or suspected malignancies at the time of validation, as well as animals with specific neurological symptoms. Sera obtained from presumably healthy dogs and cats, based on physical examination and clinical history, were also included in the validation study. Animals with conditions such as chronic infections were excluded from this study.
Animal Samples
Serum samples (0.05–0.5 mL) from animals (dogs and cats) with confirmed, suspected cancer, and presumably healthy were prepared from blood using VACUETTE tubes with serum separator. After 30–45 minutes at room temperature (which allows blood to clot), the tubes containing the collected blood were centrifuged at 2000–2500 RPM for 20 minutes to separate the serum. Sera were then transferred to Eppendorf tubes labeled with the patient’s name and date of sample collection and shipped to Pet Preferred Diagnostics laboratory to be tested for the presence of onconeural antibodies.
Routine screening for the presence of ONHR antibodies such as anti-Hu, -Yo, -CV2, -Ri, -Tr, -ZIC4, -SOX1, -GAD65, -recoverin, -amphiphysin, and anti-titin (11 ONHR antibodies) was performed by immunoblot and confirmed by indirect immunofluorescence technique (IIFT) using monkey tissue slides.
Protein Sequence Comparison
UniProt Knowledgebase (UniProtKB) has been used for the analysis of ELAV/HuD (ELAV-like protein 4), CDR2/PCD17 (cerebellar degeneration-related protein 2), CRMP5/DPYSL5 (dihydropyrimidinase-related protein 5), NOVA1 (RNA binding protein), DNER/BET (Delta/Notch-like epidermal growth factor-related receptor), ZIC4 (Zinc finger protein), SOX1 (transcription factor protein), GAD65 (glutamate decarboxylase 2), RCVRN/RCV1 (recoverin/cancer-associated retinopathy protein), AMPH/BIN1 (amphiphysin/bridging integrator1/Myc box-dependent-interacting protein 1), and TTN (Titin/Connectin) human, canine, and feline protein sequences. Human, canine, and feline protein sequences were aligned using Clustal Multiple Sequence Alignment. A sequence similarity search query was submitted in the FASTA format.
Immunoblot-Based Assay
Serum samples from dogs and cats were tested using the commercial immunoblot kit (EUROLINE Neuronal Antigens Profile 72 (IgG), DL 1111–1601-72 G; Euroimmun, Lübeck, Germany). Immunoblots were performed on the EUROBlotOne system (Euroimmun). The nitrocellulose test strips with immobilized amphiphysin, CV2, Ri, Yo, Hu, recoverin, SOX1, titin, ZIC4, GAD65 and Tr antigens were incubated with patients’ serum according to the manufacturer’s instructions, modified and validated by our laboratory to adapt the test kit for veterinary diagnostics.
The strips were washed with a buffer provided in the kit and then incubated with the secondary alkaline-phosphatase conjugated goat anti-Canine IgG (SouthernBiotech, Birmingham, AL, USA) and goat anti-Cat (Bethyl Laboratories, Montgomery, TX, USA) antibodies. The onconeural antibodies in the serum bound to the related antigens were then visualized by NBT/BCIP substrate solution (nitroblue tetrazolium chloride/5-bromo-4chloro-3 indolylphosphate, Euroimmun). The intensity of binding of 11 ONHR antibodies was evaluated using EUROLineScan software (Euroimmun). An ONHR antibody band intensity value between 0 and 7 was considered negative or Class 0, between 8 and 14 was considered borderline or Class (+), between 15–35 and 36–70 as positive or Classes +, ++, and above 70 as strongly positive or Class +++. Strips with no added serum samples were used as negative controls.
Indirect Immunofluorescence Assay
The Neurology Mosaic 8 testing kit (Euroimmun, FA 1111–1005-8) is an indirect immunofluorescence assay used to confirm the presence of autoantibodies against eight targets associated with cancers and neurological diseases. This test detects antibodies against Hu, Yo, CV2, Ri, Tr, ZIC4, GAD65, and amphiphysin.
Serum samples were applied to the slides that were filled with BIOCHIPs – cerebellum and pancreatic sections collected from a monkey, according to the manufacturer’s instructions, and were modified and validated by our laboratory to adapt the test kit for veterinary diagnostics. Sera from a dog and a cat previously diagnosed with cancer were used as positive controls. A serum sample from a healthy dog or cat was included in each evaluation as the negative control. After incubation with diluted serum samples (1:10 to 1:100) for 30 min at room temperature, BIOCHIP slides were washed with phosphate-buffered saline/Tween (PBST) for 5 min and developed with secondary fluorescent-labeled antibodies (for dog samples: fluorescein isothiocyanate (FITC)-conjugated goat anti-Dog IgG, Euroimmun; for cat samples: FITC-conjugated goat anti-Cat IgG from Jackson ImmunoResearch Laboratories, West Grove, PA, USA). The BIOCHIP slides were then incubated for 30 min at room temperature and washed with PBST for at least 5 min. Immunofluorescent images were acquired with the EUROStar III Plus fluorescent microscope at ×20 and ×40. Each power field was captured in four quadrants using the same imaging technique. Serum samples from dogs and cats with positive results were retested with positive and negative controls to ensure the repeatability of the results.
Statistical Analysis
Sensitivity and specificity were determined as the fractions of true-positive results in the cancer-diagnosed/detected group and true-negative results in the control group, respectively, and calculated for results where borderline results were interpreted as negative. Confidence intervals (CIs) were determined to be 95% for all sensitivity and specificity calculations. Microsoft Excel (Microsoft Corporation, Redmond, WA, USA) and GraphPad Prism 10 (GraphPad, San Diego, CA, USA) online tools were used for statistical analyses.
https://www.tandfonline.com/doi/full/10.2147/VMRR.S537744#d1e278
_______________________________________________________________________________________________________________
AVMA |News – FDA issues first-ever emergency use authorization for drug to combat screwworm in dogs
Meanwhile, new USDA playbook lists tools and resources for screwworm response
Published on November 12, 2025
Underscoring the gravity of the New World screwworm (NWS) threat looming just across the U.S.-Mexico border, on October 24 the Food and Drug Administration (FDA) published the first emergency use authorization (EUA) for the antiparasitic drug Credelio (lotilaner).
The is the first time the agency has invoked this emergency authority for an animal drug. In this case, it is for treatment of infestations of NWS in dogs and puppies. While most dogs in the U.S. are at low risk of contracting NWS due to their geographic location, dogs near the U.S.-Mexico border and dogs that travel to countries with active NWS cases are more likely to be exposed.
“When it comes to emerging animal health threats, we need to be proactive, not reactive,” FDA Commissioner Marty Makary, MD, said in a press release.
The Food and Drug Administration (FDA) has taken the unprecedented step of issuing an emergency use authorization for a canine flea and tick medication for treatment of New World screwworm (NWS) infestations in dogs and puppies.
Infestation
Screwworm flies target warm blooded animals, including livestock, dogs and other pets, wildlife, and humans, laying eggs in an open wound or orifice. Once the eggs hatch, NWS larvae spend the next several days consuming the host’s tissue, before reaching maturation and beginning the cycle again. NWS can cause serious damage to a host, which may die from secondary infection if not treated.
Detections of NWS have been on the rise in Central America and Mexico in recent months. In September, a screwworm case was identified in Sabinas Hidalgo, a Mexican city less than 70 miles from the Texas border, making it the northernmost detection during the current outbreak. A second detection, believed to be an isolated case and not related to the first, was detected in October in Montemorelos, about 100 miles further south in the same state.
U.S. and Mexican officials have heightened surveillance in the border region and the U.S. has closed its southern ports to imports of live cattle, bison, and horses from Mexico since July to prevent the parasite’s spread into the U.S., where it has essentially been eradicated since the 1960s.
____________________________________________________________________________________________________________________
AVMA NEWS I Winter holiday pet safety
November and December abounds with holiday celebrations, and nothing can spoil good cheer like an emergency trip to the veterinary clinic. These tips can help keep your winter holiday season from becoming not-so-happy—for your pet and for you.
Plan in advance
Make sure you know how to get to your 24/7 emergency veterinary clinic before there’s an emergency. Talk with your veterinarian in advance to find out where you would need to take your pet, and plan your travel route so you’re not trying to find your way when stressed. Always keep these numbers posted in an easy-to-find location in case of emergencies:
- Your veterinarian’s clinic phone number
- 24/7 emergency veterinary clinic (if different)
- ASPCA Poison Control Center: 888-426-4435
- Pet Poison Helpline: 855-764-7661
Food
Keep people food away from pets, and instruct everyone else to do the same. If you want to share holiday treats with your pets, make or buy treats formulated just for them. The following people foods are especially dangerous for pets:
- Chocolate is an essential part of holidays for many people, but it is toxic to dogs and cats. It’s safest to consider all chocolate off limits for pets, even though the harm it can cause varies based on the type of chocolate, the size of your pet, and the amount eaten.
- Other sweets and baked goods also should be kept out of reach. Not only are they often too rich for pets; they may contain the artificial sweetener xylitol, which has been linked to liver failure and death in dogs.
- Table scraps – including gravy, sauces, dressing, and meat or poultry fat or skin – should be kept away from pets. During the holidays, when our own diets tend toward extra-rich foods, table scraps can be especially hard for pets to digest and can cause pancreatitis. Bones can cause choking or intestinal blockage. Plus, many foods that are healthy for people are poisonous to pets, including onions, raisins, and grapes.
- Unbaked yeast dough can cause problems for pets, including painful gas and potentially dangerous bloating.
Quick action can save lives. Signs that your pet may have eaten something they shouldn’t include sudden behavior changes, depression, pain, loss of appetite, vomiting, or diarrhea. If your pet has any of these signs, call your veterinarian or nearest veterinary emergency clinic immediately. You may also want to call the ASPCA Poison Control Center (888-426-4435) or the Pet Poison Helpline (855-764-7661); note that a fee may apply.
Decorating
Holiday plants, lights, candles, and other decorations can make the holidays festive, but they also pose risky temptations for our pets.
- Ornaments can cause hazards for pets. Breakable ornaments can cause injuries, and swallowed ornaments can cause intestinal blockage or illness. Keep any ornaments, including those made from salt-based dough or other food-based materials, out of reach of pets.
- Tinsel, ribbons, wreaths, and other decorative materials also can be tempting for pets to play with and eat. These items whether swallowed in whole or in part can cause choking or intestinal blockage.
- Christmas trees can tip over if pets climb on them or try to play with the lights and ornaments. Consider tying your tree to the ceiling or a doorframe using fishing line to secure it.
- Water additives for Christmas trees can be hazardous to your pets if swallowed. Avoid adding anything to the water for your tree if you have pets in the house.
- Electric lights can cause burns when a curious pet chews the electrical cords.
- Candles and oil lamps are attractive to pets as well as people. Never leave a pet alone in an area with a lit candle or lamp; it could result in a fire.
- Flowers and festive plants can result in an emergency veterinary visit if your pet gets hold of them. Poinsettias, amaryllis, mistletoe, balsam, pine, cedar, and holly are among the common holiday plants that can be dangerous and even poisonous to pets who eat them. The ASPCA offers lists of plants that are toxic to dogs and cats.
- Potpourris should be kept out of reach of inquisitive pets. Liquid potpourris pose risks because they contain essential oils and other ingredients that can severely damage your pet’s mouth, eyes and skin. Non-liquid potpourris containing flowers, leaves, bark, herbs, and/or spices could cause problems if eaten.
Hosting parties and visitors
Visitors can upset pets, as can the noise and excitement of holiday parties and any celebratory fireworks. Even pets that aren’t normally shy may become nervous in the hubbub that can accompany a holiday gathering. The following tips will reduce emotional stress on your pet and protect your guests from possible injury.
- All pets should have access to a comfortable, quiet place indoors if they want to retreat. Make sure your pet has a room or crate somewhere away from the commotion, where your guests won’t follow, that your pet can go to anytime they want to get away.
- Inform your guests ahead of time that you have pets or if other guests may be bringing pets to your house. Guests with allergies or weakened immune systems (due to pregnancy, disease, or medications/ treatments that suppress the immune system) need to be aware of the pets (especially exotic pets) in your home so they can take any needed precautions to protect themselves.
- Guests with pets? If guests ask to bring their own pets and you don’t know how the pets will get along, you can either politely decline their request or plan to spend some time helping the pets to get to know each other, supervising their interactions, monitoring for signs of a problem, and taking action to avoid injuries to pets or people.
- Pets that are nervous around visitors should be put it in another room or a crate with a favorite toy. If your pet is particularly upset by houseguests, talk to your veterinarian about possible solutions to this common problem.
- Exotic pets make some people uncomfortable and may themselves be more easily stressed by gatherings. Keep exotic pets safely away from the holiday hubbub.
- Watch the exits. Even if your pets are comfortable around guests, make sure you watch them closely, especially when people are entering or leaving your home. While you’re welcoming hungry guests and collecting coats, a four-legged family member may make a break for it out the door and become lost.
- Identification tags and microchips reunite families. Make sure your pet has proper identification with your current contact information – particularly a microchip with registered up-to-date, information. That way, if your pet does sneak out, they’re more likely to be returned to you. If your pet isn’t already microchipped, talk to your veterinarian about the benefits of this simple procedure.
- Clear the food from your table, counters and serving areas when you are done using them and make sure the trash gets put where your pet can’t reach it. A carcass or large quantity of meat sitting out on the carving table, or left in a trash container that is easily opened, could endanger your pet if eaten. Dispose of carcasses and bones – and anything used to wrap or tie the meat, such as strings, bags and packaging – in a covered, tightly secured trash bag placed in a closed trash container outdoors (or behind a closed, locked door).
- Trash also should be cleared away where pets can’t reach it – especially sparkly ribbon and other packaging or decorative items that could be tempting for your pet to play with or eat.
When you leave the house
- Unplug decorations while you’re not around. Cats, dogs and other pets are often tempted to chew electrical cords.
- Take out the trash to make sure your pets can’t get to it, especially if it contains any food or food scraps.
Holiday travel
Whether you take your pets with you or leave them behind, take these precautions to safeguard them whenever you’re traveling. Learn more about traveling with pets.
- Interstate and international travel regulations require any pet you bring with you to have a health certificate from your veterinarian—even if you are traveling by car. Learn the requirements for any states or countries you will visit or pass through, and schedule an appointment with your veterinarian to get the needed certificate within the timeframes required by those destinations. (Even Santa’s reindeer need to get health certificates for their annual flight around the world!)
- Pets in vehicles should always be safely restrained and should never be left alone in the car in any weather. Proper restraint means using a secure harness or a carrier, placed in a location clear of airbags. Never transport your pet in the bed of a truck.
- If you’re traveling by air and considering bringing your pet with you, talk with your veterinarian first. Certain pets, such as short-nosed dogs and cats, may have difficulty with air travel. Your veterinarian is the best person to advise you regarding your own pet’s ability to travel.
- Pack for your pet as well as yourself if you’re going to travel together. In addition to your pet’s food and medications, this includes bringing copies of their medical records, information to help identify your pet if they become lost, first aid supplies, and other items. Refer to our Traveling with Your Pet FAQ for a more complete list.
- Boarding your dog while you travel? Talk with your veterinarian to find out whether and how to protect your pet from canine flu and other contagious diseases, and to make sure your pet is up-to-date on vaccines.
_____________________________________________________________________________________________________________________
AVMA – Online Publication Date: October 24, 2025
Improving noninvasive diagnosis of cutaneous and gastrointestinal forms of Pythium insidiosum infections in dogs: complementary roles of serum-based ELISA and polymerase chain reaction
Subarna Barua, PhD, Asfiha Tarannum, MS, Joseph Newton, DVM, Leonel Mendoza, PhD, Kelly Chenoweth, BS, Priscilla Barger, PhD, and Chengming Wang, DVM, PhD
Abstract
Objective
To evaluate the diagnostic performance of ELISA and PCR for detecting pathogenic oomycete infections and to explore associations with clinical presentation and treatment status.
Methods
Canine sera (n = 796) submitted to Pythium Serology Diagnostic Laboratory (from 2023 through 2025) were analyzed by ELISA to detect anti-Pythium antibodies and a PCR targeting Pythium insidiosum, Lagenidium spp, and Paralagenidium spp. Clinical records were reviewed for a subset of submissions. Analyses included χ2 tests, a linear mixed-effects model, and Cohen κ for ELISA-PCR agreement.
Results
Across all 697 samples tested by both methods, 69 (9.9%) were positive and 495 (71.0%) were negative by both assays, yielding an overall concordance of 81%. Polymerase chain reaction identified 5 cases, including cutaneous infections and Lagenidium infections tested to be negative by ELISA. Among confirmed positive submissions, ELISA detected anti-Pythium antibodies in 99.4% (182 of 183), whereas PCR detected Pythium DNA in 45.6% (72 of 158).
Conclusions
While ELISA is well suited for first-line screening due to its high sensitivity, PCR provides critical specificity and broader detection, especially in challenging cutaneous cases and when alternative pathogenic oomycetes are suspected. Enzyme-linked immunosorbent assay was highly effective for initial diagnosis and monitoring treatment response, and PCR was most useful for identifying active infections or relapses.
Clinical Relevance
The combined use of ELISA and PCR enhances diagnostic accuracy, improves detection of misdiagnosed Lagenidium infections, and guides clinical decision-making and epidemiological surveillance of this emerging and serious disease.
Keywords: gastrointestinal and cutaneous pythiosis; Pythium insidiosum; Lagenidium; ELISA; triplex PCR
Pythiosis is an emerging infectious disease of increasing concern in both veterinary and human medicine. It is caused by the oomycetes Pythium aphanidermatum, Pythium insidiosum, and Pythium periculosum, fungal-like organisms commonly referred to as the “water mold.”1,2 The disease is most commonly reported in tropical and subtropical regions,3 and within the US, it is endemic to the Southeast and Gulf Coast, though cases are increasingly documented nationwide.4,5
The infective stage of pathogenic Pythium spp is through biflagellate zoospores, which exhibit strong tropism for animal hair and tissues, initiating infection by encysting on damaged skin or gastrointestinal (GI) mucosa or by small pieces of environmental hyphae.6 While many infected animals have a history of repeated exposure to standing freshwater, other risk factors remain poorly defined.7
Canine pythiosis is typically diagnosed in 2 distinct clinical forms: cutaneous and GI, with the GI form being more commonly reported.3 Concurrent presentation of both forms is rare.8 The infection can disseminate to the pancreas, mesenteric lymph nodes, and bile ducts, whereas cutaneous lesions typically affect the limbs, face, or tail.9 Pythiosis is more frequently diagnosed in young, apparently immunocompetent dogs.8,10
Diagnosis of pythiosis is often delayed due to its nonspecific clinical presentation and the need for specialized diagnostic methods, such as culture, serology, and molecular testing.3,11,12 In the absence of pathognomonic clinical or histological features, pythiosis may be misdiagnosed as cases of zygomycosis or lagenidiosis.3,8 Phylogenetic analyses based on cytochrome oxidase II and large subunit ribosomal RNA gene sequences indicate that Lagenidium and Pythium are closely related.13,14 Given the phylogenetic and clinical similarities of these organisms, specific molecular assays are essential to distinguish Pythium from Lagenidium to guide appropriate therapy.10,15
Traditional diagnosis of pythiosis via microbiological culture is labor intensive and slow and requires skilled personnel.3,12 Immunological methods, such as ELISA16,17 and immunodiffusion,18 offer faster results by detecting serum antibodies against P insidiosum, though they are not commercially available and do not distinguish between active and past infections. Furthermore, they may be unreliable for localized infections, such as ocular pythiosis.19
Polymerase chain reaction–based molecular diagnostics have shown promise for specific detection, but most existing assays target only P insidiosum and do not differentiate it from other Oomycota pathogens.19 This limitation may delay accurate identification and negatively impact timely management and contributes to under-recognition of the disease. To address these challenges, we recently developed a multiplex molecular assay to improve accuracy in identifying P insidiosum and distinguishing it from other oomycetes.15 Building on this advancement, this study integrates serological and molecular methods in a large-scale surveillance effort to evaluate their combined diagnostic utility.
In this study, ELISA and, for the first time, PCR were both performed to detect anti-Pythium antibodies and Pythium DNA in canine sera. By comparing these results with clinical presentations (cutaneous vs GI) and treatment outcomes, we aim to evaluate the diagnostic strengths and limitations of each method and provide practical recommendations for clinical use.
Methods
Canine serum samples
A total of 796 canine serum samples submitted to the Pythium Serology & Mucormycosis Serology Laboratory at Auburn University College of Veterinary Medicine from October 2023 through February 2025 were included in this study. These samples represented 573 unique dogs, of which 456 (79.6%) had a single submission, and 117 (20.4%) had multiple submissions. Among dogs with serial submissions, 108 of 117 (92%) had 2 to 4 samples submitted, whereas 9 of 117 (8%) had 5 or more during the study period.
Of the total samples, 183 were from dogs diagnosed with pythiosis by other diagnostic modalities (eg, culture, histopathology, or immunohistochemistry) performed at external laboratories. Samples were classified as GI, cutaneous, or unusual (eg, thoracic) based on the clinical history provided by the submitting veterinarian on the test request form. Only samples submitted with adequate clinical history provided to allow definitive presentation were included in the comparison of GI and cutaneous presentations.
Samples were submitted from a wide range of breeds but were dominated by larger sporting-/working-type breeds, such as Labrador Retriever (n = 107), German Shepherd Dog (63), large mixed breed (43), Golden Retriever (31), Standard Poodle/large Doodle breeds (27), German Shorthaired Pointer (23), pit bull–type dog (23), Great Pyrenees (16), and others, whereas submissions from smaller-sized breeds, such as French Bulldog (13), Dachshund (13), Chihuahua (4), Pug (3), Boston Terrier (4), Maltese (4), and Miniature Schnauzer (4), were less common.
ELISA to detect anti-Pythium antibody
The ELISA used for the detection of anti–P insidiosum serum antibodies in this study was described previously,20,21 with minor modifications. Briefly, high-binding microtiter plates were coated with an optimal concentration of P insidiosum–concentrated minimal media antigen. After overnight incubation at 4 °C, plates were washed with PBS + 0.05% Tween 20, blocked with 1% normal goat serum for 4 hours at room temperature, and stored at −20 °C until use.
Positive controls were sera from culture-confirmed P insidiosum–infected dogs, and negative controls were from healthy dogs with no cutaneous or GI signs. Patient and control sera were diluted 1:1,000, 1:2,000, and 1:4,000 in PBS + 0.05% Tween 20, applied in triplicate to wells, and incubated at room temperature. After washing, secondary antibody (ALP-conjugated goat anti-dog immunoglobulin G; KPL ReserveAP; SeraCare) was added. Plates were incubated for 1 hour, washed, and developed with p-nitrophenyl phosphate. Optical density (OD) was read at 405 nm using a Synergy HTX microplate reader (BioTek). Blank OD values were subtracted, and titers were calculated using median values as previously described.21
Triplex PCR for P insidiosum, Lagenidium giganteum forma caninum, and Paralagenidium karlingii
Total nucleic acids were extracted from 200 µL of canine serum using an automated magnetic bead–based system (Indical Biosciences), yielding 100 µL of eluent. The triplex quantitative PCR was run on a LightCycler 96 PCR system (Roche) as described previously.15 Polymerase chain reaction products were confirmed by bidirectional Sanger sequencing (ELIM Biopharmaceuticals).
Statistical analysis
Serum samples (n = 123) from 115 dogs with confirmed pythiosis were used to assess the effects of disease presentation and sample dilution on antibody titers. Samples included 83 from dogs with GI involvement and 40 from dogs with cutaneous disease. Each sample was tested in triplicate at 3 serial dilutions (1:1,000, 1:2,000, and 1:4,000) using an in-house ELISA. The mean percentage of positivity was calculated for each dilution and used in statistical analyses. A mixed factorial design was applied, with disease presentation (GI vs cutaneous) as a between-subjects factor and dilution as a within-subject repeated measure. A linear mixed-effects model with random intercepts for individual dogs was used to account for repeated measures and unequal variances. Fixed effects included disease, dilution, and their interaction. Model assumptions were assessed using residual plots and the Levene test. Post hoc pairwise comparisons were adjusted using the Tukey method.
Diagnostic agreement between ELISA and PCR was evaluated using overall concordance and the Cohen κ coefficient (95% CI) for 697 paired samples tested by both methods. Because residuals suggested nonconstant variance across dilutions, a Kruskal-Wallis test was also performed as a nonparametric sensitivity analysis to confirm differences across dilutions. Concordance was defined as both tests yielding the same result (positive or negative). Kappa values were interpreted using standard benchmarks: < 0.20, slight; 0.21 to 0.40, fair; 0.41 to 0.60, moderate; 0.61 to 0.80, substantial; and > 0.80, almost perfect. Chi-square tests were used to compare (1) the prevalence of GI versus cutaneous pythiosis and (2) the proportion of positive results detected by ELISA versus PCR across all samples.
To assess whether certain breed groups were more likely to test positive for Pythium infection, dogs were classified into 2 groups based on primary breed as defined by the American Kennel Club. Sporting/working dogs included breeds traditionally used for hunting, herding, guarding, or similar outdoor activities (eg, Labrador Retrievers, German Shepherds, Goldendoodles, Great Pyrenees, Border Collies). Mixed-breed dogs were initially included with the sporting/working group based on typical medium-to-large body size and outdoor activity, but analyses were also repeated with mixed breeds excluded to assess the robustness of the findings.
Mixed-breed dogs without a clear primary breed were also assigned to this group. Smaller-sized dogs included primarily small indoor or lap breeds (eg, French Bulldogs, Dachshunds, Chihuahuas, Pugs, Yorkshire Terriers). Dogs whose primary breed did not clearly fit either category (eg, Greyhounds, English Bulldogs, Cocker Spaniels) were excluded to maintain consistent classification.
For each dog’s initial diagnostic submission, ELISA results were coded as strict positive (positive only based on ELISA/PCR) or inclusive positive (positive or suspect based on ELISA). For dogs that had multiple submissions, we only used the first (initial) sample to calculate sensitivity, so no single dog was counted more than once. Two-by-two contingency tables were used to compare positivity rates between these 2 groups. The Fisher exact test was applied due to the smaller sample size in the smaller-sized group, with χ2 test results also reported. Odds ratios and risk ratios with 95% CIs were calculated, applying the Haldane-Anscombe correction when needed.
All statistical analyses were performed using Python (version 3.11; scipy and statsmodels packages; Python Software Foundation), R (version 4.4.0; R Foundation for Statistical Computing), and Prism (version 10.4.2; GraphPad Software Inc). Statistical significance was set at P < .05.
Results
Concordance rate of 80% between ELISA and PCR
A total of 796 serum samples from 576 dogs were included. All samples were tested for anti-Pythium antibodies by ELISA, and 697 were also tested for Pythium DNA by PCR.
Across the 697 samples tested by both assays, 69 of 697 (9.9%) were positive and 495 of 697 (71.0%) negative by both tests. Discordant results occurred in 133 of 697 (19.1%), including 5 samples (0.7%) PCR positive but ELISA negative or suspect and 128 of 697 (18.6%) ELISA positive or suspect but PCR negative (Figure 1; Table 1; Supplementary Figure S1; Supplementary Table S1).
Positivity rates of ELISA and PCR in dogs with gastrointestinal (GI) and cutaneous pythiosis based on initial and follow-up sample submissions. Dark-blue bars represent initial diagnostic positives, and light-blue bars represent follow-up positives. Striped regions indicate suspect ELISA results from initial submissions; PCR results were binary (positive or negative). Initial samples reflect diagnostic sensitivity, whereas follow-up samples illustrate test performance during treatment monitoring.
Citation: American Journal of Veterinary Research 2025; 10.2460/ajvr.25.07.0236
Table 1
Diagnostic results and inferred infectious status for pythiosis based on combined ELISA (antibody) and PCR results from 697 canine serum samples.
PCR Antibody No. of submissions Infectious status
Positive Positive 69 Actively infected
Positive Negative 2 Actively infected
Positive Suspect 3 Actively infected
Negative Positive 86 Infected (active or waning)
Negative Suspect 42 Potentially infected
Negative Negative 495 Likely uninfected
Results reflect the interpretation of both assays to classify samples as positive, negative, or suspect for Pythium infection.
The overall concordance rate between ELISA and PCR was 80.9% (564 of 697). The Cohen κ coefficient was 0.44 (95% CI, 0.37 to 0.50), indicating moderate agreement between the 2 assays. This reflects the complementary nature of these tests: ELISA identified nearly all confirmed cases, whereas PCR was more specific for detecting active infections with circulating Pythium DNA.
Of the total submissions, 183 samples were from dogs with confirmed or suspected pythiosis based on external diagnostics. Enzyme-linked immunosorbent assay detected 177 of 183 positives (96.7%), 5 of 183 suspects (2.7%), and 1 of 183 negatives (0.5%). Polymerase chain reaction was performed on 158 of these, yielding 72 of 158 positives (45.5%), including 3 of 4 ELISA-suspect samples. Among these cases, 127 of 183 (69%) were GI, 49 of 183 (27%) cutaneous, and 7 of 183 (4%) unusual or unknown presentations (Supplementary Figure S1; Supplementary Table S1). Across all samples, ELISA showed an overall sensitivity of 96.1%, increasing to 99.4% when suspect results were grouped with positives. Polymerase chain reaction sensitivity was lower at 48.8%, driven by reduced detection in GI cases.
Based on the presentation from 115 dogs with confirmed pythiosis (No. of sera, 123), ELISA sensitivity was found to be 85.0% for cutaneous cases (97.5% including suspect results) and 100% for GI cases, whereas PCR sensitivity was 61.7% for cutaneous and 44.2% for GI cases.
Diagnosis of cutaneous and GI pythiosis by ELISA and PCR
Based on available clinical history, GI pythiosis was significantly more common than cutaneous pythiosis, with 115 individual dogs presenting with GI involvement and 40 dogs presenting with cutaneous lesions (χ2 (1, n = 155) = 70.66; P < .0001). In addition, 1 case had concomitantly cutaneous and GI pythiosis, and 2 cases were thoracic.
Among initial diagnostic submissions from dogs with confirmed cutaneous pythiosis, ELISA (n = 40) identified 34 of 40 positive cases (87.5%) and 5 of 40 suspect cases (12.5%), whereas PCR (34) detected P insidiosum DNA in 21 of 34 cases (61.7%), including 3 that were classified as suspect by ELISA. When suspect ELISA results were excluded from analysis, ELISA demonstrated a significantly higher detection rate than PCR (χ2 (1, n = 34) = 11.25; P = .0008), indicating greater diagnostic sensitivity of ELISA at the time of initial evaluation. When suspect results were grouped with positives, the difference became even more pronounced (χ2 (1, n = 34) = 13.06; P = .0003), reflecting the importance of consistent interpretation of borderline results. Notably, PCR identified a substantial proportion of cutaneous cases and provided molecular confirmation in several suspect ELISA cases. These findings underscore the complementary roles of ELISA and PCR in cutaneous pythiosis diagnosis and highlight how classification of borderline serologic results can meaningfully affect diagnostic performance metrics.
For initial submissions from dogs with confirmed GI pythiosis (n = 115), ELISA identified all cases (100%), whereas PCR (95) detected P insidiosum DNA in 42 cases (44.2%; Figure 1; Table 2). This difference in detection rates was highly significant (χ2 (1, n = 95) = 82.89; P < .0001), indicating a higher sensitivity of ELISA in detecting positive GI cases. Both thoracic cases—an uncommon presentation of pythiosis—were positive by ELISA and PCR, highlighting the utility of these noninvasive assays for diagnosing atypical manifestations of infection.
Table 2
Comparison of ELISA and PCR detection rates in dogs with confirmed gastrointestinal and cutaneous pythiosis based on initial diagnostic submissions.
Presentation Test No. (%) positive No. (%) suspect No. (%) negative Total χ2 test P value
Gastrointestinal pythiosis ELISAa 115 (100) 0 (0) 0 (0) 115 < .001
PCR 42 (44.2) N/A 53 (55.7) 95
Cutaneous pythiosis ELISA 34 (85.0) 5 (12.5) 1 (2.5) 40 .0008
PCR 21 (61.7) N/A 15 (48.4) 34
N/A = Not applicable.
Cases were confirmed by culture, PCR, and/or histopathology performed at outside diagnostic laboratories, and results are based on the initial diagnostic submissions for each dog. Enzyme-linked immunosorbent assay demonstrated significantly higher detection rates than PCR in both gastrointestinal and cutaneous pythiosis cases. However, PCR confirmed Pythium DNA in 3 cutaneous samples that were classified as suspect by ELISA, highlighting its value as a complementary tool in cases with borderline serologic results.
Among follow-up samples submitted for antibody monitoring following an initial diagnosis of GI pythiosis (n = 123), ELISA identified 18 positive, 32 suspect, and 73 negative results. Polymerase chain reaction detected Pythium DNA in 4 of these samples. In recheck samples from dogs with cutaneous pythiosis (n = 32), ELISA yielded 8 positive, 9 suspect, and 15 negative results, whereas PCR detected DNA in 3 samples, including 1 that was classified as suspect by ELISA. These findings reflect a decline in both antibody levels and circulating pathogen DNA following treatment, which can reduce the sensitivity of both assays during follow-up testing.
Interestingly, the mean copy numbers of Pythium DNA detected by PCR were not significantly different between cutaneous and GI cases (cutaneous, 5,317 ± SD 5,068/mL serum; GI, 5,338 ± SD 5,095/mL serum). However, ELISA antibody titers were significantly higher in GI pythiosis than in cutaneous pythiosis at all dilutions (P < .0001; Figure 2).
Enzyme-linked immunosorbent assay positivity at serum dilutions in dogs with GI and cutaneous pythiosis. Each box represents the IQR, with the median indicated by the horizontal line. Whiskers extend to the minimum and maximum values within 1.5 times the IQR; dots indicate outliers. Dogs with GI pythiosis exhibited significantly higher antibody levels than those with cutaneous form (P < .0001). A linear mixed-effects model revealed significant effects of both disease presentation (P < .0001) and dilution (P < .0001) on ELISA titers, with no significant interaction between these factors (P < .1129). Titers also varied significantly across dilutions, confirmed by a Kruskal-Wallis test (H = 7.90; P < .0192) due to nonconstant variance (Spearman Rs = –0.6555; P < .0001).
Citation: American Journal of Veterinary Research 2025; 10.2460/ajvr.25.07.0236
The linear mixed-effects model revealed significant main effects of both disease presentation (P < .0001) and serum dilution (P < .0001) on ELISA antibody levels, with no significant interaction between these factors (P = .1129). Antibody levels in GI cases were consistently higher than in cutaneous cases across all 3 dilutions in initial sample submissions (Figure 2). Titers also varied significantly across dilutions as confirmed by a Kruskal-Wallis test (H = 7.90; P = .0192).
Antibody levels typically declined following successful treatment (Figure 3), with titers falling to ≤ 20% positivity at all 3 dilutions as infections resolved. Conversely, when surgical resection was incomplete or medical management failed, P insidiosum could persist subclinically, resulting in rising ELISA titers, which often preceded the return of clinical signs. In some cutaneous cases, particularly those treated with limb amputation or complete excision, antibody titers dropped rapidly and remained low, reaching normal levels at all dilutions by 6 months after surgery. In contrast, cases where resection was not feasible or incomplete showed an initial decline in titers with treatment, followed by either persistently elevated levels (often classified as suspect) or a secondary spike, indicative of ongoing infection. Polymerase chain reaction showed the absence of Pythium DNA decline after treatment.
Longitudinal serologic response in dogs diagnosed with pythiosis. Each panel shows serial ELISA results for an individual dog, with percentage of positive values over time by 3 dilutions. Month 0 marks the data from the initial submission, and the dotted lines indicate the 40% positivity cutoff. Most dogs showed declining titers after diagnosis, though some had persistent or rebounding antibody levels, suggesting ongoing antigen exposure or incomplete clearance. The gold box in A at month 0 represents a positive PCR on the initial sample, whereas a PCR was not performed on the initial samples for any of the other animals. The cases in A and B were cutaneous, whereas the remaining 4 cases were in GI presentation.
Citation: American Journal of Veterinary Research 2025; 10.2460/ajvr.25.07.0236
Multiple submissions
Fifty-three cases had more than 1 sample submission. For each, serologic and molecular results were evaluated in the context of clinical history.
ELISA- and PCR-negative cases
Twenty-two cases, each with up to 5 submissions, were consistently negative by both ELISA and PCR. These were typically follow-up samples from previously diagnosed and treated cases submitted over months to years to monitor treatment response.
Case 1: A cutaneous pythiosis case initially diagnosed by culture and ELISA, with multiple negative follow-up submissions during treatment.
Case 2: Diagnosed with GI pythiosis by ELISA in May 2023 and enrolled in a clinical trial. Samples submitted ≥ 6 months after treatment began were negative by both tests, and the dog was clinically normal.
ELISA- and PCR-positive cases
Four cases tested positive by both ELISA and PCR, with intervals of up to 3 months between submissions.
Case 3: Cutaneous pythiosis diagnosed in December 2023; initial positive ELISA and subsequent recurrence of lesions in February 2024 with PCR positivity.
Case 4: Diagnosed in August 2024; persistent cutaneous lesions and strong PCR positivity in October 2024 despite ELISA titers falling to the suspect range.
Case 5: Chronic GI pythiosis with incomplete surgical resection. Enzyme-linked immunosorbent assay remained positive across multiple submissions; PCR was initially and finally positive but negative at 1 intermediate time point.
ELISA-negative and PCR-positive cases
Case 6: Presented with GI signs and masses. Two samples (January 19 and February 7, 2025) were ELISA negative (≤ 7% at all dilutions), but the first was PCR positive and the second PCR negative.
ELISA-positive/suspect and PCR-negative cases
Twenty-one cases were ELISA positive or suspect but PCR negative. Many were follow-up submissions from treated animals, where circulating antibody levels may have persisted while DNA was no longer detectable.
Case 7: A cutaneous case with 8 submissions from November 2023 through October 2024. Enzyme-linked immunosorbent assay titers declined throughout the course of treatment, and all PCR results were negative.
Case 8: Gastrointestinal case with 3 submissions. Enzyme-linked immunosorbent assay was positive on the first and negative on the remaining 2, and all PCR tests were negative.
Breed might be a risk factor for canine pythiosis
When considering cases that were PCR positive or ELISA positive/suspect as pythiosis, active outdoor breeds appeared more likely to be infected than smaller, traditionally smaller-sized dogs (Table 3). Sporting and working breeds (eg, Great Pyrenees [9 of 16 (56%)], Goldendoodle [8 of 18 (44%)], German Shorthaired Pointer [10 of 23 (43%)]) had higher positivity rates than predominantly smaller-sized breeds (eg, French Bulldog [1 of 13 (8%)], Dachshund [0 of 13 (0%)], other smaller-sized breeds [1 of 24 (4%)]; Supplementary Table S2). Among the 599 dogs classified as either sporting or smaller-sized breeds, 136 were strict positives (ELISA/PCR positive), and 160 were inclusive positives (ELISA positive or suspect). Sporting/working dogs had a significantly higher positivity rate than smaller-sized dogs. Using the Strict analysis, sporting/working dogs were 3.5 times more likely to test positive (Fisher exact P = .00088). When suspect cases were included, the association strengthened, with sporting/working dogs 4.4 times more likely to test positive (Fisher exact P = 2.31 X 10−5). To reassess the potential subjectivity of assigning mixed-breed dogs to the sporting/working group, we repeated the analysis with mixed breeds excluded. The results were unchanged, with sporting/working dogs still significantly more likely to test positive than smaller, traditionally indoor-type dogs under both strict (Fisher exact P = .0129) and inclusive (Fisher exact P = .0010) definitions. These findings indicate that sporting/working dogs are at a significantly greater risk of pythiosis, likely due to increased environmental exposure to aquatic habitats where P insidiosum thrive.
Table 3
Comparison of Pythium positivity rates between sporting/working and smaller-sized dogs.
Group Negative (strict) Positive (strict) Negative (inclusive) Positive (inclusive)
Smaller sizeda 74 7 74 7
Sporting/workingb 389 129 365 153
Total 463 136 439 160
Diagnostic submissions were classified as sporting/working (n = 518) or smaller sized (81). Strict positives include only positive results by ELISA and/or PCR, whereas inclusive positives include both positive and suspect results by ELISA. Sporting/working dogs were significantly more likely to test positive for pythiosis than smaller-sized dogs. In the strict analysis, the Fisher exact test yielded P = .00088; OR, 3.51 (95% CI, 1.57 to 7.80); and risk ratio, 2.88 (95% CI, 1.40 to 5.94). In the inclusive analysis, P = 2.31 X 10−5; OR, 4.43 (95% CI, 2.00 to 9.84); and risk ratio, 3.42 (95% CI, 1.66 to 7.02). These results indicate that sporting/working dogs were about 3.5 to 4.4 times more likely to test positive for pythiosis than smaller-sized dogs.
Smaller-sized dogs include French Bulldogs, Chihuahuas, and Pugs.
Sporting/working dogs include Labrador Retrievers, Border Collies, and mixed breeds. Breeds not clearly fitting either group were excluded.
Lagenidium-positive cases by PCR
The triplex PCR identified 6 Lagenidium-positive cases; no cases were positive for Paralagenidium. All 6 cases were reported as cutaneous presentations of pythiosis and were negative by ELISA and PCR for Pythium. In 4 of 6 cases, histopathology described hyphae suggestive of Pythium, and several were chronic or recurrent. One had previously been misdiagnosed as P insidiosum based solely on histopathology.
Case 9: A cutaneous case with a history of disseminated multifocal necrotic lesions requiring multiple aggressive surgical excisions and medical management with recurrence at the surgical site. Hyphae were noted on histopathology. Enzyme-linked immunosorbent assay negative for P insidiosum and PCR positive for Lagenidium.
Case 10: A cutaneous case was evaluated with 2 sample submissions taken 4 months apart. The initial diagnosis was pythiosis based on histopathologic evidence of hyphae despite an inconclusive pan-fungal PCR and a negative ELISA. Upon reevaluation, the second sample tested positive for Lagenidium via serum PCR, whereas the Pythium ELISA remained negative.
Discussion
This study presents the largest comparative evaluation of anti-Pythium antibody ELISA and serum-based PCR for diagnosing canine pythiosis. By integrating clinical history with test results, we provide new insights into the strengths and limitations of both diagnostic tools.
Our results support ELISA as a highly sensitive tool for both diagnosis and monitoring of pythiosis, particularly in cases with GI involvement. The ELISA used to detect anti–P insidiosum serum antibodies in this study was based on previously described protocols,16,20 with minor modifications. Specifically, instead of a single dilution as used in the original protocols, serum samples in this study were tested at dilutions of 1:1,000, 1:2,000, and 1:4,000. Each sample was then categorized as positive, suspect, or negative based on a 40% cutoff of the OD value. This 3-tiered ELISA interpretation system more accurately reflected the clinical condition than the original single-dilution approach.
Diagnostic sensitivity was found to be highest in this study when evaluating initial submissions, with ELISA identifying 182 of 183 (99.4%) confirmed positive cases as positive or suspect and PCR detecting 72 of 158 (45.6%). In contrast, sensitivity declined in follow-up samples from dogs undergoing treatment. Among follow-up samples submitted for antibody monitoring after an initial GI pythiosis diagnosis (n = 123), ELISA identified 18 of 123 positive (14.6%), 32 of 123 suspect (26.0%), and 73 of 123 negative (59.3%) results, whereas PCR detected Pythium DNA in only 4 cases. For cutaneous follow-up samples (n = 32), ELISA yielded 8 of 32 positive (25%), 9 of 32 suspect (28.1%), and 15 of 32 negative (46.9%) results, whereas PCR detected DNA in 3 of 32 samples (9.4%). This decline in detection reflects the expected reduction in both circulating antibody and pathogen DNA levels following effective treatment.12
Enzyme-linked immunosorbent assay testing on multiple submissions from the same dogs demonstrated that anti-Pythium antibody titers decline predictably over time and may signal clinical improvement. However, persistently elevated or suspect ELISA results often indicated incomplete resolution or relapse even in animals that appeared clinically normal, particularly in the postoperative setting.22 Thus, ELISA serves as an early diagnostic tool in chronic or recurrent cases.12,16 Moreover, serial monitoring using both ELISA and PCR may help identify subclinical recurrence before clinical signs reappear.23 In one such case, PCR detected Pythium DNA in a sample with a suspect ELISA result, underscoring the value of molecular confirmation when antibody levels are low.
Although PCR detection rates were numerically higher for cutaneous pythiosis (61.7%) compared to GI cases (44.2%), this difference was not statistically significant. However, PCR may still offer critical diagnostic value in cutaneous presentations, where anti-Pythium antibody titers tend to be lower and serologic results may be more ambiguous.23 Given the overlap in clinical and histologic appearance between cutaneous pythiosis and other oomycotic infections—such as Lagenidium or Paralagenidium—PCR’s ability to directly detect and differentiate pathogen DNA in serum can enhance diagnostic accuracy.8,15,23 This is especially valuable when ELISA results are borderline or inconclusive. In such cases, integrating PCR as a confirmatory tool can help resolve diagnostic uncertainty and guide appropriate clinical management. These findings support the complementary use of ELISA and PCR not only for diagnosis but also for treatment monitoring and long-term disease surveillance in dogs with pythiosis.12,23
Serum-based PCR, used here for the first time in this context, offers direct evidence of active infection by detecting circulating P insidiosum DNA. Unlike traditional tissue-based PCR,24,25 this approach is less invasive and more practical for systemic infections. While PCR had lower overall positivity than ELISA, its specificity for active infection makes it a valuable complement, especially when ELISA results are ambiguous or delayed due to host immune response.26 Serum was used for PCR in this study, but EDTA plasma, especially the buffy coat and tissues, may yield more DNA and improve PCR sensitivity. Future research should evaluate optimal sample types for molecular diagnostics in pythiosis.
Notably, some animals tested PCR positive but ELISA negative, suggesting early or silent infections.19,27 Others were ELISA positive but PCR negative, likely due to antibody persistence following treatment or low pathogen load in localized infections. These findings underscore the importance of using both tests in tandem for comprehensive diagnosis and monitoring.
The multiplex PCR used in this study also distinguished Pythium from Lagenidium, 2 morphologically similar oomycetes with different treatments.15 Six cutaneous cases initially suspected of being Pythium but negative by ELISA were confirmed to be Lagenidium, highlighting the diagnostic value of molecular testing over histopathology alone as well as the specificity of the ELISA.
Environmental exposure appears to play a role in disease risk.3,28,29 Sporting/working dogs showed significantly higher pythiosis rates, supporting the association with swampy or aquatic habitats.30 Smaller-sized dogs had lower positivity, reinforcing the environmental nature of exposure.
In total, 69 samples were positive by both ELISA and PCR, and 3 suspect ELISA samples were positive by PCR, suggesting the possible presence of Pythium immune complexes in serum or plasma, similar to what is observed in heartworm disease.31,32 Studies using heat or chemical pretreatment of serum may enhance ELISA sensitivity and deserve further investigation.31
In summary, this study provides the most comprehensive comparison of anti-Pythium antibodies by ELISA and PCR detection of Pythium DNA in sera from canines with suspected and active pythiosis to date. Enzyme-linked immunosorbent assay remains the cornerstone for diagnosing and monitoring pythiosis, and serum PCR introduces a novel noninvasive tool to confirm active infection and differentiate between pathogenic oomycetes. Used together, they offer a comprehensive diagnostic strategy for managing this challenging disease. Future studies should focus on understanding antigenemia kinetics and validating this dual-testing approach across host species and regions.
Supplementary Materials
Supplementary materials are posted online at the journal website: avmajournals.avma.org.
Acknowledgments
None reported.
Disclosures
The authors have nothing to disclose. No AI-assisted technologies were used in the composition of this manuscript.
https://avmajournals.avma.org/view/journals/ajvr/aop/ajvr.25.07.0236/ajvr.25.07.0236.xml
_________________________________________________________________________________________________________________________________
BMC Veterinary Research volume 21, Article number: 605 – Published: October 14, 2025
The impact of anxiety on canine heart function: a study using echocardiographic techniques
Delara Soghyani, Farnoosh Arfaee, Mohammad Molazem, Reihaneh Soflaei & Arman Abdous
Abstract
Canine anxiety is increasingly recognized in veterinary practice, yet its cardiovascular effects remain poorly defined. This study evaluated 36 anxious dogs and 20 healthy controls using the validated Canine Behavioral Assessment and Research Questionnaire (C-BARQ) and standardized echocardiography. Significant associations were identified between anxiety subtypes and cardiac parameters: general anxiety correlated with increased fractional shortening (FS%; r = 0.663, p = 0.001), phonophobia with larger left ventricular internal dimension in diastole (LVIDd; r = 0.681, p < 0.001), and aggression with a higher left atrial-to-aortic root ratio (LA/Ao; r = 0.475, p = 0.008). Rhythm irregularities were suspected in nearly half of anxious dogs, though classification was limited without ECG confirmation. By contrast, correlations in controls were weak and non-significant.These results provide novel evidence that chronic behavioral anxiety is associated with measurable echocardiographic alterations in dogs, paralleling mechanisms described in human psychocardiology. Incorporating behavioral screening into routine veterinary practice may support early identification and monitoring of dogs at risk for anxiety-related cardiac changes. Larger longitudinal studies with matched controls, ECG verification, and size-indexed parameters are warranted to confirm causality and guide preventive care strategies.
Introduction
Anxiety in dogs is an increasingly recognized concern in veterinary medicine, affecting a significant portion of the canine population [1]. Conditions such as separation anxiety, noise phobia, and generalized fear can lead to behavioral disturbances and reduce quality of life. Emotional stress in animals has been associated with various physiological consequences, including gastrointestinal disorders and compromised immune function; however, its specific impact on cardiovascular health remains underinvestigated [2, 3].
In contrast, a large body of human research links anxiety to cardiovascular dysfunction. Chronic anxiety in people is associated with altered heart rate variability, heightened sympathetic tone, and structural changes such as diastolic dysfunction and atrial enlargement—often detectable through echocardiography. Although parallels between human and canine physiology suggest similar psychocardiological mechanisms may occur in dogs, most veterinary studies have focused on physical stressors like exercise or anesthesia rather than emotional triggers [4, 5]. Consequently, the influence of chronic behavioral anxiety on canine cardiac function, particularly as assessed by advanced non-invasive echocardiographic techniques, remains largely unexplored [3].
Canine heart disease itself can arise from congenital abnormalities, acquired degenerative conditions such as mitral valve disease, or myocardial disorders including cardiomyopathies [6, 7]. Clinical signs may include fatigue, coughing, exercise intolerance, and dyspnea. Echocardiography—using B-mode, M-mode, and Doppler modalities—has become a cornerstone of cardiac diagnostics in veterinary practice. It provides quantitative insight into chamber size, systolic function, wall motion, and blood flow dynamics [8]. Commonly evaluated parameters include left ventricular internal dimensions at systole and diastole (LVIDs and LVIDd), ejection fraction (EF%), fractional shortening (FS%), and the left atrial-to-aortic root ratio (LA/Ao) [9].
This study was designed to address the knowledge gap by comparing echocardiographic parameters in anxious dogs and clinically healthy, non-anxious controls. By integrating validated behavioral assessment tools with echocardiographic evaluation, we aimed to determine whether chronic emotional stress contributes to subclinical or early-stage cardiovascular changes. Identifying such links could support the incorporation of behavioral screening into cardiac risk assessment, potentially enabling earlier intervention and improved clinical outcomes.
Methods
Study design
This observational, cross-sectional study was conducted over a six-month period at the Islamic Azad University, Science and Research Branch. Its objective was to assess whether anxiety-related behaviors are associated with alterations in canine cardiac parameters measured through advanced echocardiographic techniques.Dogs were assigned to either an anxiety group or a control group based on behavioral assessment. The anxiety group included dogs with owner-reported signs such as excessive fear of loud noises, separation-related distress, or generalized nervousness. The control group consisted of clinically healthy, non-anxious dogs. Both groups underwent identical echocardiographic protocols, enabling direct comparison of cardiac function. An overview of the experimental workflow is provided in Supplementary Figure S1.
Ethical considerations
The study was conducted in accordance with institutional ethical standards and approved by the Ethics Committee of the Islamic Azad University, Science and Research Branch, Tehran. Prior to participation, owners of all dogs in both groups received detailed written and verbal information about the study’s objectives, procedures, and potential risks. Written informed consent was obtained, and participants were assured of their right to withdraw at any time without consequence.All echocardiographic procedures were performed by licensed veterinary professionals with specialized training in cardiology. Humane treatment guidelines for animal research were strictly followed, with gentle handling and procedures designed to minimize stress and ensure animal welfare.
Study population
A total of 74 dogs were screened at the hospital of the Islamic Azad University, Science and Research Branch (Tehran), and affiliated veterinary clinics. Eighteen were excluded—11 due to current or recent use of psychotropic medications and 7 due to comorbid medical conditions—leaving 56 dogs enrolled. Of these, 36 met the criteria for the anxiety group, while 20 clinically healthy, non-anxious dogs served as controls.The anxious group included dogs aged 1–12 years, representing a diverse mix of breeds and sizes to enhance generalizability. Inclusion criteria required owner-reported signs of anxiety—such as noise sensitivity, separation distress, or nervousness in unfamiliar situations—persisting for at least three months, confirmed through history or clinical documentation. Dogs with prior diagnoses of heart disease were excluded to isolate the effect of behavioral anxiety on cardiac function. Additional exclusion criteria included cardiovascular, neurological, or systemic conditions that might affect echocardiographic results, as well as treatment with medications for anxiety or behavioral disorders (e.g., trazodone, fluoxetine, gabapentin).The control group consisted of 20 dogs free of cardiac or behavioral disorders, with consistently low C-BARQ scores (mean global anxiety score: 0.64 ± 0.18). Their mean age was 4.9 ± 3.0 years (range 1.1–9.7), and body weights ranged from 4.4 to 15.0 kg. To minimize environmental and handling differences, control dogs were drawn from the same clinical population and underwent identical echocardiographic evaluation.
Behavioral assessment of anxiety
Behavioral anxiety was quantified using an adapted version of the Canine Behavioral Assessment and Research Questionnaire (C-BARQ), a standardized instrument widely applied in clinical behavioral research to characterize emotional reactivity in dogs. Six domains were evaluated: separation anxiety, noise sensitivity (phonophobia), fear, aggression, obsessive-compulsive behaviors, and generalized anxiety. Owners rated the frequency of each behavior on a 5-point Likert scale, where 0 indicated “never,” 1 “rarely,” 2 “sometimes,” 3 “often,” and 4 “always.” Observable signs such as excessive barking, destructiveness when left alone, trembling, hiding, or hypervigilance were used to guide accurate scoring.Following completion of the questionnaire, total anxiety scores were calculated as the unweighted arithmetic mean of responses across the six domains, providing a standardized measure of each dog’s overall anxiety level. Dogs were then stratified into three categories: scores below 1.5 were classified as low anxiety, scores between 1.5 and 2.5 as moderate anxiety, and scores above 2.5 as high anxiety. Control dogs consistently demonstrated low scores across all domains, confirming their non-anxious status.
Echocardiographic assessment
Echocardiographic examinations were performed by a board-certified veterinary cardiologist using a Philips EPIQ 7 ultrasound system equipped with high-resolution B-mode, M-mode, and Doppler functionalities. A sector-array phased transducer probe with a frequency range of 3.5–8 MHz was used for thoracic imaging, as it provides deep penetration with high temporal resolution. For very small dogs (< 3 kg), higher-frequency probes (>8 MHz) were employed to enhance spatial resolution and minimize measurement error, ensuring consistent image quality across the range of body sizes represented in the study.This imaging platform enabled detailed evaluation of cardiac structure and function, including real-time assessment of ventricular wall motion, valve morphology, and blood flow dynamics. Standard veterinary echocardiographic protocols were followed [10] Key measurements included left ventricular internal dimensions at end-systole (LVIDs) and end-diastole (LVIDd), interventricular septal thickness (IVS), fractional shortening (FS%), and ejection fraction (EF%). These indices are central to evaluating myocardial size, contractile performance, and systolic function. M-mode echocardiography was used for precise dimensional analysis of the left ventricle, B-mode imaging for overall morphology and detection of abnormalities such as wall motion irregularities or chamber dilation, and Doppler for intracardiac blood flow and identification of valvular regurgitation across the mitral, aortic, and tricuspid valves. Together, these methods provided comprehensive insight into both structural and hemodynamic aspects of cardiac performance.All echocardiographic data were recorded systematically and analyzed in relation to behavioral anxiety classifications. Both anxious and control dogs underwent identical assessment protocols during a single clinical visit, allowing for direct comparison of structural and functional parameters and evaluation of the potential influence of emotional stress on cardiac function.
Statistical analysis
Analyses were conducted using IBM SPSS Statistics v26. Descriptive statistics (means, standard deviations, and frequencies) were used to summarize demographic, behavioral, and echocardiographic data. Linear associations between anxiety scores, age, and cardiac parameters were assessed with Pearson’s correlation coefficients. To control for multiple comparisons, p-values were adjusted using the false discovery rate (FDR), and only associations that remained significant after correction were interpreted. Statistical significance was defined as two-tailed p < 0.05. Results are reported with correlation coefficients, 95% confidence intervals, effect sizes, and p-values, with selected associations illustrated graphically. A post-hoc power analysis (α = 0.05) showed that the sample size (36 anxious and 20 control dogs) provided > 80% power to detect between-group differences of medium effect size (Cohen’s d ≥ 0.65) and correlations of r ≥ 0.44 within the anxious cases.
Results
Demographic and sample characteristics
The anxious group consisted of 36 dogs ranging in age from 1 to 12 years, with the largest proportions being 1 year (19.4%) and 2 years (16.7%). Senior dogs, defined as those aged 7 years or older for small to medium breeds, represented 25% of the anxious cases. Breed diversity was notable: Shiba Inu (22.2%), Maltese (19.4%), Miniature Poodle (16.7%), and Jack Russell Terrier (13.9%) were the most common, while French Bulldog, Terrier, Shih Tzu, and Bichon Frise were also represented, creating a heterogeneous sample of small- to medium-sized companion breeds.
Body-weight distribution in the anxious group was skewed toward very small dogs, with 58.3% weighing less than 3 kg. Dogs in the 3–5 kg and 5–10 kg categories accounted for 19.4% and 13.9% of the group, respectively, while 8.3% weighed between 10 and 15 kg. This pattern likely reflects regional breed preferences and the higher prevalence of anxiety in toy and miniature breeds.
The control group (n = 20) consisted of clinically healthy dogs with a mean age of 4.9 ± 3.0 years (range 1.1–9.7). Their body weights ranged from 4.4 to 15.0 kg, and breed distribution is shown in Table 1. In contrast to the anxious group, which was predominantly composed of dogs under 3 kg, the control group displayed a more balanced distribution across weight categories. Age was further examined in relation to both behavioral anxiety scores and echocardiographic variables, with correlation patterns presented in Sect. 3 and summarized in Table 2.
Table 1 Demographic characteristics of anxious dogs (n = 36) and control dogs (n = 20)
Table 2 Echocardiographic measurements and distribution relative to reference ranges in anxious dogs (n = 36) and control dogs (n = 20)
Cardiovascular parameter results
As summarized in Table 2, echocardiographic assessments revealed several findings related to cardiac structure and function. In the anxious group, the mean left ventricular internal dimension at end-systole (LVIDs) was 24.49 ± 4.90 mm, the average ejection fraction (EF%) was 73.61 ± 13.95%, and fractional shortening (FS%) averaged 42.14 ± 10.96%. The left atrial-to-aortic root ratio (LA/Ao) had a mean of 1.47 ± 0.24 (range 1.0–2.0).
When compared with uniform reference ranges, several parameters fell outside the suggested limits. LVIDd was above range in 33.3% and below in 5.6% of anxious dogs. FS% exceeded the standard 25–45% range in 44.4%, while LA/Ao was greater than the conventional 1.3 cutoff in 80.6%. These proportions should be interpreted cautiously, as fixed reference values may not fully account for body size differences. In contrast, interventricular septal thickness in diastole (IVSd) was within the normal range in all anxious dogs. A detailed breakdown of deviations is provided in Table 2.The control group showed values largely within normal limits. Mean LVIDd was 36.50 ± 4.00 mm, FS% was 34.50 ± 5.00%, and EF% was 67.50 ± 7.00%, with only minor deviations observed (e.g., 15% of dogs with LVIDd above reference and 10% with EF% above reference). No significant structural or functional abnormalities were detected in this group.
Suspected rhythm irregularities were identified in 47% (17/36) of anxious dogs based on auscultation and echocardiographic observation. The most frequent pattern was a non-specific irregular rhythm (38.9%), followed by findings suggestive of conduction disturbances (22.2%). Less common abnormalities included single cases of tachycardia, bradycardia, and murmurs possibly consistent with mitral regurgitation. As ECG confirmation was not available, these should be regarded as provisional rather than definitive diagnoses. Categories were not mutually exclusive, since some dogs exhibited more than one finding. The distribution of abnormalities is illustrated in Fig. 1.
Fig. 1
Frequency of detected rhythm and auscultatory irregularities in anxious dogs (n = 36) compared with control dogs (n = 20). Values reflect findings based on auscultation and echocardiographic observation. Arrhythmia types remain non-specifically categorized due to absence of ECG; no abnormalities were observed in control dogs. Categories are not mutually exclusive; a dog could contribute to more than one finding, and percentages therefore do not sum to 100%
Correlation between anxiety and cardiovascular parameters
Statistical analysis revealed significant correlations between specific anxiety subtypes and key echocardiographic parameters in anxious dogs. General anxiety was positively associated with fractional shortening (FS%) (r = 0.663, p = 0.001, q = 0.004). Phonophobia showed a strong positive correlation with left ventricular internal dimension at end-diastole (LVIDd) (r = 0.681, p < 0.001, q = 0.004). Although 61.1% of anxious dogs had LVIDd values within the reference range, those with higher phonophobia scores tended toward larger or above-reference diastolic dimensions, which may partly reflect body size variation or measurement variability. Aggression was correlated with the left atrial-to-aortic root ratio (LA/Ao) (r = 0.475, p = 0.008, q = 0.016); however, because LA/Ao values exceeded 1.3 in most anxious dogs, this association should be regarded as exploratory (Table 3, Fig. 2).By contrast, none of these associations were significant in the control group (all p > 0.6, r < 0.2), indicating that the observed relationships between anxiety traits and cardiac parameters were specific to anxious dogs.
Table 3 Correlation Analyses Related to Anxiety and Cardiac Function. Pearson correlations between anxiety subtypes and echocardiographic parameters in anxious (n = 36) and control (n = 20) dogs
Fig. 2
Mean ± standard deviation (SD) of anxiety-related behavioral parameters in anxious dogs (n = 36) compared with control dogs (n = 20), based on C-BARQ questionnaire results. Control dogs consistently exhibited minimal scores across all domains, whereas anxious dogs showed markedly elevated scores, particularly in phonophobia, fear, and separation anxiety
Heart rate also showed significant associations with multiple cardiac indices in the anxious cases. It correlated strongly with left ventricular free wall thickness in systole (LVFWs) (r = 0.833, p < 0.001, q = 0.002), FS% (r = 0.663, p = 0.001, q = 0.006), and EF% (r = 0.644, p = 0.002, q = 0.008) (Table 4, Fig. 2). A positive correlation was also observed with LVIDd (r = 0.681, p < 0.001, q = 0.002), although this finding contrasts with physiological expectations, as tachycardia typically reduces diastolic filling. This pattern may therefore reflect confounding by body size, anxiety severity, or measurement variability. Taken together, these results suggest synchronized increases in contractility and myocardial wall thickness in dogs with higher heart rates. In the control group, heart rate correlations with echocardiographic indices were weak and non-significant (all p > 0.2).
Table 4 Correlation Analyses Related to Anxiety and Cardiac Function. Correlation coefficients between heart rate and echocardiographic parameters, and among cardiac metrics, in anxious (n = 36) and control (n = 20) dogs
Further analysis of inter-parameter relationships (Table 5) demonstrated several significant associations in anxious dogs. FS% correlated positively with interventricular septal thickness in systole (IVSs; r = 0.401, p = 0.048, q = 0.049) and negatively with LVIDs (r = − 0.476, p = 0.029, q = 0.041). EF% was also negatively correlated with LVIDs (r = − 0.452, p = 0.037, q = 0.046). LA/Ao correlated significantly with IVSs (r = 0.389, p = 0.041, q = 0.049), LVIDs (r = 0.427, p = 0.042, q = 0.049), and FS% (r = 0.498, p = 0.025, q = 0.036). These findings indicate consistent internal relationships between functional and geometric cardiac parameters in anxious dogs, though interpretation is limited by the use of uniform reference ranges. In the control group, such interrelationships were again weak and non-significant (r < 0.2, p > 0.5).
Table 5 Correlation Analyses Related to Anxiety and Cardiac Function. Interrelationships between cardiac structural and functional indices in anxious (n = 36) and control (n = 20) dogs
Age also influenced several structural measures. Younger anxious dogs exhibited lower IVSs, with a positive but non-significant correlation between age and IVSs (r = 0.291, p = 0.084). By contrast, LVFWd and LVFWs were strongly correlated (r = 0.843, p < 0.001, q = 0.002), reflecting consistent myocardial wall thickening across age groups. These patterns were observed in both anxious and control dogs, although statistical significance was reached only in the anxious cases.
Anxiety profile distribution and summary
Behavioral anxiety scores in the anxious group (n = 36) demonstrated wide variability across domains. Fear-related behaviors were among the most prominent: 33.3% of dogs scored between 2 and 3, while 44.4% scored between 3 and 4, indicating moderate to high levels of fearfulness. Aggression scores, by contrast, were consistently low, with 41.7% of dogs scoring below 1 and 55.6% between 1 and 2; notably, none exceeded a score of 3.Phonophobia showed the greatest variability. About 27.8% of anxious dogs scored between 2 and 3, while 36.1% scored above 4, reflecting heightened sensitivity to loud or sudden noises. General anxiety occurred mainly at low to moderate levels, with 50.0% of dogs scoring between 1 and 2 and 47.2% between 2 and 3. Separation anxiety displayed a more balanced distribution: 41.7% scored between 2 and 3, 22.2% between 1 and 2, and 8.3% between 3 and 4. Obsessive-compulsive behaviors (OCD) were least common, with 63.9% of anxious dogs scoring below 1 and only 5.6% scoring 2 or higher.In contrast, the control group (n = 20) consistently showed minimal anxiety-related behaviors. Their mean C-BARQ scores ranged from 0.20 ± 0.18 for obsessive-compulsive behaviors to 0.64 ± 0.18 for general anxiety, confirming their role as a non-anxious comparator group (Table 6; Fig. 3).As illustrated in Fig. 3, phonophobia had the highest mean score among anxious dogs (2.74 ± 1.15), followed by fear (2.24 ± 0.89) and separation anxiety (1.96 ± 1.01). The lowest mean scores were recorded for aggression (0.57 ± 0.54) and obsessive-compulsive behavior (0.42 ± 0.62), consistent with their limited prevalence in this population. Control dogs remained near baseline across all domains. Full categorical distributions are provided in Table 6.
Table 6 Distribution of anxiety-related behavioral scores (C-BARQ) across six domains in anxious dogs (n = 36), with mean scores for the control group (n = 20)
Fig. 3
Correlation heatmaps comparing anxious dogs (n = 36) and control dogs (n = 20). Pearson correlation coefficients (r) are shown for behavioral anxiety subtypes, heart rate, and echocardiographic parameters. Darker shades denote stronger correlations. Asterisks (*) indicate correlations that remained statistically significant after Benjamini–Hochberg false discovery rate (FDR) correction across the family of correlation tests (q < 0.05). Data correspond to Tables 3, 4 and 5
Discussion
This study explored the association between behavioral anxiety and echocardiographic parameters in dogs, revealing measurable cardiac changes linked to chronic emotional stress. General anxiety was significantly correlated with increased fractional shortening (FS%), suggesting a hyperdynamic systolic state likely driven by sympathetic activation, while phonophobia was associated with relatively greater left ventricular diastolic dimensions (LVIDd) and aggression with elevated left atrial-to-aortic root ratios (LA/Ao). These findings parallel human data linking emotional stress to atrial remodeling and arrhythmia risk [11, 12]. In addition, both FS% and ejection fraction (EF%) showed negative correlations with systolic chamber size (LVIDs), consistent with expected physiological relationships, and heart rate correlated strongly with myocardial wall thickness, FS%, and EF%, indicating coordinated contractile changes under heightened arousal (Table 5, Fig. 2). By contrast, none of these associations were observed in the control group, where correlations remained weak and non-significant, reinforcing that the cardiac–behavioral relationships were specific to anxious dogs.
The association between general anxiety and elevated fractional shortening (FS%) observed in this study suggests a hyperdynamic myocardial state consistent with chronic sympathetic nervous system activation. Similar findings are well documented in human psychocardiology, where persistent emotional stress promotes catecholamine release, heightened myocardial contractility, and progressive structural adaptations. The increased FS% in anxious dogs therefore likely reflects autonomic imbalance–mediated cardiac hyperkinesis. The positive correlation between phonophobia and left ventricular diastolic dimension (LVIDd) may provide additional insight, as noise-sensitive individuals exhibited relatively larger diastolic chambers despite the group mean remaining below reference values—likely due to small body size. This pattern could represent early-stage remodeling associated with increased preload or stress-induced dilation. Likewise, the association between aggression and higher left atrial-to-aortic root ratios (LA/Ao) may indicate early atrial remodeling in response to repeated behavioral arousal, though causal inference cannot be drawn from this cross-sectional design. Finally, the strong correlations between heart rate and multiple echocardiographic indices (LVFWs, FS%, EF%) reinforce the biological plausibility of anxiety-driven cardiac remodeling, particularly since these relationships were absent in the control group.
Our findings partially align with existing veterinary literature but differ in emphasizing chronic, naturally occurring emotional stress rather than acute or pharmacological stressors. Most previous work in veterinary cardiology has examined transient stress responses or drug effects, whereas this study evaluated prolonged anxiety in client-owned dogs under real-world conditions. Pradelli et al. (2014) showed that acute emotional stress during echocardiographic handling transiently elevated aortic peak velocity in Boxer dogs, reflecting a hyperdynamic response, which parallels our observation of elevated fractional shortening (FS%; mean 42.14 ± 10.96%), exceeding reference limits in 44.4% of anxious dogs and suggesting sustained sympathetic activation [13]. In contrast, Fries et al. (2019) reported no significant changes in FS% or ejection fraction (EF%) after trazodone administration in healthy cats, highlighting the importance of excluding medicated dogs in our study and underscoring that the higher FS% in anxious dogs is more plausibly explained by chronic anxiety than pharmacological effects [14]. Neuroimaging work by Xu et al. (2023) further supports this interpretation by demonstrating disrupted connectivity in brain regions regulating emotion (e.g., amygdala, hippocampus) in anxious dogs, reinforcing the link between emotional dysregulation and physiological outcomes [15]. Similarly, Wang et al. (2025) identified early myocardial impairment under radiation-induced stress in Beagles using speckle-tracking echocardiography; unlike those dogs, our anxious cases displayed increased FS% and EF%, suggesting that emotional stress may initially induce compensatory cardiac responses before progressing to maladaptive remodeling depending on stressor chronicity [16]. Taken together, these comparisons highlight the novelty and clinical significance of our work, providing evidence that naturally occurring chronic emotional stress can drive subclinical cardiovascular remodeling in dogs, consistent with mechanisms established in human psychocardiology [17,18,19,20].
This study has important implications for veterinary cardiology and behavioral medicine. Traditionally, canine anxiety is addressed through behavioral modification, environmental enrichment, and pharmacological treatment, but our findings suggest that chronic anxiety may also act as a driver of early-stage cardiac remodeling, even in dogs without overt cardiovascular disease. Increases in fractional shortening (FS%), left ventricular internal dimension in diastole (LVIDd), and left atrial-to-aortic root ratio (LA/Ao) observed in anxious dogs indicate that sustained emotional stress can promote subclinical cardiovascular adaptations [21]. Although these parameters generally remained within reference ranges, they may represent compensatory states that precede pathological remodeling. Elevated FS%, present in 44.4% of anxious dogs, likely reflects sympathetic dominance which, in predisposed individuals or breeds, could progress to hypertrophy or diastolic dysfunction. By contrast, control dogs remained within normal limits, reinforcing that these changes were specific to anxiety rather than incidental variation.
Integrating standardized behavioral screening tools such as the validated C-BARQ into routine veterinary practice may therefore support early identification of dogs at risk, particularly those with high scores for general anxiety, phonophobia, or aggression. Such individuals could benefit from proactive cardiac monitoring, including serial echocardiography, to enable timely intervention. Importantly, clinicians should interpret echocardiographic findings in the context of behavioral status: functional hyperkinesis or mild atrial enlargement in reactive dogs should not be automatically attributed to primary cardiac pathology [3]. Recognizing the influence of emotional stress on cardiac function can improve diagnostic accuracy, guide treatment decisions, and help avoid unnecessary pharmacological interventions.Ultimately, these findings support a more integrated veterinary approach that combines behavioral and cardiac assessments. Such a holistic model could facilitate earlier, targeted interventions and improve both emotional and cardiovascular health in companion animals.
Despite providing novel insights, this study has several limitations. Although a control group of clinically healthy, non-anxious dogs was included, it was not fully matched to the anxious cases in age, body size, or breed distribution. These differences may partly explain some echocardiographic findings and limit attribution solely to anxiety. Breed predispositions—particularly in Shiba Inus and Maltese—may also have influenced baseline cardiac metrics. Reliance on owner-reported C-BARQ data, while validated, introduces subjectivity and potential reporting bias. The relatively small sample size (36 anxious, 20 controls) limits statistical power and generalizability, while the cross-sectional design prevents causal inference. In addition, multicollinearity among anxiety subtypes was not formally assessed and may have contributed to overlapping variance.Interpretation of echocardiographic parameters against fixed reference ranges is another constraint, as more than half of the anxious dogs weighed < 3 kg. Size-related misclassification is therefore possible, particularly for LVIDd, and future work should employ size-indexed indices (e.g., LVIDDN) to improve accuracy. An unexpected positive correlation between heart rate and LVIDd also emerged, which runs counter to physiological expectations. This paradox may reflect allometric scaling in very small dogs, confounding by anxiety severity, or measurement variability in fast heart rates, and should be regarded as exploratory pending replication in larger, size-matched case–control groups.Finally, the absence of electrocardiographic (ECG) data represents a significant limitation. Although rhythm abnormalities were suspected in nearly half of the anxious dogs, their classification remained provisional, based only on auscultation and echocardiographic impressions. Future studies should address these limitations by enrolling larger, multicenter case–control groups with matched controls, using longitudinal designs to explore temporal relationships, incorporating objective stress biomarkers (e.g., heart rate variability, serum cortisol, salivary alpha-amylase), and applying advanced imaging modalities such as tissue Doppler or speckle-tracking echocardiography. Interventional studies assessing whether behavioral therapy or anxiolytic medications can mitigate anxiety-related cardiac changes would also provide clinically relevant guidance for optimizing both emotional and cardiovascular health in dogs.
Conclusion
This study provides novel evidence that chronic behavioral anxiety in dogs is associated with measurable alterations in cardiac structure and function, including elevated fractional shortening, increased left ventricular dimensions, and higher LA/Ao ratios. These findings suggest that persistent emotional stress may promote early, subclinical cardiac remodeling even in the absence of overt heart disease. Recognizing anxiety as both a behavioral and cardiovascular condition underscores the importance of incorporating standardized behavioral assessments into routine veterinary care. Although limited by sample size and the absence of fully matched controls for breed and body size, the results highlight the need for larger, longitudinal studies incorporating objective stress biomarkers to clarify causality and disease progression. Such work could refine early diagnostic strategies and support preventive interventions. Ultimately, addressing anxiety as a systemic condition may improve both emotional well-being and long-term cardiovascular health in companion dogs.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author (Dr. Mohammad Molazem) on reasonable request.
References
Salonen M, Sulkama S, Mikkola S, Puurunen J, Hakanen E, Tiira K, et al. Prevalence, comorbidity, and breed differences in canine anxiety in 13,700 Finnish pet dogs. Sci Rep. 2020;10:2962.
Article PubMed PubMed Central CAS Google Scholar
Sacoor C, Marugg JD, Lima NR, Empadinhas N, Montezinho L. Gut-brain axis impact on canine anxiety disorders: new challenges for behavioral veterinary medicine. Vet Med Int. 2024;2024:2856759.
Article PubMed PubMed Central Google Scholar
Wormald D, Lawrence AJ, Carter G, Fisher AD. Reduced heart rate variability in pet dogs affected by anxiety-related behaviour problems. Physiol Behav. 2017;168:122–7.
Article PubMed CAS Google Scholar
Richter H, Kircher PR, Joerger FB, Bruellmann E, Dennler M. Assessment of myocardial perfusion at rest and during stress using dynamic first-pass contrast-enhanced magnetic resonance imaging in healthy dogs. Front Vet Sci. 2018;5:211.
Article PubMed PubMed Central Google Scholar
Koskela A, Törnqvist H, Somppi S, Tiira K, Kykyri V-L, Hänninen L, et al. Behavioral and emotional co-modulation during dog–owner interaction measured by heart rate variability and activity. Sci Rep. 2024;14:25201.
Article PubMed PubMed Central Google Scholar
Pourghasemi Z, Norouzi N, Safari N, Khakpour H, Keypoori D, Shams F, et al. Prevalence of congenital heart diseases in dogs in Tehran, Iran: a retrospective study from 2013 to 2023. Vet Med Int. 2025;2025:2994461.
Article PubMed PubMed Central Google Scholar
Lucina SB, Sarraff AP, Wolf M, Silva VB, Sousa MG, Froes TR. Congenital heart disease in dogs: a retrospective study of 95 cases. Top Companion Anim Med. 2021;43:100505.
Article PubMed Google Scholar
Gugjoo MB, Saxena AC, Hoque M, Zama MMS. M-mode echocardiographic study in dogs. Afr J Agric Res. 2014;9:387–96.
Article Google Scholar
Saini N, Uppal SK, Randhawa CS. Echocardiographic parameters and indices in healthy Labrador retriever dogs. Isr J Vet Med. 2017;72:28–32.
Google Scholar
DE MADRON ED, Chetboul V, Bussadori C. Clinical echocardiography of the dog and cat. Elsevier Health Sciences; 2015.
Google Scholar
Wang Z, Qin H, Chen G, Dai Y, Cai Y, Cheng X, et al. Anxiety is associated with increased risk for atrial cardiopathy. Acta Neurol Belg. 2020;120:1383–8.
Article PubMed Google Scholar
Wu H, Li C, Li B, Zheng T, Feng K, Wu Y. Psychological factors and risk of atrial fibrillation: a meta-analysis and systematic review. Int J Cardiol. 2022;362:85–92.
Article PubMed Google Scholar
Pradelli D, Quintavalla C, Crosta MC, Mazzoni L, Oliveira P, Scotti L, et al. The influence of emotional stress on Doppler-derived aortic peak velocity in boxer dogs. J Vet Intern Med. 2014;28:1724–30.
Article PubMed PubMed Central CAS Google Scholar
Fries RC, Kadotani S, Vitt JP, Schaeffer DJ. Effects of oral trazodone on echocardiographic and hemodynamic variables in healthy cats. J Feline Med Surg. 2019;21:1080–5.
Article PubMed Google Scholar
Xu Y, Christiaen E, De Witte S, Chen Q, Peremans K, Saunders JH, et al. Network analysis reveals abnormal functional brain circuitry in anxious dogs. PLoS ONE. 2023;18:e0282087.
Article PubMed PubMed Central CAS Google Scholar
Wang Z-Y, Huang L, Li L-Q, Zhang C-Q, Guo L-Y, Liu Y-N, et al. Quantitative evaluation of radiation-induced heart disease in beagle dogs by speckle tracking echocardiography. BMC Cardiovasc Disord. 2025;25:199.
Article PubMed PubMed Central Google Scholar
Park G, Thayer JF. From the heart to the mind: cardiac vagal tone modulates top-down and bottom-up visual perception and attention to emotional stimuli. Front Psychol. 2014;5:278.
Chen X, Xu L, Li Z. Autonomic neural circuit and intervention for comorbidity anxiety and cardiovascular disease. Front Physiol. 2022;13:852891.
Article PubMed PubMed Central Google Scholar
Rosenblum S, Rab SL, Admon R. Dynamics in physiological acute stress response trajectories: uncovering latent variability. BMC Psychiatry. 2025;25:361.
Article PubMed PubMed Central Google Scholar
Grol M, De Raedt R. The link between resting heart rate variability and affective flexibility. Cogn Affect Behav Neurosci. 2020;20:746–56.
Article PubMed Google Scholar
Avalos-Borges EE, Acevedo-Arcique CM, Segura-Correa JC, Jiménez-Coello M, Garg NJ, Ortega-Pacheco A. Echocardiographic documentation of dilated cardiomyopathy development in dogs naturally infected with trypanosoma cruzi. Animals. 2024;14:1884.
Article PubMed PubMed Central Google Scholar
https://bmcvetres.biomedcentral.com/articles/10.1186/s12917-025-05074-3
________________________________________________________________________________________________________________________
AVMA I News – Why persistence pays off when it comes to dietary management of chronic enteropathy patients
Prescribing therapeutic diets involves not only evaluating ingredients, but also strong client communication
Story by Malinda Larkin
October 20, 2025
If a patient is vomiting, has diarrhea, or is showing other gastrointestinal (GI) clinical signs, dietary management should automatically be on the table, says Dr. Adam Rudinsky, associate professor in the Department of Veterinary Clinical Sciences at The Ohio State University College of Veterinary Medicine.
He runs a laboratory dedicated to researching chronic enteropathies, pancreatic and hepatic disease, and intestinal microbiome. Dr. Rudinsky will be the first to acknowledge that getting owners to switch diets can be difficult.
“A diet switch is like a personal affront to people,” Dr. Rudinsky said. “A lot of their self worth, in terms of that animal, is tied up in the diet that they feed them. So, it can be a challenging discussion, particularly when you have a busy schedule.”
Taking a comprehensive diet history and making a purposeful diet selection are the keys to success when it comes to a nutritional management strategy, says Dr. Adam Rudinsky, associate professor in the Department of Veterinary Clinical Sciences at The Ohio State University College of Veterinary Medicine. He gave a lecture on the topic at the 2025 American Animal Hospital Association annual conference.
But he maintains it’s still worth the time and effort. He and Dr. Harry Cridge, associate professor of small animal internal medicine at Michigan State University College of Veterinary Medicine, each gave presentations on nutrition at the 2025 American Animal Hospital Association CON, held September 11-13 in Chicago. Both are board-certified specialists in small animal internal medicine.
Dietary selection
When considering a dietary management strategy, after taking a diet history, Dr. Rudinsky recommends asking these questions:
- What is the duration of clinical signs—acute or chronic?
- Where are the clinical signs localized? Stomach? Small or large bowel?
- Are the clinical signs consistent with a specific disease process, such as pancreatitis or parvovirus?
If the diagnosis warrants, a diet trial can be the first step, even before a biopsy or other invasive diagnostic procedure. Why? “Because many patients will respond to it and it’s cost effective,” Dr. Cridge said.
Dr. Rudinsky outlined five main diet categories for dogs and cats with acute or chronic GI clinical signs:
- Highly digestible or low-residue diets: These diets take the work off the GI tract by requiring less digestive effort. Dr. Rudinsky uses these diets for dogs and cats that are underweight due to vomiting, mild small intestine disease, or acute GI disease because of their nutritional density.
- Novel or limited-ingredient diets: Often associated with use in chronic GI disorders, especially small or large bowel disease, these diets can also help with food allergies. However, Dr. Rudinsky says the prevalence of food intolerance appears to be much higher than the true food allergy, as most animals are responding to the antigen load. “If you continue to feed a diet that is too rich or varied, you can get those responses frequently that appear as a chronic condition,” he said. When selecting a limited-ingredient diet, he advises choosing one with only a single carbohydrate and a single protein source in the ingredient list, both of which are ideally novel to the patient.
- Hydrolyzed diets: These diets undergo hydrolysis processing to reduce allergenicity and antigenicity by altering macronutrient structure. Typically, they are highly digestible and have a reduced fiber content. Often these are a mainstay for chronic GI symptoms, such as pancreatitis in cats and small bowel disease in dogs and cats. Specifically, Dr. Rudinsky said, “The more I have a vomiter … or a small bowel diarrhea case, I love a hydrolyzed diet.”
- Low-fat diets: These are diets with fat content generally in the range of less than 3 grams of fat per 100 kilocalories. More recently, fat content has been identified as an important component in the management of some canine GI diseases, especially pancreatitis, GI motility issues, or mixed bowel diarrhea cases, Dr. Rudinsky said. This doesn’t appear to be an issue for cats. However, “for dogs, if you use an easily digestible diet, you might as well use one that is low fat too,” he added.
- Fiber-enriched diets: Fiber is added to diets for multiple reasons, but especially diarrhea and large bowel disease in both dogs and cats. The fiber type and source will influence the type of effect that is seen in the patient. The benefits of soluble and insoluble fiber, besides helping with diarrhea (soluble) or constipation (insoluble) include fermentation, production of volatile fatty acids, benefits to enterocyte health, augmentation of the microbiota, as well as alterations in gut motility and passage of GI luminal contents.
When first getting started, Dr. Rudinsky recommends looking at hydrolyzed, novel protein, or easily digestible diets for cats. “Cats maintain the same response rate with easy digestible diets that they do with hydrolyzed and novel ingredient diets, but dogs do not,” he said, so only start with hydrolyzed or novel ingredient diets for dogs.
Dr. Harry Cridge, associate professor of small animal internal medicine at Michigan State University College of Veterinary Medicine, says he uses soluble fiber to control diarrhea and insoluble fiber to control constipation.
Also important to note is that even among a particular diet category, options can vary by ingredient list, fiber content, fat content, and more. For example, Dr. Cridge says elimination diets differ in more than just their protein. He recommends using product guides for up-to-date nutrient compositions, including looking at dietary fat and fiber content.
Both Drs. Rudinsky and Cridge suggest looking at the global nutrient profile of diets when evaluating macronutrient diet choices. Do not focus on percent as fed, which doesn’t take into account water content and other features of the diet, but rather look at grams per 100 kcal, Dr. Rudinsky said.
This information may not always be on the bag. In that case, they recommend using Tuft University Cummings School of Veterinary Medicine’s Clinical Nutrition Service Nutrient Calculator or product guides to help with comparing nutrients between diets.
Dedication pays
Recent research reaffirms the importance of purposeful nutritional management, especially for patients with chronic enteropathies.
A retrospective paper published this past May in the Journal of Veterinary Internal Medicine (JVIM) looked at 81 dogs with chronic enteropathy. Among those in which the only therapeutic change was a hydrolyzed or other alternative diet, 88% (14 of 16) had a decreased stool‐consistency score and 70% (16 of 23) saw a 69% reduction in their total Canine Inflammatory Bowel Disease Activity Index score.
Another study, published in the September-October 2024 issue of JVIM, followed 60 dogs with chronic inflammatory enteropathy for two years. The initial dietary response rate was about 45%, but over time, 73% of them ended up being diet responsive and able to come off modulators.
“Giving more evidence that even if you don’t get that initial dietary response, diet is still a fundamental component, because your ability to minimize medications later on is going to be super useful,” Dr. Rudinsky said.
Finally, he emphasized the importance of taking a complete diet history. A study out of the University of Zurich, published in 2022 in the journal Animals, found that dietary information gained from referring veterinarians and owners was often incomplete.
Among these dogs presenting to the university’s GI and internal medicine service, no dog had received more than one previous diet trial for chronic GI signs.
After prescribing all of the dogs a new diet, 58% had an initial dietary response. Then the researchers convinced all the owners with a dog that didn’t respond to switch to yet another diet, and those dogs saw a 66% dietary response rate. The researchers did this again and had a 44% dietary response rate for owners that had switched a third time.
“If you add those percentages up … they were able to drive their response rate up in chronic enteropathy animals up to 85% or 86%. So that is really powerful, especially if you think of those dogs that we’ve managed sometimes with super bad prednisone effects or other issues with chronic medications (to correct that).”
“A lot of us intuitively know this, but we’re so thankful if an owner even does one diet switch,” Dr. Rudinsky said. “Don’t be afraid to ask them to switch again if first switch isn’t working.”
So, when to switch? Dr. Rudinsky says, “If you have not seen a response in two weeks, you can abandon ship and switch. If you see a partial response at that two-week mark, then you want to go out to about four weeks.”
Dr. Cridge says two weeks for him, too, and then he’ll move onto another diet for canine patients. “I don’t push owners to try a diet for too long because that’s when they want to give up and try something else,” he said. For cats, he acknowledges the challenge is getting them to try the diet in the first place.
Another issue Dr. Rudinsky has noticed, beyond not convincing an owner to do a diet trial, is getting them to accept the cost of the new diet because many of them may be more expensive than what they are currently feeding their pet.
Finally, Dr. Cridge recommended referring patients to a board-certified veterinary nutritionist when there is a refractory case of chronic enteropathy or protein-losing enteropathy (PLE), presence of concurrent diseases, picky dogs that may need a home-cooked diet, or when owners wish to make a home-cooked diet.
“Nutritionists are invaluable resources, especially for refractory chronic enteropathy and PLE dogs and often will provide remote consultations directly,” he said.
_________________________________________________________________________________________________________________________________________________
AVMA I News – Benchmarking data plus elevating efficiency equals practice productivity
Benchmarking data plus elevating efficiency equals practice productivity
AVMA Economist Frederic B. Ouedraogo provides practice data to help with benchmarking
By Malinda Larkin
October 15, 2025
U.S. veterinary practices averaged about $1.5 million in gross revenue in 2024, but adjustment for inflation shows only modest growth in this metric since 2016.
Another sobering trend: The average number of active clients per practice has been declining since 2019 by about 95 clients per year, to a total of 3,351 clients in 2024. Over the same period, the average number active clients per full-time equivalent (FTE) veterinarian has declined by 15 clients per year, to a total of 1,499 in 2024.
In 2023, there were 34,296 veterinary practices, according to the U.S. Census Bureau. On average, approximately 362 new veterinary practices have been added each year since 2010, for an annual growth rate of 1.3%.
In 2023, there were 34,296 veterinary practices, according to the U.S. Census Bureau. On average, approximately 362 new veterinary practices have been added each year since 2010, for an annual growth rate of 1.3%.
To help create stronger and profitable practices, Frederic B. Ouedraogo, PhD, senior economist and associate director in the AVMA Veterinary Economics Division, encouraged practice owners to create their own data assets to evaluate the health of their own practice and engage in benchmarking analyses.
“This allows us to gain insights into our practice performance, monitor our performance over time and against our peers, enabling us to identify our strengths and weaknesses. More importantly, it allows us to learn things that others are doing,” he said.
Dr. Ouedraogo presented data from the 2025 AVMA Veterinary Practice Owners Survey during his talk “Unlocking Practice Health: Examining the Latest Practice Health Data” at the 2025 AVMA Veterinary Business and Economic Forum, held October 8-9 in Denver.
One of the key metrics for assessing operational efficiency in veterinary practice is gross revenue per square foot, which in 2024 was $538 per square foot on average.
“This is how much revenue each practice generates per square foot and is a good indicator of practice health,” he said, adding that this figure is especially helpful for comparison because it can be done with any other kind of practice, regardless of size. “Removing bottlenecks will result in greater efficiency and increase gross revenue per square foot.”
Rate of technology utilization in veterinary practice
Optimizing practice resources, including space and workforce, and removing inefficiencies in systems and processes, such as adopting new technologies, can help veterinary practices boost productivity and revenue, says Frederic B. Ouedraogo, PhD, senior economist and associate director in the AVMA Veterinary Economics Division.
Other productivity metrics from the survey are as follows:
Gross revenue per exam room: This is total revenue divided by the number of exam rooms in the practice. In 2024, U.S. veterinary practices generated $444,668, on average, per exam room per year. Inflation-adjusted (real) revenue per exam room has been trending down since 2020.
Gross revenue per FTE veterinarian: In 2024, the average veterinary practice generated $554,982 per veterinarian, down in inflation-adjusted terms from nearly $600,000 in 2019. Revenue per veterinarian per hour averaged $288 in 2024. “According to our data, veterinarians spend about 30 minutes per patient and complete about 15 scheduled appointments per day, on average, under normal circumstances,” Dr. Ouedraogo said.
Patients per veterinarian per day: The average veterinary practice saw 15 patients per day in 2024, compared with 16.6 in 2021, which he noted may have been unusually high during the pandemic. “If demand for veterinary services slows down, no matter how efficient you are, you will see fewer patients. So it can be a market-related issue, but let’s make sure that our systems and processes are as efficient as they can be, and again, ways to increase efficiency include but are not limited to optimizing your space, empowering your staff through training, and adopting cutting-edge technology that could boost productivity,” he said.
Veterinary practice space utilization: The average veterinary practice measured 3,845 square feet and had 3.5 exam rooms in 2024. The number of FTE veterinarians per exam room was 0.92. “It should be 0.5 or so, allowing one veterinarian to go from one room to another without waiting for the exam room to be ready for next patient,” Dr. Ouedraogo said.
Veterinary practice staffing numbers: The average veterinary practice reported 2.76 FTE veterinarians, which has varied between 2.5 and 2.8 since 2019. In addition, practices averaged 3.7 nonmedical staff members plus 5.74 veterinary technicians and veterinary assistants. But these numbers depend on the species, he noted. Companion animal predominant and exclusive practices averaged a little more than 2.6 FTE veterinarians, 6.35 FTE veterinary technicians and veterinary assistants, and around 4.1 nonmedical staff. Food animal and equine practices had between 3.34 and 2.56 FTE veterinarians, 1.55 and 2.6 veterinary technicians and veterinary assistants, and 1.25 and 1.45 nonmedical staff. Overall, the medical staff-to-veterinarian ratio was 2.21:1 in 2024.
Dr. Ouedraogo encouraged attendees to use the statistics derived from that survey as benchmarks to identify areas for improvement and drive better performance.
He added, “Keep in mind that your PIMS system may have capabilities to generate [these metrics] for you. Take the time to understand your management software, understand what it can do.”
Results Frederic B. Ouedraogo, PhD, presented at the forum will be published in the 2026 AVMA Report on the Economic State of the Veterinary Profession. The 2025 AVMA Report on the Economic State of the Veterinary Profession is available for AVMA members to download for free, and for a fee of $375 for non-members. This annual report examines the major trends through the lens of veterinary education, veterinary employment, and veterinary services.
For more practical advice from the “Unlocking Practice Health: Examining the Latest Practice Health Data” session at the 2025 AVMA Veterinary Business and Economic Forum, take a look at the 3-2-1 Insight-to-Action Guide created on the topic.
___________________________________________________________________________________________________________________________________________________________
AVMA – October 10, 2025
Integrative veterinary medicine: an emerging trend in veterinary practice
Mushtaq A. Memon, BVSc, PhD, DACT
Huisheng Xie, DVM, PhD
Abstract
The American Veterinary Medical Association’s (AVMA) House of Delegates recently adopted a revised policy on Complementary, Alternative, and Integrative Veterinary Medicine (IVM), reflecting a growing clinical and academic interest in complementary therapies. IVM is defined as “a comprehensive approach to animal health care that combines conventional veterinary practices with complementary and alternative therapies, such as acupuncture and chiropractic care.” While the utilization of IVM is increasing, a critical knowledge gap remains regarding its evidence-based integration into mainstream veterinary practice. This article provides an overview of the purpose, scope, and potential clinical contribution of commonly used modalities, including acupuncture, physical rehabilitation, manual and massage therapies, herbal medicine, and integrative nutrition. Each modality is briefly examined in terms of proposed mechanisms, therapeutic indications, and current research support. The goal is to clarify the role of IVM within veterinary clinical decision-making and promote a balanced, science-informed framework for its application. Acupuncture, a core IVM modality, uses fine needles to stimulate the body’s natural healing mechanisms. Physical rehabilitation targets neurological and musculoskeletal conditions, providing multidimensional care. Manual therapies apply hands-on techniques to soft tissues, joints, and nerves to promote relaxation, pain relief, and mobility. Integrative nutrition emphasizes personalized dietary plans that support overall health. Botanical medicine leverages the therapeutic properties of plants and their derivatives as part of a holistic treatment strategy. The takeaway emphasizes the importance of interdisciplinary collaboration, ensuring safety, and ethical practice in offering integrative veterinary care.
The AVMA House of Delegates recently passed a resolution adopting a revised policy on complementary, alternative, and integrative veterinary medicine (IVM). Integrative veterinary medicine is defined as “a comprehensive approach to animal healthcare that combines conventional veterinary practices with complementary and alternative therapies, such as acupuncture (ACP) and chiropractic care.”1
Integrative veterinary medicine reflects the growing interest in complementary therapies in both human and veterinary medicine. An analysis of national survey data reported that the proportion of US adults who have at least 1 complementary health approach (eg, ACP, chiropractic, massage, yoga, meditation) increased from 19.2% in 2002 to 36.7% in 2022.2 Similarly, a bibliometric study of veterinary literature documented a marked rise over the past 2 decades in publications referencing nonconventional therapies, particularly traditional Chinese veterinary medicine (TCVM), herbal medicine, plant products, essential oils, and other natural products.3
Integrative veterinary medicine encompasses a wide range of modalities, including ACP, physical rehabilitation, manual and massage therapies, herbal medicine, and integrative nutrition. The overarching goal is to combine the strengths of conventional and complementary approaches to enhance the quality and effectiveness of veterinary care.
This integrated framework reflects a broader societal shift toward complementary and integrative health practices, observed in both human and veterinary medicine. In 2007, approximately 38% of US adults reported using complementary therapies.4 Many of these adults may also seek complementary therapies for their pets. Even if such treatments are not a core part of a veterinarian’s practice, it is still important to have a basic understanding of the options, their uses, and when they can be effective.5 The American Animal Hospital Association’s member survey conducted from 1996 through 1999 documented an increase in the proportion of practices reporting the use of alternative therapies, rising from 6% in 1996 to 22% in 1998 and reaching 31% in 1999, indicating a marked upward trend in the provision of complementary and alternative veterinary medicine or IVM services.6
The term IVM has increasingly replaced earlier labels, such as “alternative” or “holistic” medicine, signaling a more collaborative and scientifically grounded approach. This shift is mirrored in institutional developments, such as the renaming of the NIH National Center for Complementary and Alternative Medicine to the National Center for Complementary and Integrative Health, a change that underscores a commitment to integrating complementary practices into mainstream healthcare.7
Integrative veterinary medicine encompasses a wide range of modalities, including ACP, physical rehabilitation, manual and massage therapies, herbal medicine, and integrative nutrition. As a reflection of a growing paradigm shift, IVM offers veterinarians a multifaceted, evidence-informed approach to treatment that merges traditional Western practices with complementary therapies. This evolution in veterinary medicine aligns with contemporary healthcare values and anticipates a future in which integrated care becomes the standard rather than the exception.
Acupuncture
A recent study8 evaluated the efficacy of acupuncture and electroacupuncture, integrated within a physiotherapy protocol, for postoperative rehabilitation in dogs with acute intervertebral disc extrusion. The multimodal approach was designed to enhance the recovery of ambulation. Analysis of 41 canine patients demonstrated that incorporating acupuncture into the rehabilitation protocol significantly increased the likelihood of regaining ambulatory function. Another study9 compared the therapeutic outcomes of decompressive surgery (DSX), electroacupuncture (EAP), and a combined approach (DSX + EAP) in dogs with thoracolumbar intervertebral disc disease (IVDD) and severe neurological deficits persisting for more than 48 hours. Findings indicated that EAP alone was more effective than DSX in promoting ambulation and improving neurological function in dogs with prolonged, severe deficits secondary to thoracolumbar IVDD.
According to this system, Qi circulates through defined pathways called meridians, and disease is understood as a disruption or blockage in this flow. Diagnostic evaluation within TCVM relies heavily on observational methods, such as tongue appearance, pulse quality, and other subtle physical cues.10,11
Acupuncture is one of the most commonly utilized modalities within IVM and is supported by the most extensive body of efficacy literature among complementary therapies. The term ACP originates from 2 Latin words: acus, meaning needle, and pungere, meaning to pierce or prick. Acupuncture involves the insertion of sterile, fine needles to stimulate the body’s natural healing mechanisms, aiming to diagnose, treat, and prevent disease. The core objective of ACP is to achieve therapeutic benefits with minimal trauma.12
Acupuncture is thought to relieve pain and reduce inflammation through multiple biological pathways. Needle stimulation triggers the release of natural pain-relieving chemicals, such as endorphins and enkephalins, from immune cells, which act on nerve endings and the sympathetic nervous system to block pain signals. Electro-ACP can also activate the hypothalamic-pituitary-adrenal axis and influence the autonomic nervous system, leading to broader anti-inflammatory effects. Research further suggests it may mobilize mesenchymal stem cells via hypothalamic activation, supporting tissue repair and regeneration.13
The use of fine, sterile needles is referred to as dry-needle ACP. Other common techniques include aqua-ACP, which involves injecting small amounts of fluid (eg, vitamin B12 or saline) into ACP points for prolonged stimulation—particularly useful in patients who cannot tolerate retained needles—and electro-ACP, which involves the application of electrical stimulation to inserted needles. Electro-ACP is employed to enhance neuromodulation and stimulate the release of neurochemicals by varying electrical frequencies.14
Acupuncture is commonly utilized for pain management. A recent clinical trial tested ACP in 32 dogs with osteoarthritis. Dogs received either ACP or placebo treatment once a week for 4 weeks. Measures of mobility and activity did not show a clear difference between the 2 groups. However, dog owners reported noticeable improvement in their pets’ comfort and ability to do daily activities after ACP.15 While ACP is practiced globally—in China, Korea, Japan, Europe, and the Americas—its application varies, much like the regional differences observed in Western medical practice, despite shared foundational principles.
Physical Rehabilitation
Animal physical rehabilitation stands out as a rapidly expanding domain within veterinary medicine. It is gaining prominence in treating small animals and equine and is increasingly recognized as a crucial facet of veterinary care, especially for animals with physical limitations stemming from surgeries or illnesses.16 This trend is accentuated by advancements in the life expectancy of companion animals and the rise in chronic conditions often linked to movement disorders.17 The American Association of Rehabilitation Veterinarians defines animal physical rehabilitation as the “diagnosis and management of patients with painful or functionally limiting conditions, particularly those with injury or illness related to the neurological and musculoskeletal systems.”18 Similar observations were made in a retrospective analysis of 5,195 integrative patient treatment sessions in a veterinary academic teaching hospital, with 39% involving a combination of therapeutic modalities. Among 274 patients receiving multiple modalities, neurologic and orthopedic diseases were the prominent concerns: 50.7% and 49.6%, respectively. Older neutered or spayed dogs, particularly Dachshunds, were more frequently treated, with common modalities including ACP, laser therapy, electro-ACP, and hydrotherapy (> 50% of patients).19
The overarching objective of rehabilitation is to alleviate pain, reduce edema, facilitate tissue healing, restore gait and mobility, rebuild strength, prevent further injury, and enhance overall quality of life.20 A foundational understanding of the anatomy of animals, such as canines, felines, and equines, is imperative for formulating safe and effective rehabilitation plans. The ideal rehabilitation strategy involves a comprehensive patient assessment to identify musculoskeletal or neurological impairments, discern TCVM patterns, address pain, and employ a combination of rehabilitation therapies (ACP, manual therapy [MT], and therapeutic exercises), along with herbal and pharmaceutical supplements. Additionally, commonly utilized therapeutic modalities encompass cryotherapy, thermotherapy, photobiomodulation, and electrical stimulation.
Therapeutic exercises, whether on land or in water, serve as the linchpin of veterinary rehabilitation, irrespective of the specific problem or diagnosis. Regular reassessment intervals are vital for monitoring the patient’s progress. For a more in-depth exploration of various facets of veterinary rehabilitation, readers are directed to the comprehensive information provided by Koh and Huntingford.21
Manual Therapies
Manual therapy is a comprehensive term encompassing veterinary manipulative therapy, massage, touch, osteopathy, and related techniques.22 Manual therapy involves a skilled, specific hands-on approach utilized by clinicians and therapists to address soft tissues, joints, and nerves of diverse etiologies. The primary objectives of MT are to induce relaxation, enhance circulation, alleviate muscle tension, diminish pain, increase the flexibility of soft tissues, and restore joint mobility.23 The precise therapeutic mechanism behind MT is not fully elucidated, likely involving a combination of physiological, biomechanical, physical, and psychological effects.24
Physiologically, MT may reduce pain through the pain gate theory and stimulate descending inhibitory tracts, leading to pain inhibition and tissue relaxation.25 Biomechanically, MT can alter tissue extensibility and fluid dynamics by mechanically pressuring and mobilizing soft tissues and joint capsules, thereby facilitating tissue repair and remodeling and improving tissue function.26
While MT plays a vital role in the rehabilitation of many patients, there is a scarcity of studies validating its clinical benefits in animals. Two studies27,28 suggest that MT, in combination with ACP, may be effective and safe for improving pain relief, physical function, and quality of life in dogs with degenerative diseases, chronic pain, or musculoskeletal pain.
A survey of equine practitioners revealed a generally positive view of MT therapies, with over half referring owners to veterinary chiropractic (manipulative therapy) or massage.29 The reason for equine practitioners’ patient referrals is not entirely clear but may be linked to an understanding of the importance of soft tissue pathology or due to the restrictions on certain drug therapies in performance horses.30 Studies in horses suggest increased tolerance to pressure and improved vertebral flexibility following spinal manipulative therapy.31 Massage of equine hind limb muscles has been shown to increase active and passive protraction.32
Massage therapy
Massage therapy, defined by Hippocrates as “the art of rubbing,” boasts an extensive history dating back to 2,500 BC, making it one of the earliest documented treatment modalities.33 Initially described in China, ancient Greeks and Indians also practiced massage, but it was the Roman Empire that notably embraced massage therapy for sports and war injuries.34,35 For detailed massage techniques and descriptions, readers are referred to Maler.33
Tui-na
Tui-na (pronounced twee-nah) is a Chinese MT used for preventing and treating disease. Tui-na practitioners use various manipulations applied to ACP points (acupoints) and Meridians as well as special limb-stretching movements to prevent and treat diseases.36 Traditional Chinese veterinary medicine practitioners, from the novice to the senior clinician, can use this unique modality to help more animals.
Tui-na is generally very safe to practice on animals, and they also tend to enjoy the administration of Tui-na techniques. It is especially well suited for fractious or anxious patients or those with an aversion to needles. Tui-na can be effectively used alone for soft tissue injury, pain management, and anxiety. It can also enhance the effects of ACP and herbal medicine for a variety of chronic illnesses.37 Many Tui-na techniques can be applied as a tool for TCVM pattern diagnosis, which is central to TCVM practice. In addition, it is a tool that can be taught to caregivers to enhance the human-animal bond.37
Integrative Nutrition
The timeless maxim “Let food be thy medicine and medicine be thy food” often underscores the pivotal role of nutrition in maintaining optimal health. In the realm of veterinary medicine, contemporary clinical assessment guidelines underscore the significance of nutritional evaluation, with conventional veterinary nutrition forming a cornerstone of the veterinary curriculum.38
Embracing an integrative approach to veterinary nutrition empowers practitioners to craft tailored, all-encompassing nutrition plans for each patient. This involves drawing from both established and emerging insights into nutrition and supplements, strategically employed for both nutritional support and therapeutic purposes.39
Nutritional assessment and intervention are seamlessly integrated with other IVM modalities, such as physical rehabilitation and sports medicine. Dietary modification can improve performance, preserve lean body mass, reduce inflammation, and improve recovery from injury.40 Notably, the pet owners seeking physical rehabilitation for their pets commonly grapple with the issue of being overweight or obese. Therefore, a comprehensive exploration of the physiological implications and contemporary guidelines for managing obesity is pertinent.41–43 The role of diet in various diseases, especially in cancer, has been reported.44
Beyond the realm of nutrition’s direct impact on the musculoskeletal system, its interplay with other bodily systems is worth examining. For instance, TCVM emphasizes the incorporation of food therapy as an energetic approach to diets applied by IVM practitioners in their dietary recommendations. Explaining all the details of TCVM’s food therapy is beyond the scope of this article; readers are encouraged to explore the comprehensive insights provided by Fowler and Xie.45
Botanical and Herbal Medicine
Herbal medicine, also known as botanical medicine, involves the utilization of plants or plant derivatives with medicinal or therapeutic effects on the body. This encompasses the use of entire plants or specific parts, such as flowers, berries, leaves, stems, and roots, which may contain various active constituents producing therapeutic effects.46 The application of herbal supplements is on the rise among veterinarians and pet owners, with a notable prevalence in veterinary oncology.47,48 Traditional medical systems, including Chinese, Western, Ayurvedic, and indigenous North American practices, incorporate natural products into treatments. Plant-derived products offer potential avenues for drug discovery, exemplified by the reported effects of medical mushrooms on cancer.49 A Chinese herbal researcher was awarded a Nobel Prize in medicine for the discovery of Artemisia derivatives, now utilized in treating resistant malarial infections in humans.50,51 Silymarin, a derivative of milk thistle, is commonly prescribed by veterinarians for hepatic diseases.50,51 Numerous publications detail hundreds of herbs and herbal combinations for animal use, drawing from case reports, clinical experience, expert opinions, or data from other species.52
Conclusions
In conclusion, IVM represents a progressive approach that blends conventional Western veterinary practices with complementary therapies, driven by the growing interest of humans and animal owners. The term “integrative” reflects a preference over “alternative” or “holistic,” aligning with the broader shift observed in medical practices.
The diverse spectrum of IVM includes ACP, physical rehabilitation, manual therapies, integrative nutrition, and herbal medicine. Acupuncture, rooted in ancient Chinese practices, employs fine needles to stimulate the body’s healing abilities. Physical rehabilitation is crucial for animals with mobility issues, emphasizing a comprehensive approach to veterinary care. Manual therapies involve hands-on techniques to address soft tissues and joints, contributing to pain reduction and improved mobility. Integrative nutrition recognizes the role of tailored diets in promoting overall health. Herbal medicine offers therapeutic benefits in veterinary treatments.
In essence, IVM strives to provide comprehensive, evidence-based care that integrates the best of conventional and complementary approaches for the well-being of animal patients. The evolving landscape of IVM necessitates ongoing education to ensure that veterinarians are adept at navigating this integrative paradigm.
____________________________________________________________________________________________________________________________________________________________
TVP – Today’s Veterinary Practice Logo Articles | October 6, 2025 | Issue: November/December 2025
Anesthetic Strategies for the Dental Patient
General practice encompasses both anesthesia and dentistry skill sets on a continuum from basic to advanced care, and it is up to the discretion of the practitioner to determine when a case should be referred.
Brook A. Niemiec, DVM, DAVDC, DEVDC, FAVD
Stephanie Hon, DVM, DACVAA
Abstract
Dental care for small animal veterinary patients requires high-quality anesthesia and pain management. Dental patients often have significant comorbidities along with their need for dental care, continuing into the geriatric period. With appropriate preanesthetic preparation and perianesthetic management, these patients can still receive quality dental care to support them throughout their life. Robust pain management and techniques that decrease inhalant anesthetic minimum alveolar concentrations can help manage common comorbidities such as mitral valve disease and chronic kidney disease. However, complex patient status or advanced dental disease may present a challenge to the provider. For such patients, anesthesia-free dentistry is not recommended; instead, referral to a specialist team will reduce surgery and anesthesia time and enable use of advanced monitoring techniques.
Take-Home Points
Pain control intervenes in the pain pathway at multiple points, and a multimodal, preemptive approach supports a continuum of analgesia throughout the patient experience.
Locoregional anesthesia offers numerous benefits for pain management and techniques for reducing intraoperative inhalant anesthetic minimum alveolar concentrations.
Periodontal disease can increase the risk for development of new renal or cardiac comorbidities and can exacerbate preexisting disease; although dental work should be encouraged for patients with these conditions, patients must be properly prepared and their conditions managed.
Anesthesia-free dentistry is not a viable treatment option.
A specialist dentistry and anesthesia team can help improve patient outcomes, especially for patients with complex status or advanced dental disease.
Pain interventions should be aligned with the degree of pain stimulus experienced by the animal.1 Ideal pain management would consist of a preventive, multimodal approach, intervening in the pain pathway at multiple points and attempting to avoid any perception of pain by the patient.1-3 To achieve this goal, a combination of oral, intravenous, locoregional, and nonpharmaceutical agents can be used to create a full complement of strategies to mitigate pain.
What are the commonly used analgesics in dental patients?
Analgesia has been proven to be more effective when administered preemptively (before the procedure) than postoperatively.4-6 Oral preemptive analgesia and sedation are often achieved with premedication designed to be administered by the client before bringing the patient to the hospital.7 Agents generally consist of gabapentin and trazodone, which have wide safety ranges. Pregabalin has also been investigated for preemptive use.8 Although generally aimed at reducing anxiety and allowing for ease of obtaining intravenous access, gabapentinoids can also be administered before the procedure. Although NSAIDs are also powerful analgesics, they are reserved for postprocedure administration due to their potential to lead to renal insult if hypotension has been difficult to mitigate under general anesthesia. Acetaminophen (dogs only) is also being increasingly added to the armamentarium of perioperative oral pain control options.1,9,10
NSAIDs provide excellent pain control for oral surgery, typically for 24 hours, and can be given intraoperatively or postoperatively (especially advantageous in the initial postoperative period when a patient may resent oral manipulation). NSAIDs should be used with aligned standards in feline patients (robenacoxib or low-dose meloxicam only) and in all patients with metabolic disease, especially renal impairment.11 Preoperative testing (including CBC, serum chemistry, and urinalysis) and close intraoperative blood pressure monitoring are critical for minimizing any anesthetic complications.
Opioids are the mainstay analgesic for acute pain, and their safety index is favorable.12 Full µ agonists (e.g., methadone, fentanyl, hydromorphone, morphine) are commonly used during surgery. For postoperative pain control, buprenorphine provides a step-down partial µ agonist effect, but it can be a useful option to send home with patients or administer in a sustained-release formulation. Butorphanol is generally not adequate for pain control because its effect size is limited.13
Additional constant-rate infusions (CRIs) have been investigated to complement opioids. Dexmedetomidine (an α2 agonist) has been shown to be as effective as morphine for postoperative pain control in dogs undergoing invasive surgery.3 Ketamine, an NMDA (N-methyl-D-aspartate) receptor antagonist, has been shown to effectively reduce scores indicating acute pain but also to prevent conversion to or treatment of chronic pain for oncologic palliative care.3
Which locoregional techniques are commonly used in dental practice?
Locoregional techniques are excellent for blocking transduction and transmission of the pain stimulus, beginning before any surgical stimulus is created until long into the postoperative period if long-term techniques are used.14 Locoregional anesthesia reduces overall systemic drug consumption, recovery times, and inhalation anesthetic minimum alveolar concentrations (MACs).15
Commonly used locoregional techniques include infraorbital, mental, and caudal mandibular blocks, which are best learned in a hands-on laboratory and are taught in most extraction courses. For a list from the lead author, including a virtual option, see vdspets.com.
Infraorbital block (FIGURES 1 AND 2): If properly performed, the infraorbital block affects the ipsilateral maxilla teeth, as well as associated soft tissues.16,17 The infraorbital canal runs rostrally just above the apices of the maxillary fourth premolar and exits the maxilla over the distal root of the third premolar. To approximate the dorsoventral location, imagine the fourth premolar being approximately the same size mesiodistally as corona-apically. Measure the width of the tooth and then that same distance dorsally from the cusp tip. The infraorbital canal is just apical to this point. Starting approximately over the second premolar parallel to the maxilla, advance the needle caudally, close to the maxillary bone. The depth is controversial; some veterinary dentists barely enter the needle into the canal, and others place the needle all the way caudally to the level of the second molar. These authors generally recommend insertion to the level of the medial canthus of the eye in mesocephalic and dolichocephalic dogs.16 Correct placement can be confirmed by gentle lateral movement of the needle, allowing it to engage the canal wall.
In brachycephalic dogs and in cats, the infraorbital canal is very short, which may easily allow for orbital penetration.16 The block will diffuse distally to the molars if a finger is placed over the foramen for 30 to 60 seconds after injection.17
Mental block (FIGURES 3 AND 4): The mental block affects the inferior alveolar nerve and anesthetizes the ipsilateral canine tooth to the central incisor, including the surrounding bone and associated soft tissue.16,17 For the block to be effective, the needle must be placed slightly within the canal.
In dogs, the middle mental foramen is located rostrocaudally apical to the mesial root of the second premolar; in cats, it is midway in the diastema between the canine tooth and the third premolar.16,18 Dorsoventrally, the middle mental foramen is approximately two-thirds of the distance down from the dorsal border of the mandible.
To perform a mental block, retract the mandibular labial frenulum ventrally and insert the needle at the rostral aspect of the frenulum and advance it at an approximately 45° angle along the mandibular bone until it just enters the canal. Confirm placement by moving the syringe laterally to encounter the lateral aspect of the canal.
Caudal mandibular block (FIGURES 5 AND 6): The inferior alveolar nerve enters the mandibular foramen on the lingual aspect of the caudal mandible.16,17 The caudal mandibular block is performed by infiltrating the nerve at this level before its entry into the canal.
To perform the intraoral approach, place the patient in dorsal recumbency with the mouth open. Approximate the width of the last molar tooth (M3 in dogs and M1 in cats), and insert the needle starting at 1 width of the tooth caudally from the caudal edge of the last molar. The needle is inserted into the mucosa on the lingual aspect of the mandible and advanced right along (ideally touching) the mandible. Insert the needle at a 45° angle caudally, advancing it along the bone approximately halfway ventrally from the dorsal aspect of the mandible. If this insertion is correctly performed, all mandibular teeth, bone, and soft tissue on the treated side are affected by this block.18
Utilize your dentistry veterinary nurses/techs for locoregional blocks and share this TVN article!
Is there a use for anesthesia-free dentistry, especially given the prevalence of patients with comorbidities such as advanced cardiac disease?
In the opinion of the authors (as well as numerous veterinary dentists and veterinary dental associations), the short answer is no; anesthesia-free dentistry (also called nonanesthetic dentistry) serves no medical purpose whatsoever. It is well established that it is not possible to fully diagnose oral and dental pathology without a thorough anesthetized oral examination (including periodontal probing and imaging) of an anesthetized patient.19-25 Furthermore, a professional dental cleaning includes supragingival and subgingival scaling as well as polishing of the teeth with power and/or hand instrumentation performed by a trained veterinary healthcare provider while the patient is under general anesthesia.23,26
One study showed that anesthesia-free dentistry may result in periodontal health that is worse than it was before the procedure.27 In addition, there are concerns about the emotional stress and potential physical injury that animals may endure during the procedure,26 which may be particularly applicable for patients with comorbidities for which sympathetic tone increase can have a significant negative effect. Last, when heart murmurs are properly diagnosed and optimized for anesthesia, morbidity has not been shown to be increased for those patients.28
What are the anesthetic strategies for dogs with cardiac disease?
In dental practice, one of the most common conditions among dogs is mitral valve disease. If not previously diagnosed, patients should be screened for heart disease by physical examination, history, murmur auscultation, NT-proBNP (N-terminal pro–B-type natriuretic peptide) testing, and/or electrocardiography.29 In specialty dental practice, cardiology consultation and echocardiography are recommended and often pursued by the client, which provides the anesthetist the most robust database to formulate a risk-management plan.
Protocol design can be guided by echocardiography findings (e.g., left atrium to aortic root ratio, systolic function, degree of regurgitation, presence of arrhythmias).29 Staging of heart disease will help determine if the dental procedure should be rescheduled to a later date to allow time for further stabilization. For instance, dental procedures for dogs with newly diagnosed stage B2 heart disease should be delayed to allow for commencement of medical therapy (e.g., pimobendan), and dogs with stage C heart disease should be confirmed free of any acute signs of heart failure before being admitted for dental procedures. The increased risk associated with anesthesia should be discussed with the client, including options for specialty dentistry and anesthesiology referral.
Before the patient is anesthetized, pimobendan and diuretics should be continued and a minimum database obtained (including volume and electrolyte status) to help guide intraoperative fluid therapy. To avoid refractory intraoperative hypotension, anesthesiologists often request that angiotensin-converting enzyme inhibitors and angiotensin receptor blockers be discontinued for at least 24 hours before arrival at the hospital.30 Patient handling should involve minimal stress as endogenous catecholamine surge can worsen cardiac regurgitation and precipitate disease exacerbation or an acute heart failure event. If intravenous access can be obtained, premedication with an opioid is ideal, followed by 3 to 5 minutes of diligent preoxygenation and, if possible, placement of preanesthesia monitoring (e.g., electrocardiography, noninvasive blood pressure measurement, blood oxygen saturation) to ensure vital signs are stable before induction. Induction can be achieved with carefully titrated alfaxalone or propofol, often with benzodiazepine coinduction. Partial intravenous anesthesia techniques (e.g., fentanyl CRI) can reduce the vasodilation and negative contractility effects of inhalant anesthetics.
Anesthetic management strategies focus on maintaining high-normal heart rates, avoiding afterload increases, and supporting contractility; patients with advanced disease may benefit from receiving care at a facility with advanced monitoring capabilities.31 Dobutamine (pure β agonist) is a preferred agent for treating hypotension.28 Fluid volume delivery (including any CRI) should not exceed 2.5 to 3 mL/kg/hour.
During anesthesia and continuing into the postoperative period, the anesthetist should monitor for signs of fluid overload and congestive heart failure. Desaturation and decreased chest compliance are signs that the patient may benefit from furosemide. During recovery, the patient should receive oxygen and flow-by oxygen should be available. Before discharge, respiratory rate and effort and clear respiratory auscultation should be documented.
Even if the patient’s dental disease is not severe, referral may be warranted for anesthesia management capability. When heart disease is properly screened for and managed, morbidity among affected dogs that were anesthetized by trained personnel and monitored diligently during routine dental procedures was no higher than that among dogs with lower American Society of Anesthesiologists status.32
What are the anesthetic strategies for cats with chronic renal disease?
Among geriatric cats, chronic kidney disease (CKD) is highly prevalent (up to 80%).33 In addition, periodontal disease has been implicated as a risk factor for CKD.33,34 Geriatric cats should be screened for CKD and, if present, their renal perfusion should be optimized and clients should be counseled as to potential exacerbation of CKD. The disease process is not expected to resolve, but the progression varies based on many risk factors and the control thereof.35 Therefore, the risk-to-rewards balance of pursuing dental treatment should be weighed, considering the staging of the CKD, management and stability of the condition, risks from the dental disease, and overall prognosis for survival from the primary disease process, all of which may involve multiple specialties (e.g., dentistry, internal medicine, anesthesia). For example, patients with CKD at International Renal Interest Society (IRIS) stage I and with moderate to severe dental disease may be candidates for pursuing dental treatment, whereas patients at IRIS stage IV with mild dental disease may not be.
Helpful diagnostics to screen for CKD in new patients include physical examination, CBC and chemistry, SDMA (symmetric dimethylarginine), and blood pressure.36 Common abnormalities include azotemia, anemia, electrolyte imbalances, acidemia, and hypertension. Previously undiagnosed CKD patients should be referred for CKD staging and management before general anesthesia is pursued for dental care. Comorbidities (especially concurrent heart disease) add to the difficulty as the management strategies for heart and kidney disease may directly oppose each other.
As with cardiac disease, low-stress handling avoids the increased renal vascular resistance and decreased renal perfusion that result from sympathetic stimulation.36 Oral pretreatment can be helpful; however, if intramuscular premedication is required for catheterization, as is often the case for cats, low-dose dexmedetomidine or alfaxalone can avoid major shifts in hemodynamics. Benzodiazepines or opioids are also appropriate, but these agents alone will not confer adequate sedation if temperament is difficult; often they are used adjunctively with the former 2 options. Alfaxalone and propofol are equivalently adequate choices for induction as long as care is taken to prevent hypotension. Ketamine is more extensively renally excreted in cats and therefore is not a good choice for patients with advanced CKD.36
To protect against further renal insult, techniques that reduce inhalant MACs (e.g., fentanyl CRI, locoregional blocks) and aggressive blood pressure management with adrenergic agonists help prevent hypotension. Target mean arterial pressure should be 70 to 80 mm Hg, unless the cat has been chronically hypertensive, in which case 20% of baseline should be achieved. Invasive pressure monitoring is a helpful advanced monitoring technique to tightly regulate blood pressure.36,37 In addition, electrolyte imbalances or metabolic acidosis are common among patients with CKD; therefore, these values should be monitored and ventilation should be controlled. An arterial catheter also facilitates serial bloodwork as needed.
Discretion should be used regarding whether to continue fluid therapy during the postoperative period until discharge; additionally, NSAID therapy, if elected, should be administered with extreme caution. New literature is redefining when NSAIDs may be appropriate for patients with underlying kidney disease, but the findings are new and broad applications are not yet clear. However, if the degree of inflammatory dental pain warrants it, it may be worth the risk for CKD exacerbation to cautiously administer NSAIDs.29
Summary
Anesthesia and dentistry are specialties that have long existed in concert with each other. General practice encompasses both anesthesia and dentistry skill sets on a continuum from basic to advanced care, and it is up to the discretion of the practitioner to determine when a case exceeds the expertise of their clinical practice and should be referred. With anesthesia consultation, remote guidance, or in-person referral, patients with intricate comorbidities can still be provided dental care and returned to oral health, either by their primary veterinarian or through referral to a specialty practice.
References
- Monteiro BP, Lascelles BDX, Murrell J, Robertson S, Steagall PVM, Wright B. 2022 WSAVA guidelines for the recognition, assessment and treatment of pain. J Small Anim Pract. 2023;64(4):177-254. https://doi.org/10.1111/jsap.13566
- Bell A. The neurobiology of acute pain. Vet J. 2018;237:55-62. doi:10.1016/j.tvjl.2018.05.004
- Folino TB, Mahboobi SK. Regional anesthetic blocks. StatPearls. Updated January 29, 2023. Accessed September 9, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563238
- Kelly DJ, Ahmad M, Brull SJ. Preemptive analgesia I: physiological pathways and pharmacological modalities. Can J Anaesth. 2001;48(10):1000-1010. doi:10.1007/BF03016591
- Kelly DJ, Ahmad M, Brull SJ. Preemptive analgesia II: recent advances and current trends. Can J Anesth. 2001;48(11):1091-1101. doi:10.1007/BF03020375
- Lanz GC. Regional anesthesia for dentistry and oral surgery. J Vet Dent. 2003;20(3):181-186
- Erickson A, Harbin K, MacPherson J, Rundle K, Overall KL. A review of pre-appointment medications to reduce fear and anxiety in dogs and cats at veterinary visits. Can Vet J. 2021;62(9):952-960.
- Madan RD, Cenani A, Montgomery E, Azevedo T, Vernau KM, Brosnan RJ. Pregabalin produces similar effects as gabapentin for preanesthetic sedation in cats. JAVMA. 2023;262(3):359-363. doi:10.2460/javma.23.09.0493
- Taylor S, Gruen M, KuKanich K, et al. 2024 ISFM and AAFP consensus guidelines on the long-term use of NSAIDs in cats. J Feline Med Surg. 2024;26(4):1098612X241241951. doi:10.1177/1098612X241241951
- Bello AM, Dye C. Current perceptions and use of paracetamol in dogs among veterinary surgeons working in the United Kingdom. Vet Med Sci. 2023;9(2):679-686. doi:10.1002/vms3.1058
- Di Cesare F, Negro V, Ravasio G, Villa R, Draghi S, Cagnardi P. Gabapentin: clinical use and pharmacokinetics in dogs, cats, and horses. Animals (Basel). 2023;13(12):2045. doi:10.3390/ani13122045
- Gomes VH, Barbosa DJ, Motta AS, Corrêa CG, Moreno DJ, da Silva MF. Evaluation of nalbuphine, butorphanol and morphine in dogs during ovariohysterectomy and on early postoperative pain. Vet Anaesth Analg. 2020;47(6):803-809. doi:10.1016/j.vaa.2020.07.035
- Valtolina C, Robben JH, Uilenreef J, et al. Clinical evaluation of the efficacy and safety of a constant rate infusion of dexmedetomidine for postoperative pain management in dogs. Vet Anaesth Analg. 2009;36(4):369-383. doi:10.1111/j.1467-2995.2009.00461.x
- Iocolano KE, Looney A, Balkman CE, Hume KR, Boesch JM, Sylvester SR. Retrospective evaluation of outpatient intravenous ketamine-lidocaine infusions for the palliation of cancer pain in dogs and cats. JAVMA. 2024;263(4):499-506. doi:10.2460/javma.24.09.0595
- Niemiec BA: Regional anesthesia: In: Niemic BA, ed. Dental Extractions Made Easier. 2nd ed. Practical Veterinary Publishing; 2022:12-16.
- Woodward TM. Pain management and regional anesthesia for the dental patient. Top Companion Anim Med. 2008;23(2):106-114. doi:10.1053/j.tcam.2008.02.007
- Evans HE, Christesen GC. Cranial nerves. In: Hermanson JW, de Lahunta A, Evans HE, eds. Miller’s Anatomy of the Dog. 2nd ed. Saunders; 1970:903-934.
- Wallis C, Patel KV, Marshall M, et al. A longitudinal assessment of periodontal health status in 53 Labrador retrievers. J Small Anim Pract. 2018;59(9):560-569. doi:10.1111/jsap.12870
- Huffman LJ. Oral examination. In: Niemiec BA, ed. Small Animal Dental, Oral and Maxillofacial Disease: A Color Handbook. CRC Press; 2010:39-61.
- Colmery B. The gold standard of veterinary oral health care. Vet Clin North Am Small Anim Pract. 2005;35(4):781-787. doi:10.1016/j.cvsm.2005.02.005
- Niemiec BA. Professional teeth cleaning. J Vet Dent. 2003;20(3):175-180.
- Bellows J. Equipping the dental practice. In: Bellows J, ed. Small Animal Dental Equipment, Materials, and Techniques: A Primer. 1st ed. Blackwell Publishing; 2004:13-55.
- Niemiec B, Gawor J, Nemec A, et al. World Small Animal Veterinary Association Global Dental Guidelines. J Small Anim Pract. 2020;61(7):395-403. doi:10.1111/jsap.13113
- Holmstrolm SE, Fitch PF, Eisner ER. Dental prophylaxis and periodontal disease staging. In: Holmstrolm SE, Fitch PF, Eisner ER, eds. Veterinary Dental Techniques. 3rd ed. Saunders; 2002:175-232.
- Bellows J, Berg ML, Dennis S, et al. 2019 AAHA Dental Care Guidelines for Dogs and Cats. JAAHA. 2019;55(2):49-69. doi:10.5326/JAAHA-MS-6933
- Niemiec BA. The complete dental cleaning. In: Niemiec BA, ed. Veterinary Periodontology. Wiley-Blackwell; 2013:129-153.
- Stella JL, Bauer AE, Croney CC. A cross-sectional study to estimate prevalence of periodontal disease in a population of dogs (Canis familiaris) in commercial breeding facilities in Indiana and Illinois. PLoS One. 2018;13(1):e0191395. doi:10.1371/journal.pone.0191395
- Carter JE, Motsinger-Reif AA, Krug WV, Keene BW. The effect of heart disease on anesthetic complications during routine dental procedures in dogs. JAAHA. 2017;53(4):206-213. doi:10.5326/JAAHA-MS-6512
- Perkowski SZ, Oyama MA. Pathophysiology and anesthetic management of patients with cardiovascular disease. In: Lamont L, Grimm J, Robertson S, Love L, Schroeder C, eds. Veterinary Anesthesia and Analgesia. 6th ed. Wiley-Blackwell; 2024:680-696.
- Bailey K, Briley J, Duffee L, et al. The American College of Veterinary Anesthesia and Analgesia Small Animal Anesthesia and Sedation Monitoring Guidelines 2025. Vet Anaesth Analg. 2025;52(4):377-385. doi:10.1016/j.vaa.2025.03.015
- Goya S, Wada T, Shimada K, Hirao D, Tanaka R. Dose-dependent effects of isoflurane and dobutamine on cardiovascular function in dogs with experimental mitral regurgitation. Vet Anaesth Analg. 2018;45(4):432-442. doi:10.1016/j.vaa.2018.03.010
- Congdon JM. Cardiovascular disease. In: Johnson RA, Snyder LBC, eds. Canine and Feline Anesthesia and Co-existing Disease. John Wiley & Sons; 2014:1-54.
- Finch NC, Syme HM, Elliott J. Risk factors for development of chronic kidney disease in cats. J Vet Intern Med. 2016;30(2):602-610. doi:10.1111/jvim.13917
- Trevejo RT, Lefebvre SL, Yang M, Rhoads C, Goldstein G, Lund EM. Survival analysis to evaluate associations between periodontal disease and the risk of development of chronic azotemic kidney disease in cats evaluated at primary care veterinary hospitals. JAVMA. 2018 Mar 15;252(6):710-720. doi:10.2460/javma.252.6.710
- Cowgill LD, Polzin DJ, Elliott J, et al. Is progressive chronic kidney disease a slow acute kidney injury?. Vet Clin North Am Small Anim Pract. 2016;46(6):995-1013. doi:10.1016/j.cvsm.2016.06.001
- Clark-Price SC, Fischer JR. Physiology, pathophysiology, and anesthetic management of patients with renal disease. In: Lamont L, Grimm K, Robertson S, Love L, Schroeder C, eds. Veterinary Anesthesia and Analgesia. 5th ed. Wiley-Blackwell; 2024:922-843.
- KuKanich K, George C, Roush JK, et al. Effects of low-dose meloxicam in cats with chronic kidney disease. J Feline Med Surg. 2021;23(2):138-148. doi:10.1177/1098612X20935750
____________________________________________________________________________________________________________________________________________________________
TVP – Today’s Veterinary Practice Logo Articles | October 6, 2025 |
Diagnosing and Treating Chronic and Recurrent Otitis Externa
Avoiding cycles of inflammation that can lead to end-stage otitis depends on early recognition of primary, secondary, predisposing, and perpetuating factors and aggressive intervention.
Helena Montin Mills DVM, Annette Lundberg DVM, MS, DACVD
Abstract
Otitis is a common disease among dogs. Without early and effective intervention, it can progress to chronic and recurrent otitis externa (CROE), which can be challenging to treat. The clinical framework of primary causes, secondary contributors, predisposing factors, and perpetuating factors is useful for creating treatment plans. General physical examination helps practitioners prioritize primary causes. Otoscopic examination can identify early and reversible chronic changes. External palpation of the ear canals can help detect suspicion for mineralization. The most valuable diagnostic tool is cytology because it enables identification and targeted treatment of the organisms involved in the infection. For treatment, most patients with CROE will benefit from oral and/or topical glucocorticoids, the selection of appropriate antimicrobials, ear cleaning, and control of the primary causes. Regular recheck examinations evaluating otalgia and treatment response are crucial to treatment success, after which maintenance therapy is needed to prevent relapse.
Take-Home Points
Successful management of chronic and recurrent otitis externa requires an understanding of primary causes, secondary contributors, predisposing factors, and perpetuating factors. Chronic and recurrent otitis externa is the result of inadequately controlled primary causes. Oral and/or topical glucocorticoids can reverse early chronic changes to the ear canals such as hyperplasia, but mineralization cannot be reversed. Recheck examinations with cytology are crucial in monitoring patient comfort and treatment response. Bacterial culture and susceptibility is controversial for otitis externa, but may have a place in identifying organisms and in a public health perspective. Maintenance therapy with ear cleanings, low-potency corticosteroids, and control of underlying diseases can help to prevent relapses. Otitis remains one of the most common diseases among dogs.1 To help categorize clinical cases and formulate therapy, a recent review proposed definitions for recurrent and chronic otitis (see GLOSSARY).2 Successful management and treatment of chronic and recurrent otitis externa (CROE) can be challenging because they require an understanding of the pathogenesis, diagnostics, and multimodal therapy.2-5 Preventing chronic and irreversible changes to the ears requires early recognition and medical intervention.2,3,5 Furthermore, patience and adherence from clients and patients are vital for positive outcomes.3,6 Appropriate intervention leads to better patient outcomes, client satisfaction, improved antimicrobial stewardship,2,6 and ultimately saves ears from end-stage CROE requiring surgical removal through total ear canal ablation and bulla osteotomy (TECABO).3,7-9
Glossary Otitis Inflammation of the pinnae and/or ear canals Ear infection Clinically significant microbial overgrowth or infection
Recurrent Clinically significant ear inflammation and/or infection within 3 months of complete resolution of previous episode
Acute Otitis without acquired proliferative changes in the ear canals
Chronic Otitis with acquired proliferative changes in the ear canals
Pathogenesis of CROE Understanding the primary, secondary, predisposing, and perpetuating (PSPP) factors of otitis externa (OE) can help clinicians create effective treatment plans (TABLE 1).2,3,5,10,11 Identifying the causes through a detailed history and physical examination prioritizes differentials and diagnostic workup.2-5,10 CROE may have more than one primary cause and multiple other contributing factors.3,5,10
Primary Causes Primary causes of OE are conditions that can induce clinical disease in a healthy ear with no secondary, predisposing, or perpetuating factors (i.e., dermatologic diseases that also affect the ear epithelium).2-4,10 Atopic dermatitis is the primary cause in 43% to 75% of dogs with OE and in 31% to 50% of dogs with CROE.12-14 Cases of unilateral otitis should prompt prioritization of other causes such as foreign bodies (e.g., plant awns), otitis media (OM), polyps, and neoplasia (particularly in cats as allergic otitis is less common).15 For successful management of CROE, obtaining an accurate diagnosis for the underlying cause and its appropriate management is crucial.2-4,10
Secondary Contributors Secondary contributors to otitis lead to clinical disease in abnormal ears,3-5 which compound the effects of primary causes, exacerbating clinical signs.2,3 Bacterial or yeast infections of the ear can incite inflammation3; however, they result from a combination of primary causes, predisposing factors, and perpetuating factors.3 Ear infections become chronic or recurrent despite treatment of the infection itself because of uncontrolled underlying disease.3
Predisposing Factors Predisposing factors are present before the onset of otitis, alter the ear canal anatomy and physiology, and increase the likelihood of OE development.3,11 Alone, each factor is unlikely to cause disease.2,11 However, the presence of any predisposing factor(s) warrants active surveillance for the development of primary causes, which may then lead to otitis.10,11 A classic example is the pendulous pinnae of basset hounds, especially because not every dog breed with pendulous pinnae is at increased risk for OE.11,16 Some individuals and breeds may have multiple predisposing factors.3,11,17
An often overlooked predisposing factor is iatrogenic trauma from hair plucking or vigorous cleaning that induces inflammation or injury to delicate epithelial cells.11 The authors believe that hair plucking may be required for select patients (e.g., those with entrapped biofilm or matting) and should be done responsibly with use of appropriate analgesia and anti-inflammatory medications.8
Perpetuating Factors Perpetuating factors include changes to the normal anatomy and physiology resulting from chronic inflammation and OM; ultimately, they are the result of uncontrolled otitis.2,18 These changes are initially very mild but become prominent with time. The significance of perpetuating factors is that they sustain otitis regardless of resolution or control of the primary cause.2,3,5
The first change detectable during otoscopic examination is a cobblestone appearance of the lining of the canal walls, resulting from epidermal nodular and glandular hyperplasia (FIGURE 1).2,3,5 Treatment at this stage can most effectively reverse the changes and prevent end-stage otitis; however, if untreated, the changes progress to epidermal and dermal hyperplasia, stenosis, fibrosis, and finally mineralization (FIGURE 2).2,3 Although medical management may be able to reverse hyperplasia and fibrosis, it cannot reverse mineralization of the canal; therefore, ears with mineralized canals are often called end-stage ears,2,3 at which stage a TECABO may be recommended to improve patient comfort by removing a source of pain.2,3,7,9
The above changes can all result in OM and tympanic membrane alterations, if not rupture.2,3,18,19 The natural, self-cleaning mechanism of the ear becomes disrupted, and cerumen along with other debris will no longer migrate out of the canal.2,3,5,8
Diagnosis of otitis media/interna (OMI) can be more apparent when patients exhibit Horner syndrome, facial paralysis, or peripheral vestibular signs.3,19 However, in a study evaluating 121 dogs with CROE and no overt evidence of OMI in either their history or clinical presentation, magnetic resonance imaging (MRI) detected subclinical OM in 20.8% of dogs.18 OM should be considered for patients with recurrent otitis, an abnormal tympanic membrane, and a history of treatment failures.3,19,20
Clinical Presentation of CROE History Patients with CROE can exhibit a variety of clinical signs (e.g., head shaking, malodorous ears, hearing loss).3 Hearing loss caused by OE can be conductive or sensorineural.3,5,21,22 In a study of dogs referred for chronic OE, hearing deficits were bilateral for 53% and unilateral for 25%.21 The more severe cases of hearing loss were statistically significantly associated with OM; therefore, patients with hearing loss should be referred for advanced imaging so they can receive appropriate diagnosis and treatment.21
OE always results from an underlying disease that must be addressed to minimize recurrence. A detailed history regarding recurrent skin infections, pruritus or pruritic behaviors, and seasonal changes may reveal concerns for atopic dermatitis. Alternatively, changes in appetite, thirst, urination, and energy levels may indicate an endocrinopathy.
Physical Examination The entire patient should always be examined because general examination can provide valuable insight into the underlying cause, perpetuating factors, or adverse drug effects. Oral examinations are relevant for detecting potential diseases affecting the middle ear or temporomandibular joint.3,19
The clinical signs of OE depend on the primary cause, secondary infections, and chronicity.3 Ear canal palpation is a valuable part of examination, tolerated to some degree by most patients. A healthy ear canal is made up of soft and pliable cartilage.2,3,23,24 Chronic inflammation makes the ears firm, thickened, and nonpliable, often corresponding to the degree of hyperplasia and calcification.2,3 Palpation also helps with assessment of facial asymmetry and identifying unilateral versus bilateral disease.
External visual examination of the ear canal can reveal hyperplasia and stenosis of the vertical canal (FIGURE 3),3,8 which may be reversible depending on whether the cartilage in the canal has mineralized.3 A subset of patients will have purulent exudate with erosions to ulcerations, which may indicate Pseudomonas species infection.2,25 These conditions typically are particularly painful, although pain is likely underrecognized in most patients with chronic otitis.3,25 Characterizing the exudate can provide insight as to expected cytologic findings; however, ear cytology should never be replaced by smell or macroscopic visual inspection.
FIGURE 3. Canine hyperplastic otitis. Courtesy: Dr. Sandra Koch
When otoscopic examination is possible, a cobblestone appearance of the canal epithelium indicates ceruminous gland hyperplasia. Concurrent stenosis or masses can be seen.2,3,5 For dogs of certain breeds, especially brachycephalic breeds, the normal horizontal canal is often slightly stenotic, which is a predisposing factor for OE and should not be confused with pathologic stenosis.3,8,25 Hair may be present within the canals of breeds such as poodles, bichon frises, and terriers, making deep structures difficult to visualize.3,8
A healthy tympanic membrane is translucent, but when chronically inflamed it becomes opaque and thickened and/or ruptures.2,3,5,19 Depending on their size, partial ruptures can be difficult to diagnose. In stenotic ears, visualization of the tympanic membrane may not be possible.18,26 Note that an intact tympanic membrane does not rule out OM.3,18,27,28
OMI may be identified during examination of facial symmetry and neurologic examination. Head tilt can sometimes be a nonspecific finding because it can result from pain, causing abnormal head carriage, or from temporary or permanent sequelae to vestibular syndrome.3 Middle ear disease can damage the postganglionic sympathetic neurons, leading to Horner syndrome: miosis, third eyelid elevation, enophthalmos, and ptosis.3,22 In dogs, it is more common to also see facial nerve paralysis from middle or inner ear disease.3,22 These dogs can have neurogenic keratoconjunctivitis sicca, making tear production evaluation relevant.3,22 Patients with neurologic deficits benefit greatly from early referral for diagnosis and intervention.
Diagnostics for CROE Ear Cytology Ear cytology is a foundational part of chronic otitis management because it enables quick identification and quantification of infectious organisms and inflammatory cells. Ear cytology should be performed at every visit to help measure treatment effectiveness and particularly at the end of a treatment course to prove infection resolution. Cytology may reveal cocci (usually Staphylococcus species), rods (e.g., Pseudomonas, Proteus), or yeasts (e.g., Malassezia species).2,5,29,30 Rarely, filamentous fungi (e.g., Aspergillus) are also identified.3,30 Inflammatory and red blood cells are not seen in healthy ears and enter the lumen of the canal only through ulceration, exudate, masses, or middle ear effusion.29 The presence of rods and inflammatory cells is associated with a higher risk for OM.18,28
Biofilm can be produced by a variety of organisms, including Malassezia and Staphylococcus, but predominantly Pseudomonas.2,31 Most bacteria noted on cytology are in their planktonic form; however, a biofilm is an aggregate of bacteria suspended in a liquid matrix.2,31 Presence of biofilm can be suggested by clinical and cytologic evaluation of sticky mucus and a net-like cluster of bacteria and neutrophils (FIGURE 4).2,31 However, a definitive diagnosis requires specialized testing, such as periodic acid–Schiff staining.32 Identification of biofilm is valuable because biofilm makes the bacteria less susceptible to antibacterial agents and increases adherence to the epithelium, both of which can make infections more difficult to resolve.2,31
FIGURE 4. Oil immersion showing biofilm containing rod bacteria and inflammatory cells from a dog with chronic otitis externa secondary to a mass. Diff-Quik stain, 1000× magnification. Courtesy: Dr. Kim Devos Davies.
Bacterial Aerobic Culture and Susceptibility The usefulness of ear aerobic bacterial culture and susceptibility for OE treatment is controversial. When interpreted along with cytology, culture results can provide insight to predominant pathogens.2,3,29 The controversy arises from the lack of clinical breakpoints for topical formulations, and the in vivo therapeutic concentrations for topical therapy are significantly higher than plasma drug concentrations.2,3,5,33 Culture results do not seem to be predictive of in vivo response.34 The literature reports that cultures are potentially useful for cases of atypical bacterial infections or failure to respond despite adequate treatment and adherence.2,5 It is the authors’ opinion that with adequate treatment of the underlying disease and anti-inflammatory therapy, proper application technique, and selection of medications and cleanser, along with compliant clients and patients, true treatment failure resulting from resistance is uncommon. However, from a public health perspective, identifying multidrug-resistant bacteria is useful because resistance genes can be shared between bacteria. In addition, bacterial culture and susceptibility of material collected from within the tympanic bulla during a myringotomy is recommended because OM is frequently treated with a systemic antibiotic, whereas OE is treated topically.19 Although the relevance of ear cultures will remain controversial, a smaller sample study showed cultures better correlated with cytology than with 16S rRNA gene profiling.35
Advanced Imaging Advanced imaging is crucial for detecting masses, congenital abnormalities, mineralization of the external ear canals, and OMI. The decision between computed tomography (CT) and MRI depends on case presentation, differentials, and availability.18,28 CT is better for evaluating osseous tissue, including evaluating the thickness of the bullae and mineralization of the external canal (FIGURE 5).28 For patients with neurologic deficits, depending on the constellation of clinical signs, MRI may be preferred because it enables better evaluation of soft tissue (FIGURE 6). When a mass is suspected, contrast should be used if available and not contraindicated.
Treatment of CROE At-Home Ear Cleaning When done correctly, ear cleaning is a crucial component of managing OE because it removes exudate, debris, and foreign material, which affect the effectiveness of medications and serve as nidi for infection.8 Clients must be shown how to correctly clean the ears, including introducing the product into the ear, gently massaging the ears if it is comfortable for the patient, and then allowing the patient to shake its head.8 Clients can gently clean debris from the visible parts of the ears but should never insert objects (e.g., a cotton-tip swab) into the canal as it can cause an impaction and trauma.8
The market for ear cleaners is continually changing; therefore, being familiar with desired active ingredients is recommended (TABLE 2).8,36 Many of the active ingredients can potentially irritate the middle ear or be ototoxic; therefore, when the status of the tympanic membrane is unknown, a good option for minimizing risk for ototoxicity is use of tromethamine (tris) and EDTA-tromethamine (tris-EDTA).37
Patients with CROE may be very sensitive to topical medications.8 If a contact reaction is suspected, discontinue topical therapy and allow the ears to recover; saline should be considered as a safe alternative if cleaning is necessary.8 Some ear cleaners may also contain topical glucocorticoid(s). If one of these cleaners is to be used, choose a product with low-potency glucocorticoid(s) and use it infrequently to avoid the adverse effects of steroids.8,20
In-Clinic Ear Cleaning At-home cleaning may be sufficient for patients with acute OE; however, in-clinic cleaning or deep ear flushing may need to be performed in clinic for patients with more chronic OE. When material is tightly adhered deep in the canal, as is common for patients with CROE, deep ear flushing by a veterinarian is usually recommended. Recent studies on collaborative care showed deep ear flushing leads to faster patient improvement and resolution of otitis.6,38,39 The distinction between in-clinic ear cleaning and deep ear flushing is that in-clinic ear cleaning using commercial cleaners can be helpful when the client is unable to perform ear cleaning at home40; however, deep ear flushing involves visualization of the canal and tympanic membrane to remove as much material as possible while often using specialized equipment to mitigate damage.8,20
Patients undergoing deep ear flushing need to be anesthetized with a protected airway in case of tympanic membrane rupture or compromise because the fluid will drain through the auditory canal and into the oropharynx.8,20 Ideally, irrigation is performed through a video otoscope.3,8,20 If a video otoscope is not accessible, a hand-held otoscope with a red rubber catheter and a syringe may enable adequate flushing, although visualization throughout the procedure may be more difficult.3,8,20 Saline is typically considered the best option for flushing because it is safe for the middle ear and provides excellent visibility.3,8,20 Soaking with a ceruminolytic product could be considered before flushing.8
Complications may result from deep ear flushing and anecdotally seem to be more common among cats; therefore, the risks should be discussed with clients before flushing.8 Commonly reported complications include ruptured tympanic membrane, peripheral vestibular syndrome, facial nerve paralysis, Horner syndrome, and hearing loss.3,8,20
Antimicrobial Therapy Topical
Historically, topical antimicrobial therapy has been empirically based on cytology and tympanic membrane status.2,3,41,42 If tympanic membrane integrity is compromised, ototoxic medications (TABLE 3) should not be used.3,41-44 It has recently been suggested to also base therapy on antibiotic tiers, similar to those outlined in the International Society for Companion Animal Infectious Diseases guidelines for antimicrobial use for pyodermas.2,45,46
A study evaluating chronic, unresponsive Malassezia infections found that within 4 weeks of initiating a course of glucocorticoids, performing a deep ear flush, and following up with otic antifungal ointment resolved the infections.39 Common antifungals include the azoles (clotrimazole, miconazole, and posaconazole), allylamines (terbinafine), and polyene (nystatin).2,41 Empiric treatment failure is often a sign that a PSPP factor is unidentified or inadequately addressed or that cleaning and medication application have been improperly performed.3,5,8
Systemic
Use of systemic antimicrobials for OE has been controversial and is now discouraged with the emergence of antimicrobial resistance and emphasis on antimicrobial stewardship as the likelihood of obtaining therapeutic drug concentrations within the ear lumen is low.2,41,47 Use of systemic antimicrobials should be reserved for patients with OM with or without otitis interna and ideally based on culture and susceptibility of material from within the tympanic bulla.2,20,41,46,47
Glucocorticoids
Glucocorticoids provide the best chance for reversing chronic changes from inflammation and should be considered for OE patients with stenosis, erythema, ulcerations, or pain.48,49 Early use of systemic glucocorticoids is associated with better outcomes and fewer relapses of CROE.6 Prescribing considerations include case selection, treatment phase (induction or maintenance), formulation, potency, and route of administration. Patients that are sensitive to glucocorticoids may experience fewer adverse effects from different formulations of triamcinolone, methylprednisolone, or dexamethasone.49 For some patients, glucocorticoids are contraindicated and alternative therapy should be investigated, which may make treatment challenging.
Treatment Phases of CROE Induction The goals for the induction phase are to improve patient comfort, identify and address PSPP factors, treat infection, and determine whether permanent physiologic changes are present.48 The success of the treatment plan strongly depends on patient and client adherence.2,3 Patients often have otalgia and form negative associations with the ears.2,3,50 Reassessing pain and behavior at every visit enables the practitioner to intervene with pain medication and anxiolytics to improve patient comfort and therefore adherence to the treatment plan.
Typically, an induction dose of glucocorticoids starts at the high end of the anti-inflammatory dosing range and continues for 1 to 3 weeks.2,20,41,48,49 Depending on the degree of pain, it can take a few days of systemic glucocorticoids before the patient will tolerate topical ear medications, after which it is recommended to start a topical glucocorticoid for a local effect.49 Similar to systemic glucocorticoids, the potencies of topical formulations vary (TABLE 4). Fluocinolone, which is in Synotic (fluocinolone acetonide 0.01% and dimethyl sulfoxide 60%; Zoetis, zoetis.com), is among the most potent glucocorticoids. Synotic also has the benefits of dimethyl sulfoxide, and together these ingredients can effectively address hyperplasia.20,41,48,49 Betamethasone, dexamethasone, hydrocortisone aceponate, and mometasone furoate are all potent glucocorticoids and are appropriate for use during the induction phase. Hydrocortisone aceponate and mometasone furoate are considered “soft steroids” and are less likely to cause local and systemic adverse effects.51 However, even these steroids can be systemically absorbed.48,49,52,53 The steroid vehicle also contributes to potency.3,41,49 Ointments are more potent because they form an occlusive barrier.49 For adequate effect, recommended frequency for administration of most otic glucocorticoids is twice daily.49
During the induction stage, ear cleaning and topical antimicrobials are started. The topical antimicrobial selection should be based on cytology results. Many commercially available otic medications contain a combination of a topical steroid, an antibacterial agent, and an antifungal agent. However, more targeted treatment is based on cytology (e.g., using a sole topical azole for Malassezia infections). Furthermore, an in vitro study showed that miconazole has some antistaphylococcal activity.54 Because mechanical removal of material from the ear canal via cleaning is a valuable component of treatment, it is the authors’ opinion that long-lasting topical gels be used only for patients for which at-home treatment is not possible and that lanolin-based products be avoided entirely. When used, long-lasting topical gels should be applied after thorough in-clinic cleaning and verification of an intact tympanic membrane. If otalgia precludes medicating at home, long-lasting topical gels can be considered to improve patient comfort while clients can practice veterinarian-guided behavior modification to help reset negative associations with ear interactions to enable future at-home ear treatment.
To monitor treatment response, patients with CROE should ideally be evaluated every 2 to 4 weeks until resolution of infection and abnormal otoscopic findings.41 The induction phase ends when only irreversible chronic changes remain and infection is resolved.
Maintenance The goal for the maintenance phase is to prevent recurrence of OE. The focus should be to reduce inflammation in the ear as a noninflamed ear canal is less likely to support bacterial or yeast overgrowth.5,55 After the infection is treated and stenosis is improved, systemic glucocorticoids should be tapered down to the lowest effective dose and frequency.48,49 PSPP factors should be reassessed to ensure that infection and stenosis remain controlled. Depending on the primary etiology, this can be a time to transition to other allergy medications. For patients with severe cases, modified cyclosporine is frequently a good option for long-term control and may prevent the need for surgery.56
For patients with CROE, at-home maintenance ear cleaning should be performed 1 to 2 times weekly.8 Regular intermittent administration of the least-potent steroid possible can help decrease systemic medications needed for long-term control and prevent flares.48,55 In addition, topical otic solutions containing aqueous solution of aluminium triacetate (Burow’s solution) or enzymatic solutions can be used a few times per week to help prevent infection return.48,49
Effects of CROE Treating CROE is associated with a significant emotional and financial burden for clients.2,6,38 Patients and clients experience reduced quality of life as a result of CROE, and it can also fracture the human–animal bond.6,38 In the authors’ experience, initial encounters at the referral level for patients with CROE can be very emotionally charged, particularly when concern for end-stage otitis is raised. Recent studies showed that outcomes are worse for patients that have had CROE for more than 6 months from diagnosis to referral.6,38 Another benefit of early referral is availability of the special equipment for video otoscopy and deep ear flushing, which has been associated with improved patient outcomes.6,38,39
When clinical signs concerning CROE are found during examination, appropriate expectations should be explained to the client (i.e., if these changes are not reversed, they can lead to end-stage otitis). Medical management of end-stage otitis is unlikely to be successful over the long term without flares, and affected ears are a source of pain. A TECABO is considered a salvage procedure to remove the painful and infected canals.2,3,7,9,57 Although surgery is not free of potential complications, if surgery goes well, clients usually report that their dog is more playful and has a significantly improved demeanor, underscoring how much unaddressed pain accompanies CROE.7
Summary CROE is a complex, multifaceted condition with a significant emotional and physical burden to patients and clients. Understanding all the factors leading to its development is crucial for treatment success. For patients with CROE, glucocorticoids provide the best chance of reversing detrimental changes and improving patient comfort. Topical treatment of secondary infections should be based on cytology, with or without culture. Systemic antimicrobials are indicated for treatment of OMI. Chronic cases may require advanced diagnostics and procedures that are not always available in a primary care setting. When treatment for the initial stages of CROE is successful, long-term management is necessary to prevent further recurrence. Ultimately, avoiding cycles of inflammation that can lead to end-stage otitis depends on early recognition of PSPP factors and aggressive intervention.
_____________________________________________________________________________________________________________________________________________________________
Journal of Veterinary Internal Medicine ACVIM I 02 October 2025
Evaluation of a Rapid, Low-Cost Broth Turbidity Test for Detecting Ampicillin-Resistant Lower Urinary Tract Infections in Dogs and Cats
W. Cooper Brookshire | Larry Ballard | Anna Collinsgru | Josh Burnette | Khadija Ferdous | Joo You Park |Keun Seok Seo
Funding: This work was supported by the National Institutes of Health NIGMS (U54GM115428).
ABSTRACT
Background
Effective treatment of urinary tract infections (UTIs) in dogs and cats relies on timely recognition of antimicrobial resistance, but traditional microbiological culture and susceptibility testing require 48–72 h and can be cost-prohibitive.
Objective
To evaluate a novel, low-cost broth turbidity test for detecting ampicillin-resistant UTIs in dogs and cats compared to the gold standard microbiological methods.
Animals
One hundred sixty urine samples from 145 dogs and 15 cats at the Mississippi State University College of Veterinary Medicine.
Methods
From September 2023 to February 2024, urine samples were tested using an ampicillin-supplemented Mueller–Hinton broth turbidity test and gold standard culture with Sensititre MIC plates. Sensitivity, specificity, Cohen’s κ, and McNemar’s test were calculated.
Results
The broth turbidity test showed 89.47% sensitivity (95% CI: 66.86%–98.72%) and 100% specificity (95% CI: 97.42%–100%) for the detection of ampicillin-resistant organisms in the urine, with substantial agreement (κ = 0.94, p < 0.001) and no classification difference (p = 0.500). Notably, all 36 samples with ampicillin-susceptible organisms identified by the gold standard cultures test were correctly identified as negative for ampicillin resistance by the broth turbidity test.
Conclusions
The broth turbidity test’s simplicity and affordability make it a promising tool for in-house use by veterinary practitioners to guide first-line therapy decisions, though its sensitivity could be limited in cases with low bacterial loads or confounding factors such as recent antibiotic administration.
Abbreviations
|
|
|
|
|
|
|
|
|
|
1 Introduction
Urinary tract infections (UTIs) are prevalent in dog and cat populations, often requiring prompt antimicrobial therapy to prevent complications such as pyelonephritis or recurrent infections. Amoxicillin and potentiated sulfonamides are both considered first-line treatments for uncomplicated UTIs, but amoxicillin is preferred by many practitioners due to its efficacy, safety, and cost-effectiveness [1]. Amoxicillin is excreted into the urine at a high concentration, which likely reduces the need for clavulanic acid for successful treatment of many infections [1]. However, the rising prevalence of ampicillin-resistant bacteria, particularly methicillin-resistant Staphylococcus pseudintermedius and extended-spectrum beta-lactamase-producing Enterobacterales, has complicated empirical therapy, leading to treatment failures and the use of second-line antibiotics [2]. Traditional microbiological culture and susceptibility testing, while the gold standard, are time-consuming (48–72 h) and usually require submission to off-site diagnostic laboratories [3]. Published veterinary guidelines state that “aerobic bacterial urine culture is preferred for all cases of suspected bacterial cystitis,” but surveys suggest that practitioners commonly prescribe antibiotics empirically for these cases, with cost being a major limiting factor [1, 4–7]. Low-cost diagnostic tools are essential for veterinarians working in resource-limited settings and for those providing care across the spectrum of care model. These tools support decision-making by enabling diagnostic testing that aligns with clients’ financial limitations while still promoting responsible antimicrobial use [8].
Rapid, cost-effective point-of-care diagnostic tools to detect ampicillin resistance in dog and cat UTIs are lacking, hindering timely and appropriate treatment decisions. Such tools are needed to guide first-line therapy, reduce unnecessary antibiotic escalation, and support antimicrobial stewardship.
The objective of this study was to develop a rapid, cost-effective, user-friendly microbial assay to reliably detect ampicillin-resistant bacteria in urine samples within 16–24 h as a point-of-care diagnostic. If the pathogen grows in the presence of the indicator drug (ampicillin), the broth will become visibly turbid, indicating positive growth of an amoxicillin-resistant microbe. A positive result will prompt the clinician to submit the sample for traditional culture and susceptibility testing at an accredited laboratory, thus avoiding inappropriate first-line therapy resulting in likely improved patient outcomes. This study aimed to evaluate the performance of the experimental broth turbidity test against traditional microbiological methods in detecting ampicillin-resistant lower UTIs in dogs and cats. We hypothesized that the test would exhibit high sensitivity and specificity for the detection of ampicillin-resistant bacteria in dog and cat urine, making it a practical tool for in-house use by veterinary practitioners.
2 Materials and Methods
2.1 Study Design and Sample Collection
This study was conducted in accordance with the Standards for Reporting of Diagnostic Accuracy Studies (STARD) guidelines to ensure transparent and comprehensive evaluation of the broth turbidity test’s diagnostic accuracy [9]. Urine samples submitted for routine culture and antibiotic susceptibility testing (AST) at the Mississippi State University College of Veterinary Medicine (MSU-CVM) Diagnostic Laboratory from September 2023 to February 2024 were tested in parallel using the experimental broth turbidity test. A total of 160 de-identified urine samples collected via cystocentesis from 145 dogs and 15 cats were used, obtained as residual samples from routine diagnostic submissions unrelated to this study. The MSU-CVM Institutional Animal Care and Use Committee (IACUC) reviewed the study and determined that no IACUC protocol was required, as no live animals were involved and only pre-existing diagnostic samples were analyzed. Owner consent for diagnostic testing was obtained as part of standard clinical procedures at the MSU-CVM Diagnostic Laboratory.
Due to the absence of published data on the broth turbidity test’s performance, a sample size of 160 urine samples was selected based on unpublished antibiogram data from Mississippi State University (2022–2023), estimating a 15% prevalence of ampicillin-resistant bacteria in dog and cat urine cultures. Assuming a 15% prevalence and at least a 90% sensitivity for the broth turbidity test (informed by our unpublished pilot experiments), approximately 24 resistant cases were expected, yielding a 95% confidence interval (CI) for sensitivity with a width of approximately ±10% using the Clopper–Pearson method [10]. This precision was deemed sufficient for evaluating diagnostic accuracy, consistent with STARD guidelines [9].
2.2 Gold Standard Traditional Microbiological Methods
Gold standard testing followed standard protocols for urinary bacterial culture and AST, as described by the Clinical Laboratory Standards Institute (CLSI). In summary, a calibrated 1 μL sterile loop was used to inoculate blood agar, Columbia nalidixic acid (CNA) agar, MacConkey agar, and an enrichment broth with urine. After 24 h of aerobic incubation at 37°C, colonies were manually selected from agar plates, isolated, and identified using Sensititre gram positive or negative ID plates (Thermo Fisher Scientific). If agar plates failed to grow after 24 h but the enrichment broth exhibited growth, subcultures were performed for bacterial isolation. Ampicillin susceptibility was determined using Sensititre CMV1BURF minimum inhibitory concentration (MIC) plates, with results interpreted per CLSI guidelines. An organism was classified as resistant (positive) if its MIC for ampicillin exceeded the CLSI susceptible breakpoint for lower UTIs in dogs and cats (8 μg/mL) [11].
2.3 Experimental Broth Turbidity Test
The broth turbidity test was designed for simplicity and affordability. A clear sterile polystyrene tube containing 2.5 mL of sterile cation-adjusted Mueller–Hinton (MH) broth and two 10 μg ampicillin discs (corresponding to the CLSI breakpoint of 8 μg/mL) were added. The tube was vortexed to ensure disc dissolution, then inoculated with 20 μL of urine (two 10 μL sterile loops). Twenty-microliters of urine was used as the inoculation volume based on our unpublished pilot studies that suggested it would be a reasonable volume. Broth tubes were incubated aerobically in a non-shaking incubator at 37°C overnight for 16–24 h. After incubation, tubes were gently hand-rocked to suspend any bacteria and visually assessed by two independent and blinded observers for turbidity compared to a negative control tube, with apparent turbidity indicating growth of ampicillin-resistant bacteria (positive), as demonstrated in Figure 1. Tubes with no visible turbidity were classified as no growth of ampicillin-resistant bacteria (negative). Observers were blinded to the results of the gold standard test at the time of turbidity assessment; gold standard results were not available until 24–48 h after turbidity was assessed.
FIGURE 1
Open in figure viewerPowerPoint
Broth turbidity test visual comparison. Tube A = negative control tube; Tube B = positive sample.
2.4 Statistical Analysis
The performance of the broth turbidity test was compared to the gold standard for detecting ampicillin-resistant bacteria. A 2 × 2 contingency table was constructed to calculate true positives, true negatives, false positives, and false negatives. Sensitivity and specificity were computed, with 95% CIs determined using the exact binomial (Clopper–Pearson) method via an online calculator [10]. Cohen’s κ was calculated to assess agreement beyond chance, and McNemar’s test was used to evaluate differences in classification between the two methods. Statistical analyses were performed using SPSS (version 28, IBM Corp.) or the Soper online calculator, with significance set at p < 0.05.
3 Results
3.1 Summary of Bacterial Species and Ampicillin MICs
Urine from 145 dogs and 15 cats submitted for routine AST was used for the study. Of the 160 urine samples analyzed, the gold standard method identified bacterial growth in 55 samples (34.4%), while 105 samples (65.6%) showed no growth. Among the 55 samples with bacterial growth, 36 (65.5%) contained only ampicillin-susceptible isolates (MIC ≤ 8 μg/mL), and 19 (34.5%) had at least one ampicillin-resistant isolate (MIC > 8 μg/mL). The experimental broth turbidity test, designed to detect ampicillin-resistant organisms, showed growth in 17 samples (10.6%) and no growth in 143 samples (89.4%). The broth test correctly classified all 36 ampicillin-susceptible samples as negative for ampicillin-resistant organisms (no growth) but misclassified 2 of the 19 gold standard identified ampicillin-resistant samples as no growth (negative for ampicillin resistance), indicating false negatives. Interobserver agreement for the broth turbidity test was 100%, with both observers classifying all samples the same (negative vs. positive). A 2 × 2 contingency table summarizing the detection of resistant organisms is presented in Table 1. The broth turbidity test demonstrated a sensitivity of 89.47% (95% CI: 66.86%–98.72%) and a specificity of 100% (95% CI: 97.42%–100%) for detecting ampicillin-resistant organisms. Cohen’s κ was 0.94 (p < 0.001), indicating almost perfect agreement between the broth turbidity test and the gold standard. McNemar’s test showed no significant difference in classification between the two methods (p = 0.500, exact test, two-sided).
TABLE 1. Contingency table for the broth turbidity test versus gold standard test.
|
Gold standard |
Broth turbidity positive |
Broth turbidity negative |
Total |
|
Pos |
17 (True positives) |
2 (False negatives) |
19 |
|
Neg |
0 (False positives) |
141 (True negatives) |
141 |
|
Total |
17 |
143 |
160 |
Susceptible isolates included a variety of species, predominantly Escherichia coli (n = 10) and Enterococcus faecalis (n = 7), with MICs ranging from < 0.12 to 4 μg/mL. Of the 36 ampicillin-susceptible cultures, 6 contained two isolates each. Resistant isolates were primarily E. coli (n = 11), with MICs ranging from 32 to > 256 μg/mL, alongside other species such as Klebsiella pneumoniae and Enterobacter cloacae. Of the 19 ampicillin-resistant cultures, 6 contained two isolates. Three of these cultures contained two morphologically distinct E. coli isolates, which were identified based on differences in colony morphology noted during isolation. Two discrepant result cultures were noted: one contained a mixed culture of S. pseudintermedius (MIC < 2 μg/mL) and Proteus spp. (MIC 16 μg/mL), and the other contained a β-lactamase-producing E. coli isolate (MIC > 256 μg/mL). Both discrepant result samples tested negative in the broth turbidity test despite demonstrating resistance by the gold standard. Table 2 summarizes the bacterial species and MIC ranges, categorized by susceptibility, resistance, and discrepant results.
TABLE 2. Summary of bacterial species and ampicillin MICs.
|
Category |
Species |
Number of isolates |
MIC range (μg/mL) |
|
Susceptible |
Streptococcus canis |
3 |
< 2 to ≤ 2 |
|
Susceptible |
Enterococcus faecalis |
7 |
< 2 to ≤ 2 |
|
Susceptible |
Coagulase-negative Staphylococcus spp. |
1 |
< 0.12 |
|
Susceptible |
S. warneri |
1 |
< 2 |
|
Susceptible |
S. aureus |
1 |
< 2 |
|
Susceptible |
Escherichia coli |
10 |
≤ 2 to 4 |
|
Susceptible |
Staphylococcus pseudintermedius |
5 |
< −0.25 to < 2 |
|
Susceptible |
Proteus spp. |
5 |
< 2 to ≤ 2 |
|
Susceptible |
Bacillus spp. |
2 |
< 0.25 to < 2 |
|
Susceptible |
Staphylococcus epidermidis |
1 |
2 |
|
Resistant |
E. coli |
11 |
32 to > 256 |
|
Resistant |
E. faecalis |
2 |
> 8 |
|
Resistant |
Klebsiella pneumoniae |
2 |
32 to > 256 |
|
Resistant |
S. pseudintermedius |
2 |
16–64 |
|
Resistant |
Enterobacter cloacae |
2 |
> 8 to > 256 |
|
Resistant |
Pseudomonas aeruginosa |
1 |
> 256 |
|
Resistant |
Proteus spp. |
1 |
16 |
|
Discrepant Result 1a |
Proteus spp.a |
1 |
16 |
|
Discrepant Result 1a |
Staphylococcus pseudintermediusa |
1 |
< 2 |
|
Discrepant Result 2 |
E. coli |
1 |
> 256 |
a Discrepant Result 1 had two bacterial isolates: Ampicillin-susceptible S. pseudintermedius and ampicillin-resistant Proteus spp.
Notably, all 36 cultures identified by the gold standard as positive for ampicillin-susceptible organisms (e.g., Streptococcus spp., Staphylococcus pseudintermedius, E. coli) were correctly classified as negative for ampicillin resistance by the broth turbidity test. This further confirms the test’s 100% specificity for detection of ampicillin resistance (zero false positives), even in the presence of uropathogenic bacteria.
The two discrepant results (false negatives) involved samples with low bacterial loads. One sample (Discrepant Result 1) had 1000 CFUs/mL in the gold standard test and contained ampicillin-susceptible S. pseudintermedius and ampicillin-resistant Proteus spp., while the other (Discrepant Result 2) had < 1000 CFUs/mL, grew only in the gold standard’s enrichment broth, and contained ampicillin-resistant E. coli. These discrepant results were included in the analysis to ensure a conservative estimate of the test’s sensitivity. Further investigation revealed that Discrepant Result 1 was from an animal treated with cefovecin 2 days before sample collection, likely resulting in high urinary concentrations of cefovecin at the time of culture [12]. This almost certainly contributed to suppresion of bacterial growth in the broth turbidity test, rendering the result invalid [13]. Discrepant Result 2 involved a Proteus species with an ampicillin MIC of 16 μg/mL on the Sensititre MIC plate, which is within one dilution of the CLSI breakpoint (8 μg/mL). Due to the design of MIC plates (antibiotic concentrations double with each well), a reported MIC of 16 μg/mL actually indicates that the true MIC is > 8 but ≤ 16 μg/mL.
4 Discussion
This study evaluated a novel, low-cost broth turbidity test for detecting ampicillin-resistant lower UTIs in dogs and cats, with the goal of guiding first-line therapy decisions. The test exhibited high specificity (100%) and substantial agreement with the gold standard (κ = 0.94), making it a reliable tool for ruling out ampicillin resistance. Its sensitivity (89.47%) was slightly lower, primarily due to two false negatives in samples with low bacterial loads (≤ 1000 CFUs/mL). These discrepant results were retained in the analysis to provide a conservative estimate of sensitivity, ensuring that the test’s performance is not overestimated. The findings suggest that the test is a promising in-house option for veterinary practitioners, particularly in resource-limited settings where rapid results can inform whether amoxicillin is appropriate or if a formal culture is warranted.
The test’s 100% specificity is a key strength, as demonstrated by its ability to correctly classify all 36 cultures positive for ampicillin-susceptible organisms as negative for resistance. This ensures that practitioners can confidently use amoxicillin in such cases without risking inappropriate treatment escalation. The high positive predictive value in this sample population (100%) further supports its utility in identifying true resistant cases, prompting submission for formal culture and susceptibility testing. However, an important limitation of the broth turbidity test is that it is specifically designed to detect ampicillin-resistant organisms and does not distinguish between antibiotic-susceptible infections and cultures with no bacterial growth. As a result, a negative test result could indicate either the absence of infection or the presence of a susceptible infection, but the test cannot differentiate between these scenarios. Therefore, this test should only be used when the clinician has a high clinical suspicion of a lower UTI that warrants empiric treatment. If the turbidity test on a lower UTI urine sample suggests ampicillin susceptibility, clinicians can treat with confidence using guideline-recommended amoxicillin. If the turbidity test suggests amoxicillin resistance, amoxicillin could still be an effective treatment, but results will be less predictable. In this case, the client could be given the option to opt for a different empirical drug (e.g., potentiated sulfonamide) based on local antibiogram data or submit the urine sample for traditional bacterial culture and AST at a diagnostic lab.
The two false negatives highlight additional limitations. Discrepant Result 1 was likely confounded by recent cefovecin administration, which could have suppressed bacterial growth in the broth turbidity test due to high urinary concentrations of the antibiotic. Cefovecin, a third-generation cephalosporin, has a long half-life in dogs and cats, and it is well documented that antibiotics excreted into urine at the time of bacterial culture can inhibit the growth of organisms [12, 13]. In standard AST protocols, organisms are first isolated and grown in antibiotic-free media, then tested for susceptibility using purified colonies [14]. This approach allows bacteria to undergo exponential growth under ideal conditions before antibiotic exposure. In contrast, the broth turbidity test exposes bacteria to ampicillin without a prior isolation step. So, the bacteria were effectively transferred from cefovecin-containing urine directly to ampicillin-containing MH broth, potentially compounding the inhibitory effects of the antibiotics. These findings suggest that the broth turbidity test might be unreliable in patients recently treated with antibiotics and that clinical history, particularly recent antimicrobial use, should be carefully considered when interpreting results.
Discrepant Result 2 involved a Proteus species with an MIC of 16 μg/mL, just one dilution above the CLSI breakpoint of 8 μg/mL for ampicillin resistance and the concentration of ampicillin in the broth turbidity test. CLSI guidelines state that a margin of error of ±1 dilution is acceptable for MIC testing [14]. Additionally, reported MIC values are the highest value within the dilution range, meaning a reported MIC value of 16 has a true MIC value > 8 but ≤ 16 μg/mL [3]. Variability in the antibiotic content of Kirby–Bauer discs is also well documented, with studies reporting high variability in the true antibiotic content of discs [15]. If the true MIC of the Proteus isolate was closer to 8.1 μg/mL and the disc content was slightly above 10 μg (e.g., 11 μg), this could explain why the organism did not grow in the broth turbidity test, leading to a false negative. This highlights the importance of considering the inherent variability in both MIC testing and disc-based methods when interpreting diagnostic test results near the resistance breakpoint.
The CLSI susceptible breakpoint of 8 μg/mL for ampicillin susceptibility is likely conservative for lower UTIs in companion animals, as amoxicillin (ampicillin is used in vitro to predict amoxicillin susceptibility) achieves very high urinary concentrations—averaging > 200 μg/mL over a typical dosing interval for typical doses in both dogs and cats [16, 17]. In human medicine, amoxicillin has been shown to effectively treat lower UTIs caused by bacterial pathogens with MICs far exceeding this breakpoint, owing to these elevated urinary concentrations [18]. However, these high urinary concentrations are not applicable to upper UTIs, such as pyelonephritis, where tissue penetration is critical. Treatment of upper UTIs should be guided by AST using tissue-specific breakpoints to ensure adequate therapeutic concentrations at the site of infection.
The low prevalence of ampicillin-resistant isolates resulted in a small number of positive cases (n = 19), leading to a wide CI for sensitivity (66.86%–98.72%). Future studies with larger sample sizes or higher resistance prevalence could provide more precise estimates of the test’s performance. Additionally, the test’s reliance on visual turbidity assessment introduces subjectivity; future iterations could incorporate more objective turbidity assessments with a microbiology densitometer or simply by comparing to a set of standardized McFarland turbidity reference tubes to enhance reproducibility. Excluding patients with recent antibiotic exposure could further improve the test’s reliability, as demonstrated by the probable confounding effect of recent cefovecin administration in Discrepant Result 1.
The broth turbidity test’s simplicity—requiring only MH broth, ampicillin discs, sterile tubes, sterile inoculating loops, and a basic incubator—makes it accessible for in-house use, with very low material costs (e.g., under $5 per test). Compact low-cost incubators are widely available for purchase and could be used for other useful purposes in veterinary hospitals. For example, a broth without ampicillin could be inoculated to rule out uro-pathogens in cases such as cystitis in young, otherwise healthy cats. Traditional culture and susceptibility testing often exceed $100 per sample and can easily double the total cost of diagnosing and treating a UTI. In addition to the financial burden of the gold standard test, results typically require 48–72 h, delaying treatment decisions. By providing results within 16–24 h at a very low cost, the broth turbidity test enables practitioners to make timely decisions, potentially improving outcomes while encouraging the use of a first-line antimicrobial, a critical consideration in the context of antimicrobial stewardship [19].
5 Limitations
This study has several limitations. First, the low prevalence of resistant bacteria limited the number of positive cases, affecting the precision of sensitivity estimates. Second, the test’s performance in samples with low bacterial loads or recent antibiotic exposure suggests that modifications (e.g., larger inoculum volumes, exclusion of recently treated patients) might be needed to enhance sensitivity and reliability. Third, the study was conducted at a single diagnostic laboratory, and regional variations in resistance patterns might influence the test’s generalizability. While the broth turbidity test detects ampicillin resistance, it does not provide organism identification or susceptibility profiles for alternative antibiotics, necessitating follow-up testing for positive results. Finally, the study is limited by its in vitro design. Future studies should assess patient outcomes, along with in vitro susceptibility of bacterial isolates, to determine the usefulness of the diagnostic test more accurately.
6 Conclusion
The ampicillin broth turbidity test offers a rapid, low-cost method for detecting ampicillin-resistant lower UTIs in dogs and cats, with high specificity and substantial agreement with gold standard traditional microbiological methods. Its perfect specificity in identifying ampicillin-susceptible infections underscores its reliability in avoiding unnecessary treatment escalation. However, its sensitivity might be affected by low bacterial loads, recent antibiotic administration, or variability in MIC and disc content near the resistance breakpoint. The CLSI breakpoint of 8 μg/mL for ampicillin susceptibility is likely conservative for lower UTIs, given that amoxicillin achieves urinary concentrations greatly exceeding 8 μg/mL. The test’s clinical value could likely be maximized by limiting its use to infections with high CFUs, as identified by urinalysis, and excluding animals with recent antibiotic exposure.
Acknowledgments
The authors acknowledge Sarah Duncan for her technical assistance with sample processing. This work was supported by the National Institutes of Health NIGMS under Award Number U54GM115428.
Disclosure
Authors declare no off-label use of antimicrobials.
Ethics Statement
Authors declare no institutional animal care and use committee or other approval was needed. Authors declare human ethics approval was not needed.
Conflicts of Interest
The authors declare no conflicts of interest.
________________________________________________________________________________________________________________________________
DVM 360 | Managing brachycephalic obstructive airway syndrome
Author(s)Katie Hoddinott, DVM, DVSc, BSc, DACVS-SA
Fact checked by: Kristen Coppock Crossley, MA
September 29, 2025
Surgery can help improve respiratory and gastrointestinal clinical signs of the disease.
Brachycephaly is a breed conformation that consists of short and wide skull proportions.1 The short and wide conformation of the skull results in compression of the nasal and laryngeal airways, which may lead to reduced or absent frontal sinuses, aberrant nasal turbinates, and a reduction in overall airway space caused by a lack of alteration in the local soft tissue structures. The tongue, tonsils, soft palate, and nasopharyngeal mucosa are proportionally larger than the space within which they are housed, reducing the area for airflow through the upper respiratory tract.1
Despite these conformational changes, not all brachycephalic dogs will be affected by brachycephalic obstructive airway syndrome (BOAS). Breeds with more severe brachycephalic conformation, including French bulldogs, pugs, and other bulldog breeds, are at greatest risk.1-3 BOAS is a spectrum of disease characterized by multilevel, partial, or complete obstruction of the upper airway secondary to brachycephalic anatomic conformation and progressive secondary changes to the local anatomy.1,3
The primary anatomic components of BOAS include the following1,3:
Stenotic nares
Elongated and hyperplastic soft palate
Hypoplastic trachea
Aberrant nasal turbinates
The secondary anatomic components of BOAS include the following1,3:
Everted laryngeal saccules (stage 1 laryngeal collapse)
Hiatal hernia
Everted tonsils
BOAS primarily affects the respiratory system, with secondary consequences to the gastrointestinal system.1,3 A less commonly considered consequence of BOAS is sleep-related disorders, particularly obstructive sleep apnea, which can impact overall quality of life.4
Clinical signs of BOAS include stertorous breathing, snoring, stress, heat and exercise intolerance, regurgitation, sleep disturbances, and, in extreme circumstances, respiratory distress, collapse, and cyanosis.1,3 These clinical signs result from the overall reduction in airway diameter because of the brachycephalic conformation. Stenotic nares and aberrant nasal turbinates reduce the size of the nasal passages, making movement of air through the nasal cavity challenging, which often results in open-mouth breathing.1,3 Movement of air through the nasal passages is important for thermoregulation, making these animals prone to hyperthermia.1,3 The elongated and hyperplastic soft palate extends across the rima glottidis, resulting in a reduction in upper airway diameter, thus increasing the negative pressure required for air to pass. The increased negative airway pressures can result in eversion of the laryngeal saccules and tonsils locally but also result in increased negative pressure within the thoracic cavity, leading to gastroesophageal reflux and, in severe circumstances, hiatal hernias.1,3 The hypoplastic tracheal component of this disease may exacerbate the respiratory signs but likely plays a lesser role in the airway obstruction.3
Many dogs exhibit mild signs such as stertor or snoring early in life, which many owners may consider normal for the breed. It is important to begin discussing BOAS early in these animals’ lives, so that owners can better understand the consequences of the disease and monitor for progression of clinical signs. Early intervention when clinical signs are mild is ideal, as the longer the upper airway tissues are exposed to increased upper airway pressures, the more affected they become (ie, further hyperplasia, redundancy, and eversion of local soft tissues), leading to more severe clinical signs.1,3
Diagnostic evaluation for BOAS can vary greatly based on the severity of clinical signs, the disease’s chronicity, and the surgeon’s preferences. Stenotic nares are easily identified on routine physical examination; however, further investigation of the nasal passages for aberrant nasal turbinates requires advanced imaging such as a CT scan or rhinoscopy.1,3 Evaluation of the palate, laryngeal saccules, tonsils, and other nasopharyngeal tissues can be achieved with a sedated oral and laryngeal examination. This can be performed with or without endoscopic equipment. However, greater assessment of soft palate thickness and overall airway diameter can be achieved via CT scan.1,3,5 Thoracic imaging (3-view thoracic radiographs or CT) can rule out aspiration pneumonia because of gastroesophageal reflux/regurgitation and can be used to assess for evidence of a hiatal hernia. Hiatal hernias can be dynamic in nature and therefore may not always be identified on static imaging.1,3
Surgical interventions are typically aimed at the respiratory components of the disease, with the goal of reducing the negative pressures within the upper airway, thus improving air flow.1,3 By reducing the upper airway pressures, the negative intrathoracic pressures may also be improved, resulting in the improvement or resolution of gastrointestinal-related clinical signs. However, if gastrointestinal signs persist, medical management with antacids and promotility agents may be required, or direct surgical intervention for treatment of a hiatal hernia may be warranted.3
Surgical interventions may include any combination of the following procedures1,3:
Rhinoplasty for stenotic nares
Partial staphylectomy for elongated and hyperplastic soft palate
Laryngeal sacculectomy for everted laryngeal saccules
Laser ablation turbinectomy for aberrant nasal turbinates
Tonsillectomy for enlarged and everted tonsils
The individual dog’s anatomic abnormalities will determine the combination of procedures. Surgeon preference and experience will dictate the specific surgical techniques used to address each component of the disease.
Preoperatively, a brachycephalic risk score may be determined to understand the relative risk for a negative outcome following BOAS surgical interventions.6 This score can help set appropriate expectations for owners and inform the veterinary team of the individual risk for each dog undergoing surgical treatment for BOAS. In addition to the specific risk associated with airway surgery, brachycephalic dogs are at increased risk of complications associated with anesthesia.7 Preanesthetic interventions are recommended and often include medications used to reduce the risk of vomiting and regurgitation, as aspiration pneumonia is the most common complication following brachycephalic anesthesia.7,8
In addition to aspiration pneumonia, the risk for postoperative upper airway obstruction following BOAS surgery is significant. There are many ways to reduce the risk of upper airway obstruction, such as administration of injectable steroids, topical application of mannitol in the oropharynx, and a slow and controlled anesthesia recovery. However, being prepared for an emergency temporary tracheostomy is always recommended.3,8
Takeaway
The overall prognosis for dogs following BOAS surgery remains unclear because of the multilevel nature of the airway obstruction with this disease.1,3 Overall, dogs who recover from BOAS surgery should show improvement in respiratory and gastrointestinal clinical signs. If clinical signs persist, further investigations and interventions may be warranted. It is important to note that not all components of BOAS may be addressed with surgical interventions; thus, a return to normal airway function (nonbrachycephalic breeds) is unrealistic.
- REFERENCES
- Ekenstedt KJ, Crosse KR, Risselada M. Canine brachycephaly: anatomy, pathology, genetics and welfare. J Comp Pathol. 2020;176:109-115. doi:10.1016/j.jcpa.2020.02.008
- Tomlinson F, O’Neill E, Liu NC, Sargan DR, Ladlow JF. BOAS in the Boston terrier: a healthier screw-tailed breed? PLoS One. 2024;19(12):e0315411. doi:10.1371/journal.pone.0315411
- Krainer D, Dupré G. Brachycephalic obstructive airway syndrome. Vet Clin North Am Small Anim Pract. 2022;52(3):749-780. doi:10.1016/j.cvsm.2022.01.013
- Hynes JM, Menard JV, Lopez DJ. Quality of life improvement in 3 dogs with sleep-disordered breathing managed by permanent (crico)tracheostomy. Am J Vet Res. 2024;86(2):1-9. doi:10.2460/ajvr.24.09.0270
- Stordalen MB, Bray S, Stringer F, Stonebrook C, Guilherme S, Bray JP. Awake 160-slice computed tomography for upper airway evaluation in 17 dogs. Vet Sci. 2024;11(8):342. doi:10.3390/vetsci11080342
- Tarricone J, Hayes GM, Singh A, Davis G. Development and validation of a brachycephalic risk (BRisk) score to predict the risk of complications in dogs presenting for surgical treatment of brachycephalic obstructive airway syndrome. Vet Surg. 2019;48(7):1253-1261. doi:10.1111/vsu.13291
- Gruenheid M, Aarnes TK, McLoughlin MA, et al. Risk of anesthesia-related complications in brachycephalic dogs. J Am Vet Med Assoc. 2018;253(3):301-306. doi:10.2460/javma.253.3.301
- Hill M, Lepiz ML, Ida KK, Rutter C, Mankin KT, Dickerson V. Implementation of a standard perioperative protocol reduces postoperative respiratory distress events in dogs undergoing surgical correction of brachycephalic obstructive airway syndrome. J Am Vet Med Assoc. 2025;263(5):590-598. doi:10.2460/javma.24.09.0598
https://www.dvm360.com/view/managing-brachycephalic-obstructive-airway-syndrome
________________________________________________________________________________________________________________________________
AVMA Publications – Development and validation of a sandwich ELISA for measurement of angiotensinogen concentration in canine urine and serum
September 26, 2025
Jane H. C. Huang DVM, MSc, Bianca N. Lourenço DVM, PhD, DACVIM and Canaan Whitfield-Cargile DVM, PhD, DACVS
Abstract
Objective
To develop and analytically validate a sandwich ELISA that measures angiotensinogen concentration in canine urine and serum samples.
Methods
Serum (n = 4) and urine (10) samples submitted to a clinical pathology laboratory were used for assay development. Recombinant canine angiotensinogen and both rabbit-derived polyclonal and mouse-derived monoclonal antibodies against it were obtained from a commercial source. A sandwich ELISA was developed and validated via assessment of precision, limit of blank, dilutional linearity, recovery, and cross-reactivity with albumin and angiotensin II.
Results
Intra- and interassay variability ranged from 8.31% to 14.52% and 1.88% to 16.87%, respectively. The limit of blank of the assay was 1.01 ng/mL, which was lower than all observed concentrations in 4 serum (27,130 to 64,690 ng/mL) and 10 urine (3.24 to 8,970.24 ng/mL) samples. Both serum and urine demonstrated ideal dilutional linearity following 2-fold serial dilutions (R2 > 0.99). The assay showed moderate recovery (61.8% to 113.9%). Minimal cross-reactivity was observed with albumin and angiotensin II, as both yielded values below the assay’s limit of blank.
Conclusions
The angiotensinogen ELISA we developed is precise, linear, and sufficiently sensitive for measuring angiotensinogen in the urine and serum of domestic dogs.
Clinical Relevance
Urinary angiotensinogen quantification is a promising noninvasive marker of intrarenal renin-angiotensin system activity in humans and rodents; this assay enables evaluation of its utility in dogs.
Keywords: ELISA; immunoassay; renin-angiotensin-aldosterone system; intrarenal renin-angiotensin system; kidney disease
Inappropriate, maladaptive activation of the renin-angiotensin-aldosterone system is widely implicated in the pathophysiology of kidney diseases and systemic arterial hypertension.1,2 Following the recognition of local, tissue-specific renin-angiotensin systems (RAS), accumulating evidence has identified the intrarenal RAS as a key contributor to the development and progression of various nephropathies and hypertension in people.1,3,4 Importantly, the intrarenal RAS is regulated independently from its circulating counterpart,5,6 and circulating renin-angiotensin-aldosterone system markers do not accurately reflect the status of intrarenal RAS activity.7–9
It is believed that the availability of angiotensinogen serves as a regulatory step for intrarenal RAS activation.7 Accordingly, urinary angiotensinogen has been widely used as a noninvasive biomarker of intrarenal RAS activity in rodents and people,10–15 for whom several studies11,16–21 have documented its positive correlation with renal angiotensin II immunostaining. In humans with acute or chronic kidney disease, the urinary angiotensinogen-to-creatinine ratio is positively correlated with severity of histopathologic renal damage,10,11 is negatively correlated with estimated glomerular filtration rate,12–14 and is predictive of future kidney function deterioration.10,16,22
Recently, sandwich ELISA has been the preferred method for quantifying angiotensinogen in human and rodent urine or plasma and was used in all the above-mentioned studies following the publication and commercialization of species-specific assays in 2007 and 2008, respectively.23,24 Currently, there is a lack of published, validated assays for quantifying angiotensinogen in companion animals. Veterinary researchers have, therefore, historically relied on renal tissue to assess intrarenal RAS activity.25–28 The availability of a urinary angiotensinogen assay would enable noninvasive investigation of intrarenal RAS in kidney diseases and other relevant conditions in veterinary species.
The objective of this study was to develop and analytically validate an ELISA for quantifying canine angiotensinogen. Given the high angiotensinogen concentration in blood (28 to 71 μg/mL) relative to that in urine (7.1 to ∼2,000 ng/mL) reported in humans,10–12,14,16,22,23 and the research interest in comparing circulating and urinary concentrations,1,7 we aimed for the assay to be suitable for both urine and serum samples from dogs.
Methods
Recombinant canine angiotensinogen and anti-canine angiotensinogen antibodies
Recombinant canine angiotensinogen, polyclonal anti-canine angiotensinogen antibody, and monoclonal anti-canine angiotensinogen antibody were purchased from a commercial source (GenScript). The protein sequence of canine angiotensinogen was obtained from the National Center for Biotechnology database (National Center for Biotechnology Reference Sequence: XP_038518489.1). Protein was expressed using the manufacturer’s mammalian cell platform (TurboCHO-Express 2.0) with sequence modification deemed necessary by the manufacturer. The final expressed sequence is as follows:
MGWSCIILFLVATATGVHSDRVYIHPFHLLVYSKSSCEQLEKSNTEMPKDLTFLPVPIQAKTSPVDEEALREELVLVSERLEGEDKMRATKVGMMLNFLGFHMYRMLSESQNAASGTILSPTTLFGTLASFYLGALDPTASRLQAFLGVPGEDQGCTSRLDGHKVLSALQTIQSLLVAQGGASGQSTPLLSTVVGLFTAPGLRLKQPFVRALAPFAPITFPRSLDLSMDPDLAAEKINRFMQAVTGWRMDRPMSGISPDSTLLFNTYVHFQGKLKGLSPLAGLREFWVDNTTSVSVPMLSGTGTFQHWSDPQSNLSMTRVPLGKSACLLLVRPLSTWGLQTVEALTFQHNFPTWVKNLSPRAIRLTMPQLTLRGSYDLQDLLAQAKLPTLLGAEANLGKISDDNVRVGKVLNSILLELQADQEEQPTESAPQPDGPEVLEVTLDHPFLFAIYEQESTALHLLGRVANPLSGVHHHHHH.
The recombinant protein was then purified with AmMag Ni Magnetic beads by the manufacturer.
Purified recombinant protein was used as the antigen to produce rabbit-derived polyclonal antibody and mouse-derived monoclonal antibodies against canine angiotensinogen, both of which were affinity purified by the manufacturer. For the latter, 20 clones of hybridoma were initially produced, and 5 were selected for subcloning based on their supernatant’s reactivity to recombinant canine angiotensinogen, canine serum, and canine urine.
After subcloning, all 5 supernatants underwent purification and were again tested against recombinant canine angiotensinogen, canine serum, canine urine, and native canine albumin (Abbexa; catalog No. abx655874) to confirm the antibodies’ specificity. These purified supernatants were tested using the same sandwich ELISA protocol as described below, except that monoclonal and polyclonal antibodies were both tested as potential detecting or capturing antibodies. Briefly, the candidate supernatant or the rabbit-derived polyclonal antibody was coated on the ELISA plate. Recombinant canine angiotensinogen, serially diluted canine serum, serially diluted canine urine, and native canine albumin were then loaded into the plate. Rabbit-derived polyclonal antibody or the candidate supernatant (whichever that was not used as the coating antibody) was then added. The ideal candidate supernatant would show minimal signal toward canine albumin and a clear signal toward recombinant canine angiotensinogen and canine serum and urine, with reasonable changes in signal strength following dilution of these samples. Based on the testing results, 1 out of 5 mouse-derived monoclonal antibody clones was selected and used alongside the rabbit-derived polyclonal antibody in all experiments presented below.
Sample collection
Residual canine urine and serum samples submitted to the University of Georgia’s Veterinary Clinical Pathology Laboratory for diagnostic purposes were used for assay validation. These samples, obtained from client-owned dogs and otherwise designated for disposal, were stored at 4 °C for up to 7 days before being transferred to our laboratory, aliquoted, and frozen at −80 °C until use. Studies23,24 in rodents and humans showed no significant changes in plasma angiotensinogen concentration after a 1-week incubation at 37 °C. All samples were deidentified, and their use was qualified for exemption from IACUC review.
Sandwich ELISA protocol
A sandwich ELISA was developed using a rabbit-derived polyclonal anti-canine angiotensinogen antibody for capture, a mouse-derived monoclonal anti-canine angiotensinogen antibody for detection, and a horseradish peroxidase–conjugated anti-mouse IgG secondary antibody (Jackson ImmunoResearch; product 115-035-146). All other reagents, including coating buffer, diluents, stop solution, 3,3′,5,5′-tetramethylbenzidine substrate, wash buffer, and 96-well ELISA plates, were obtained from a commercial kit (DuoSet ELISA Ancillary Reagent Kit 2; R&D Systems; catalog No. DY008B). Reagent diluents were prepared by 10-fold dilution of 1% bovine serum albumin in phosphate-buffered saline, per the manufacturer’s instructions. Absorbance was measured at 450 nm with a reference wavelength of 630 nm using a BioTek Epoch Microplate Spectrophotometer and Gen5 software (Agilent). Multiple antibody pairings were tested using recombinant canine angiotensinogen, urine, and serum to determine the optimal configuration, as described in section “Recombinant canine angiotensinogen and anti-canine angiotensinogen antibodies”; the final protocol is described below.
The rabbit-derived polyclonal antibody was diluted 1:2,000 and used to coat 96-well ELISA plates (100 μL per well), which were incubated at 4 °C overnight. Plates were then washed twice and blocked with 150 μL per well of reagent diluent and incubated at 37 °C for 1 hour. After 2 additional washes, 100 μL of standard or sample was loaded into each well, and plates were incubated overnight at 4 °C. Following another 2 washes, mouse-derived anti-canine angiotensinogen monoclonal antibody (1:2,000 dilution) was loaded into each well (100 μL per well) and incubated at 37 °C for 1 hour. After being washed twice, the horseradish peroxidase–conjugated secondary antibody was diluted 1:5,000 per the manufacturer’s instructions, and 100 μL was added to each well. Plates were again incubated at 37 °C for 1 hour. Subsequently, plates were washed 5 times, and 100 μL of 3,3′,5,5′-tetramethylbenzidine substrate was added to each well. Plates were protected from light and incubated at room temperature for 8 minutes. The reaction was stopped with 50 μL of stop solution per well, and absorbance was read immediately. All washing steps were performed manually using 250 μL washing buffer per well, with washing buffer diluted 1:25 in distilled water to working concentration, following the manufacturer’s instructions.
Recombinant canine angiotensinogen was used to establish the standard curve. Standards were run in duplicate for each plate, while samples were run in duplicate, triplicate, or higher replication as required (eg, for intra-assay variability). The concentrations for the standard curve were determined based on optimal performance under a 4-parameter logistic model,29 which was computed in R (version 4.4.1)30 using the drc package (version 3.0-1).31 Model quality was assessed for each plate via adjusted coefficient of determination (R2) and a goodness-of-fit F test that incorporated pure error from duplicate standards.29 All plates achieved an adjusted R2 > 0.98. Fitted 4-parameter logistic model parameters were then used to calculate angiotensinogen concentrations for individual samples.
The established ELISA was examined for its limit of blank and potential cross-reactivity and validated for dilutional linearity, precision (ie, inter- and intra-assay variability), and recovery. For the recovery tests, spiking was performed in diluted serum and urine. Preliminary tests indicated that undiluted serum produced suboptimal signal recovery, consistent with matrix inhibition, as has been reported for other immunoassays.32 Dilution was, therefore, conducted to reduce potential interference from matrix components. Given the assay’s working range and the concentration of angiotensinogen we observed in the canine urine and serum samples tested, dilution of samples is also expected to be required as a standard procedure when using this assay for future diagnostic or research purposes.
Results
Standard curves
Standard curves were generated using recombinant canine angiotensinogen at concentrations of 20, 10, 5, 2.5, 1.25, 0.625, 0.156, and 0 ng/mL (diluent only). Curve performance was assessed via adjusted R2 and goodness-of-fit analyses, as described in the Methods section.29
All standard curves demonstrated excellent fit, with adjusted R2 values exceeding 0.98. Representative curves are shown in Figure 1.
View Full Size
Figure 1
Representative standard curves generated using recombinant canine angiotensinogen (AGT) for the sandwich ELISA quantifying canine AGT. Curves were fitted using a 4-parameter logistic model in R (version 4.4.1)30 with the drc package (version 3.0-1).31 O.D. = Optical density. SS = Sum of squares.
Citation: American Journal of Veterinary Research 2025; 10.2460/ajvr.25.07.0231
Download figure as PowerPoint slide
Limit of blank
The assay’s limit of blank was determined following the Clinical and Laboratory Standards Institute guidelines.33 Specifically, the mean and SD of the measured concentrations from 33 replicates of the blank sample (diluent buffer) were used to calculate the limit of blank, which was 1.095 ng/mL, using the following equation:
meanblank+ 1.645(SDblank)
Linearity
Adequate linearity following serial dilution was observed for both serum and urine samples. Two serum and 2 urine samples from 4 different dogs were initially diluted 1:10,000 (serum) or 1:4 (urine), followed by 2-fold serial dilutions. Observed concentrations were plotted against dilution factors, and linearity was assessed using the average of duplicate measurements for each point. For all 4 samples, the R2 was greater than 0.99 between dilution and observed concentrations (Figure 2).
View Full Size
Figure 2
Linearity following serial dilution of serum (A) and urine (B) samples using the ELISA developed to quantify canine AGT. All dilutions were performed in duplicate, and the mean of each pair of observations was used for analysis. Conc. = Concentration.
Citation: American Journal of Veterinary Research 2025; 10.2460/ajvr.25.07.0231
Download figure as PowerPoint slide
Precision
The inter- and intra-assay variability was evaluated using 3 urine and 3 serum samples from 6 different dogs. All serum samples were diluted at 1:10,000, while urine samples were diluted at 1:4, 1:40, and 1:400, respectively.
Interassay variability was calculated using results from 3 separate assay runs performed on different days. Intra-assay variability was determined from 21 replicates of the same sample within a single plate. Coefficients of variation for intra- and interassay precision ranged from 6.9% to 13.8% and 1.88% to 16.87%, respectively (Table 1).
Table 1
Intra-assay variability of the ELISA quantifying canine angiotensinogen, evaluated using 21 replicates per sample from the urine and serum of 6 different dogs.
|
Variable/sample |
n |
Mean ± SD (ng/mL) |
CV (%) |
|
Intra-assay |
|||
|
Urine 1 (diluted 400 times) |
21 |
5.15 ± 0.4282 |
8.31 |
|
Urine 2 (diluted 40 times) |
21 |
6.41 ± 0.8836 |
13.80 |
|
Urine 3 (diluted 4 times) |
21 |
1.36 ± 0.0935 |
6.90 |
|
Serum 1 |
21 |
6.58 ± 0.9559 |
14.52 |
|
Serum 2 |
21 |
2.28 ± 0.2836 |
12.42 |
|
Serum 3 |
21 |
2.99 ± 0.3784 |
12.66 |
|
Interassay |
|||
|
Urine 1 (diluted 400 times) |
3 |
5.46 ± 0.4087 |
7.49 |
|
Urine 2 (diluted 40 times) |
3 |
6.67 ± 0.6280 |
9.41 |
|
Urine 3 (diluted 4 times) |
3 |
1.33 ± 0.2238 |
16.87 |
|
Serum 1 |
3 |
6.18 ± 0.6026 |
9.75 |
|
Serum 2 |
3 |
2.46 ± 0.2243 |
9.11 |
|
Serum 3 |
3 |
2.93 ± 0.0551 |
1.88 |
Serum samples were diluted 1:10,000, and urine samples were diluted 1:400, 1:40, or 1:4, as indicated.
CV = Coefficient of variation.
Recovery
Recovery was assessed by spiking various concentrations of recombinant canine angiotensinogen into diluted urine and serum samples. Recovery was calculated using the following equation:34
measured concentrationspiked sample−measured concentrationneat sampletheoretical concentrationspiked
Recovery rates ranged from 61.8% to 113.9% (Table 2). Neat samples that aimed at having nondetectable concentrations of angiotensinogen were prepared by diluting 3 urine samples 1:100 and 3 serum samples 1:800,000. Each was spiked with 5% volume of recombinant canine angiotensinogen at 0, 40, 100, or 200 ng/mL, resulting in final theoretical concentrations of 0, 2, 5, and 10 ng/mL, respectively.34 Each condition was evaluated in triplicate, and the mean of the 3 observed values was used to calculate recovery.
Table 2
The ELISA recovery rates of canine angiotensinogen, evaluated in 3 urine samples and 3 serum samples from 6 different dogs.
|
Sample/amount added (ng/mL) |
Observed concentration (ng/mL) |
Recovery (%) |
|
Urine 1 |
||
|
10 |
13.04 |
113.9 |
|
5 |
6.53 |
97.7 |
|
2 |
2.88 |
61.8 |
|
0 |
1.65 |
NA |
|
Urine 2 |
||
|
10 |
11.82 |
111.8 |
|
5 |
4.70 |
81.0 |
|
2 |
1.95 |
65.1 |
|
0 |
0.64 |
NA |
|
Urine 3 |
||
|
10 |
10.93 |
101.4 |
|
5 |
5.60 |
96.3 |
|
2 |
2.18 |
69.5 |
|
0 |
0.79 |
NA |
|
Serum 1 |
||
|
10 |
8.30 |
70.3 |
|
5 |
5.72 |
88.9 |
|
2 |
3.15 |
93.8 |
|
0 |
1.27 |
NA |
|
Serum 2 |
||
|
10 |
10.56 |
96.5 |
|
5 |
6.05 |
102.9 |
|
2 |
2.76 |
92.6 |
|
0 |
0.91 |
NA |
|
Serum 3 |
||
|
10 |
7.94 |
68.9 |
|
5 |
5.97 |
98.4 |
|
2 |
2.75 |
84.4 |
|
0 |
1.06 |
NA |
Recovery was assessed following spiking with known concentrations of recombinant canine angiotensinogen.
NA = Not applicable.
Specificity
To evaluate cross-reactivity, native canine albumin (100 μg/mL) and angiotensin II (100 ng/mL) were tested in quadruplicate. Both yielded signals below the assay’s limit of blank (Table 3), indicating minimal cross-reactivity at physiologically relevant concentrations.
Table 3
Cross-reactivity testing of the ELISA developed to quantify canine angiotensinogen.
|
Angiotensin II (100 ng/mL) |
Albumin (100 μg/mL) |
|
|
Estimated concentration (ng/mL) |
0.5853 |
0.9343 |
|
Ratio to limit of blank (1.095 ng/mL) |
0.535 |
0.853 |
Angiotensinogen concentration in individual dogs
Serum and urine from 4 and 10 dogs, respectively, were tested using the developed ELISA. Serum angiotensinogen concentrations ranged from 27,130 to 64,690 ng/mL, while urine concentrations showed greater variability, ranging from 3.24 to 8,970.24 ng/mL.
Discussion
Measurement of angiotensinogen concentration in blood and urine is common in studies of intrarenal RAS in people.10–14,16,22 In the present study, a sandwich ELISA was successfully developed and analytically validated for quantifying angiotensinogen in canine urine and serum, enabling similar investigations in dogs. The range of angiotensinogen concentrations observed in canine samples submitted to a clinical pathology laboratory for diagnostic purposes is similar to that observed in healthy people23 and in individuals with conditions known to alter intrarenal RAS activity.10–12,14,22 However, it should be noted that our sample size is small, and convenience samples were used. Our study was designed for assay development and validation only and did not aim to establish a reference interval for angiotensinogen concentration in canine serum or urine. For such purposes, a structured sampling approach and a larger sample size would be required.
The assay demonstrated strong linearity, with consistent and reproducible standard curves (adjusted R2 > 0.98) across multiple runs. Intra- and interassay coefficients of variation ranged from 8.31% to 14.52% and 1.88% to 16.87%, respectively, indicating acceptable precision both within and across assay plates. These results support the assay’s reliability for measuring angiotensinogen in both urine and serum from dogs within the tested range of approximately 1.3 to 6.5 ng/mL. Considering the concentration range observed in canine urine and serum samples in this study and with those reported in human samples,10–14,22,23 the assay is well suited for its intended clinical application. Although recovery testing at 10 ng/mL showed acceptable performance, for samples approaching the upper end of the standard curve (8 to 10 ng/mL), further dilution is recommended to minimize variability.
Due to the unavailability of urine and serum that does not contain angiotensinogen, the recovery tests were conducted using diluted samples, which might have weakened any potential matrix effects. It is, therefore, recommended that urine and serum are diluted before their angiotensinogen concentration is determined using this assay. According to our data and previous data from humans,10–14,22,23 dilutions will also likely be needed for both biological fluids to achieve an angiotensinogen concentration within the assay’s working range. Therefore, we consider that our recovery test provides a realistic assessment of assay performance under the applied conditions, which mimic those of its future use.
Our study has several limitations that should be noted. This study utilized a small number of samples (n = 4 for serum; 10 for urine), which limits generalizability to a larger population of animals. Although dilution linearity and recovery were evaluated, other potential matrix effects likely to be found in clinical samples were not evaluated, and the assay’s recovery was only validated in diluted urine and serum but not in undiluted ones. Validation was conducted in a single laboratory using one lot of antibodies and reagents, so interlaboratory reproducibility was not assessed. Ideally, our assay would be validated by comparing our results to a reference method; however, there is currently no reference or validated quantification method for canine angiotensinogen. Formal limits of detection and quantification were not established; however, consistent dilutional linearity and acceptable precision were observed down to approximately 1.35 ng/mL, which we consider the practical lower limit of reliable quantification. Recovery was suboptimal for low-concentration spikes in urine samples (61.8% to 69.5%) and for high-concentration spikes in serum samples (68.9% to 96.5%). The dynamic range of the assay was narrow, but the limit of blank (1.095 ng/mL) was well below all measured values in all samples, suggesting that appropriate dilution can address this limitation.
Finally, urine and serum samples used in this study were stored at 4 °C for up to 1 week before freezing. While this could theoretically affect analyte stability, prior studies23,24 in both rodents and humans have demonstrated that incubation at 37 °C for up to 7 days does not alter the concentration of angiotensinogen in plasma evaluated through ELISA. Thus, it is unlikely that storage at 4 °C for 1 week had impacted the assay results.
In summary, the ELISA developed in this study is sensitive, reproducible, and suitable for quantifying angiotensinogen in canine urine and serum samples. The development and validation of this assay enable future investigation of urinary angiotensinogen as a noninvasive marker of intrarenal RAS activity in dogs. Based on the methods used and the limitations identified, we recommend the assay be applied to diluted urine or serum and within a practical working range of approximately 1.35 to 8 ng/mL; for samples approaching the upper end of this range, additional dilution is advised to minimize variability. Within this range, the assay is well suited for both research and potential clinical applications, provided that preanalytical conditions are consistent with those used in the present study.
Acknowledgments
The authors gratefully acknowledge Mary Byrd, Lab Manager of the University of Georgia’s Veterinary Clinical Pathology Laboratory, for assistance with the accrual of samples.
Disclosures
Dr. Lourenço has received research funding from Ceva Animal Health, Boehringer Ingelheim Vetmedica GmbH, and Elanco Animal Health and speaker honoraria from Boehringer Ingelheim Vetmedica, Ceva Animal Health, IDEXX Ltd, Antech Diagnostics, and Nestlé Purina.
No AI-assisted technologies were used in the composition of this manuscript.
Funding
This study was supported by the authors’ discretionary funds.
ORCID
Jane H. C. Huang https://orcid.org/0009-0001-4528-9245
Bianca N. Lourenço https://orcid.org/0000-0001-5249-4645
Canaan Whitfield-Cargile https://orcid.org/0000-0002-9743-8783
References
1.↑Lin H, Geurts F, Hassler L, et al. Kidney angiotensin in cardiovascular disease: formation and drug targeting. Pharmacol Rev. 2022;74(3):462–505. 10.1124/pharmrev.120.000236
2.↑Brewster UC, Perazella MA. The renin-angiotensin-aldosterone system and the kidney: effects on kidney disease. Am J Med. 2004;116(4):263–272. doi:10.1016/j.amjmed.2003.09.034
3.↑Carey RM, Siragy HM. Newly recognized components of the renin-angiotensin system: potential roles in cardiovascular and renal regulation. Endocr Rev. 2003;24(3):261–271. doi:10.1210/er.2003-0001
4.↑Coffman TM. Under pressure: the search for the essential mechanisms of hypertension. Nat Med. 2011;17(11):1402–1409. doi:10.1038/nm.2541
5.↑Siragy HM, Howell NL, Ragsdale NV, Carey RM. Renal interstitial fluid angiotensin. Hypertension. 1995;25(5):1021–1024. doi:10.1161/01.HYP.25.5.1021
6.↑Navar LG, Lewis L, Hymel A, Braam B, Mitchell KD. Tubular fluid concentrations and kidney contents of angiotensins I and II in anesthetized rats. J Am Soc Nephrol. 1994;5(4):1153–1158. doi:10.1681/ASN.V541153
7.↑Nishiyama A, Kobori H. Independent regulation of renin-angiotensin-aldosterone system in the kidney. Clin Exp Nephrol. 2018;22(6):1231–1239. doi:10.1007/s10157-018-1567-1
8.Li XC, Zhu D, Zheng X, Zhang J, Zhuo JL. Intratubular and intracellular renin-angiotensin system in the kidney: a unifying perspective in blood pressure control. Clin Sci (Lond). 2018;132(13):1383–1401. doi:10.1042/CS20180121
9.↑Huang JH, Lourenço BN, Schmiedt CW, Tarigo JL, Coleman AE. Post hoc comparison of the intrarenal and circulating renin-angiotensin (-aldosterone) systems in cats with ischemia-induced chronic kidney disease. Physiol Rep. 2025;13(12):e70417. doi:10.14814/phy2.70417
10.↑Dou Y, Song D, Wang C, et al. The role of urinary angiotensinogen in kidney interstitial inflammation and renal prognosis. Iran J Kidney Dis. 2019;13(6):372–379.
11.↑Wu L, Yang M, Feng X, et al. Urinary angiotensinogen: an indicator of active antineutrophil cytoplasmic antibody-associated glomerulonephritis. Nephrol Dialysis Trans. 2018;34(5):838–847. doi:10.1093/ndt/gfy112
12.↑Juretzko A, Steinbach A, Hannemann A, et al. Urinary angiotensinogen and renin excretion are associated with chronic kidney disease. Kidney Blood Press Res. 2017;42(1):145–155. doi:10.1159/000474932
13.Kobori H, Ohashi N, Katsurada A, et al. Urinary angiotensinogen as a potential biomarker of severity of chronic kidney diseases. J Am Soc Hypertens. 2008;2(5):349–354. doi:10.1016/j.jash.2008.04.008
14.↑Mills KT, Kobori H, Hamm LL, et al. Increased urinary excretion of angiotensinogen is associated with risk of chronic kidney disease. Nephrol Dial Transplant. 2012;27(8):3176–3181. doi:10.1093/ndt/gfs011
15.↑Gonzalez AA, Visniauskas B, Reverte V, et al. Urinary angiotensinogen displays sexual dimorphism in non-diabetic humans and mice with overweight. Int J Mol Sci. 2024;25(1):635. doi:10.3390/ijms25010635
16.↑Yamamoto T, Nakagawa T, Suzuki H, et al. Urinary angiotensinogen as a marker of intrarenal angiotensin II activity associated with deterioration of renal function in patients with chronic kidney disease. J Am Soc Nephrol. 2007;18(5):1558–1565. doi:10.1681/asn.2006060554
17.Zhang XY, Ding XQ, Lv WL, Teng J, Zhong YH. ELISA examining urinary angiotensinogen as a potential indicator of intrarenal renin-angiotensin system (RAS) activity: a clinical study of 128 chronic kidney disease patients. Mol Biol Rep. 2013;40(10):5817–5824. doi:10.1007/s11033-013-2687-z
18.Nishiyama A, Konishi Y, Ohashi N, et al. Urinary angiotensinogen reflects the activity of intrarenal renin-angiotensin system in patients with IgA nephropathy. Nephrol Dial Transplant. 2011;26(1):170–177. doi:10.1093/ndt/gfq371
19.Urushihara M, Nagai T, Kinoshita Y, et al. Changes in urinary angiotensinogen posttreatment in pediatric IgA nephropathy patients. Pediatr Nephrol. 2015;30(6):975–982. doi:10.1007/s00467-014-3028-8
20.Cao W, Jin L, Zhou Z, et al. Overexpression of intrarenal renin-angiotensin system in human acute tubular necrosis. Kidney Blood Press Res. 2016;41(6):746–756. doi:10.1159/000450564
21.↑Kobori H, Nishiyama A, Harrison-Bernard LM, Navar LG. Urinary angiotensinogen as an indicator of intrarenal angiotensin status in hypertension. Hypertension. 2003;41(1):42–49. doi:10.1161/01.hyp.0000050102.90932.cf
22.↑Jang HR, Kim SM, Lee YJ, et al. The origin and the clinical significance of urinary angiotensinogen in proteinuric IgA nephropathy patients. Ann Med. 2012;44(5):448–457. doi:10.3109/07853890.2011.558518
23.↑Katsurada A, Hagiwara Y, Miyashita K, et al. Novel sandwich ELISA for human angiotensinogen. Am J Physiol Renal Physiol. 2007;293(3):F956–F960. doi:10.1152/ajprenal.00090.2007
24.↑Kobori H, Katsurada A, Miyata K, et al. Determination of plasma and urinary angiotensinogen levels in rodents by newly developed ELISA. Am J Physiol Renal Physiol. 2008;294(5):F1257–F1263. doi:10.1152/ajprenal.00588.2007
25.↑Taugner F, Baatz G, Nobiling R. The renin-angiotensin system in cats with chronic renal failure. J Comp Pathol. 1996;115(3):239–252. doi:10.1016/s0021-9975(96)80082-x
26.Mitani S, Yabuki A, Taniguchi K, Yamato O. Association between the intrarenal renin-angiotensin system and renal injury in chronic kidney disease of dogs and cats. J Vet Med Sci. 2013;75(2):127–133. doi:10.1292/jvms.12-0314
27.Mitani S, Yabuki A, Sawa M, Chang HS, Yamato O. Intrarenal distributions and changes of angiotensin-converting enzyme and angiotensin-converting enzyme 2 in feline and canine chronic kidney disease. J Vet Med Sci. 2014;76(1):45–50. doi:10.1292/jvms.13-0314
28.↑Lourenço BN, Coleman AE, Berghaus RD, Tarigo JL, Schmiedt CW, Brown SA. Characterization of the intrarenal renin-angiotensin system in cats with naturally occurring chronic kidney disease. J Vet Intern Med. 2022;36(2):647–655. doi:10.1111/jvim.16361
29.↑O’Connell M, Belanger B, Haaland P. Calibration and assay development using the four-parameter logistic model. Chemometric Intell Lab Syst. 1993;20(2):97–114. doi:10.1016/0169-7439(93)80008-6
30.↑R: a Language and Environment for Statistical Computing. Version 4.4.1. R Foundation for Statistical Computing; 2024. Accessed August 24, 2025. https://www.R-project.org/
31.↑Ritz C, Baty F, Streibig JC, Gerhard D. Dose-response analysis using R. PLoS One. 2015;10(12):e0146021. doi:10.1371/journal.pone.0146021
32.↑Rosenberg-Hasson Y, Hansmann L, Liedtke M, Herschmann I, Maecker HT. Effects of serum and plasma matrices on multiplex immunoassays. Immunol Res. 2014;58(2):224–233. doi:10.1007/s12026-014-8491-6
33.↑Armbruster DA, Pry T. Limit of blank, limit of detection and limit of quantitation. Clin Biochem Rev. 2008;29(suppl 1):S49–S52.
34.↑Andreasson U, Perret-Liaudet A, van Waalwijk van Doorn LJC, et al. A practical guide to immunoassay method validation. Front Neurol. 2015;6:179. doi:10.3389/fneur.2015.00179
_____________________________________________________________________________________________________________________________________________________
DVM 360 | Managing canine chylothorax
Katie Hoddinott, DVM, DVSc, BSc, DACVS-SA
Fact checked by: Kristen Coppock Crossley, MA
September 5, 2025
There are many methods for surgically managing idiopathic canine chylothorax, but diagnostic information collected from an individual dog will guide the specific approach.
Chylothorax is the accumulation of lymphatic fluid (also known as chyle) within the thoracic cavity.1 Because of the rib cage’s rigid confines, fluid accumulation within the thoracic cavity is not well tolerated and results in respiratory signs, such as increased respiratory rate and effort, after reaching a critical volume. This volume depends on the rate at which it accumulates, the individual dog’s size, respiratory adherence, and overall respiratory function.2
While fluid gradually builds up in the thoracic cavity over time, dogs are often presented on an emergency basis, as the fluid hits the critical volume that results in a respiratory crisis. Once the respiratory system is affected, fluid removal via thoracocentesis is required to improve respiratory function.
To make a chylothorax diagnosis, a diagnostic thoracocentesis is required. This is often performed in conjunction with a therapeutic thoracocentesis to improve the dog’s respiratory function. Thoracic fluid triglycerides will be increased relative to the dog’s serum levels, whereas fluid cholesterol will be reduced relative to the dog’s serum levels.1 It is important that the pleural fluid values are compared to a concurrently collected serum sample and not historical data. Cytological evaluation is important to rule out other sources of pleural effusion, but it can have various cell types present with a chylothorax diagnosis.1
Canine chylothorax is most commonly considered to be idiopathic in nature, but it can also be caused by any disease process that results in a disruption of normal lymphatic flow.1 Primary causes resulting in secondary chylothorax have included cranial mediastinal mass effects, primary cardiac disease, pericardial effusion, trauma, and lung lobe torsions.1-3 When investigating chylothorax, it is important that all possible causes are ruled out before making the presumptive diagnosis of idiopathic chylothorax, as treatments will differ based on the primary cause. When chylothorax is the secondary result of another disease process, the primary disease process must first be addressed before considering direct interventions for treating the chylothorax itself.
Diagnostic evaluation may include thoracic and abdominal imaging to evaluate intrathoracic and intra-abdominal structures, echocardiography to evaluate for primary cardiac disease and pericardial effusion, and a CT lymphangiogram (CTLA) to rule out compressive lesions of the lymphatic and venous systems.1,2,4-7 If advanced imaging is not immediately available, thoracic and abdominal radiography and/or ultrasonography may be used to rule out space-occupying lesions, with CT imaging being reserved for dogs more likely to have idiopathic chylothorax and a desire for surgical interventions.2
Following the diagnosis of idiopathic canine chylothorax, treatment options may include medical or surgical therapies.
Medical management may include the following8:
Intermittent therapeutic thoracocenteses
Low-fat diets
Medications such as octreotide
Therapeutic thoracocenteses are used as needed to remove the buildup of chylous effusion within the thorax to improve clinical respiratory signs. Overall success has been reported in only 26% of animals with idiopathic chylothorax, likely because of the progressive inflammatory changes within the thoracic cavity resulting in difficulty with complete pleural evacuation over time, as well as the development of pleural fibrosis.2,8
Low-fat diets have been used to reduce the overall production of chyle, thus reducing the flow of chyle through the lymphatic system, which may reduce the volume of chylous effusion buildup and allow time for defects in the thoracic duct to heal. Evidence in dogs suggests that the lipid content of chyle can be reduced with this treatment, but the flow through the thoracic duct is not directly reduced.8
Octreotide, a somatostatin analog, has been used to reduce the flow of chyle through the thoracic duct. This has been used with success in human patients who have chylous effusion secondary to trauma to the thoracic duct but has been less successful in dogs with idiopathic chylothorax.8
Surgical interventions may include the following1-3:
Thoracic duct ligation
Subtotal pericardiectomy
Cisterna chyli ablation
PleuralPort placement
Thoracic duct ligation is ideally planned based on CTLA, as this imaging can best identify the anatomic variations of an individual thoracic duct.4-7 The duct can be accessed through open or minimally invasive approaches and is ligated with suture or hemoclips as far caudally in the thoracic cavity as possible to ensure all contributing branches are obstructed. The forward flow of chyle is prevented in the short term while the body develops new lymphaticovenous connections to restore chyle flow.9,10 Intraoperative use of near-infrared fluorescence (NIRF) imaging can be used to confirm complete occlusion of the thoracic duct.11
Subtotal pericardiectomy can also be performed via open or minimally invasive approaches. Removal of the pericardium is aimed at reducing the systemic venous pressure as chyle is being returned to the heart via the cranial vena cava. Increased pressure from primary cardiac disease and/or a constrictive pericardium can result in back pressures within the vena cava and lymphatic extravasation into the thoracic cavity. Determining whether constrictive pericarditis is present can be challenging and invasive;12,13 thus, combining a subtotal pericardiectomy with a thoracic duct ligation has been reported without a definitive diagnosis of constrictive pericarditis.4,10,14-16
Cisterna chyli ablation can be performed through an open or minimally invasive approach. This procedure aims to reduce the forward flow of lymphatic fluid into the thoracic duct by opening the cisterna chyli and allowing lymphatic fluid to drain into the abdominal cavity.9 When combined with thoracic duct ligation, this can reduce the pressure of any forward flow of chyle on the thoracic duct ligation and allow time for the new lymphaticovenous connections to be developed. These new connections may be made within the abdominal cavity, reducing the risk of further chyle leakage within the thoracic cavity.9
PleuralPort placement is a long-term solution for thoracic drainage of chylous (or other) effusion.2,17,18 A fenestrated catheter is surgically placed within a hemithorax and is connected to a drainage port that is placed within the subcutaneous space on the dorsolateral aspect of the ipsilateral hemithorax. The port can be palpated externally and accessed percutaneously with a Huber needle to intermittently drain the thoracic cavity without requiring repeated puncturing of the pleura for thoracocentesis.2,17 Unilateral or bilateral PleuralPorts can be placed based on the volume of effusion and accessibility of the fluid. A PleuralPort is often considered when surgical management has failed, but it can also be considered in place of surgical intervention.
Many combinations of surgical procedures have been reported.3 The most reported combinations include thoracic duct ligation with concurrent subtotal pericardiectomy and thoracic duct ligation with concurrent cisterna chyli ablation.3,4,9,10,12,14-16 A combination of thoracic duct ligation, subtotal pericardiectomy, and cisterna chyli ablation has also been reported in veterinary literature.19
Takeaway
Medical management has a limited success rate for idiopathic chylothorax overall.8 Surgical interventions have a variety of reported success rates, with marked improvement in success being noted with the more recent use of preoperative (CTLA) and intraoperative (NIRF) imaging techniques.3,4,9,10,12,14-16 With these improved imaging techniques, the previously reported need to combine surgical techniques has recently been challenged.12 Although there are many approaches to surgically managing idiopathic canine chylothorax, the specific approach will be made based on the diagnostic information collected from an individual dog, along with the skill set, knowledge, and preference of the surgical care team.
Katie Hoddinott, DVM, DVSc, BSc, DACVS-SA, is a native of Nova Scotia, Canada. She graduated with her DVM from the Atlantic Veterinary College, University of Prince Edward Island, in 2012. She then completed 2 internships and a surgery residency at the Ontario Veterinary College, University of Guelph. She became a diplomat of the American College of Veterinary Surgeons – Small Animal in 2019. Hoddinott is currently working at the Atlantic Veterinary College as an assistant professor in small-animal surgery, where she enjoys teaching undergraduate veterinary students, interns, and residents. Her professional interests lie mainly in surgical oncology and minimally invasive soft tissue surgery. Her current research focuses on advances in clinical teaching for surgery residents and surgical site infections.
REFERENCES
- Hawker W, Singh A. Advances in the treatment of chylothorax. Vet Clin North Am Small Anim Pract. 2024;54(4):707-720. doi:10.1016/j.cvsm.2024.02.006
- Johnston SA, Tobias KM, eds. Veterinary Surgery: Small Animal. 2nd ed. Elsevier; 2018:5468-5542.
- Reeves LA, Anderson KM, Luther JK, Torres BT. Treatment of idiopathic chylothorax in dogs and cats: a systematic review. Vet Surg. 2019;49(1):70-79. doi:10.1111/vsu.13322
- Mayhew PD, Steffey MA, Fransson BA, et al. Long-term outcome of video-assisted thoracoscopic thoracic duct ligation and pericardectomy in dogs with chylothorax: a multi-institutional study of 39 cases. Vet Surg. 2018;48(S1):O112-O120. doi:10.1111/vsu.13113
- Johnson EG, Wisner ER, Kyles A, Koehler C, Marks SL. Computed tomographic lymphography of the thoracic duct by mesenteric lymph node injection. Vet Surg. 2009;38(3):361-367. doi:10.1111/j.1532-950X.2008.00473.x
- Lee N, Won S, Choi M, et al. CT thoracic duct lymphography in cats by popliteal lymph node iohexol injection. Vet RadioL Ultrasound. 2011;53(2):174-180. doi:10.1111/j.1740-8261.2011.01892.x
- Lin LS, Chiu HC, Nishimura R, Fujiwara R, Chung CS. Computed tomographic lymphangiography via intra-metatarsal pad injection is feasible in dogs with chylothorax. Vet Radiol Ultrasound. 2020;61(4):435-443. doi:10.1111/vru.12865
- Singh A, Brisson B, Nykamp S. Idiopathic chylothorax in dogs and cats: nonsurgical and surgical management. Compend Contin Educ Vet. 2012;34(8):E3.
- Sicard GK, Waller KR, McAnulty JF. The effect of cisterna chyli ablation combined with thoracic duct ligation on abdominal lymphatic drainage. Vet Surg. 2005;34(1):64-70. doi:10.1111/j.1532-950x.2005.00012.x
- da Silva CA, Monnet E. Long-term outcome of dogs treated surgically for idiopathic chylothorax: 11 cases (1995-2009). J Am Vet Med Assoc. 2011;239(1):107-113. doi:10.2460/javma.239.1.107
- Steffey MA, Mayhew PD. Use of direct near-infrared fluorescent lymphography for thoracoscopic thoracic duct identification in 15 dogs with chylothorax. Vet Surg. 2018;47(2):267-276. doi:10.1111/vsu.12740
- Mayhew PD, Balsa IM, Stern JA, et al. Resolution, recurrence, and chyle redistribution after thoracic duct ligation with or without pericardiectomy in dogs with naturally occurring idiopathic chylothorax. J Am Vet Med Assoc. 2022;261(5):696-704. doi:10.2460/javma.22.08.0381
- Adams TE, Marvel SJ, Monnet E. Constrictive physiology is not present in all dogs with idiopathic chylothorax. J Am Vet Med Assoc. 2024;262(10):1354-1362. doi:10.2460/javma.24.02.0124
- Fossum TW, Mertens MM, Miller MW, et al. Thoracic duct ligation and pericardectomy for treatment of idiopathic chylothorax. J Vet Intern Med. 2004;18(3):307-310. doi:10.1892/0891-6640(2004)18<307:tdlapf>2.0.co;2
- McAnulty JF. Prospective comparison of cisterna chyli ablation to pericardectomy for treatment of spontaneously occurring idiopathic chylothorax in the dog. Vet Surg. 2011;40(8):926-934. doi:10.1111/j.1532-950X.2011.00902.x
- Allman DA, Radlinsky MG, Ralph AG, Rawlings CA. Thoracoscopic thoracic duct ligation and thoracoscopic pericardectomy for treatment of chylothorax in dogs. Vet Surg. 2010;39(1):21-27. doi:10.1111/j.1532-950X.2009.00623.x
- Bianchi A, Collivignarelli F, Paolini A, et al. Thoracoscopic assisted PleuralPortTM application in seven dogs affected by chronic pleural effusion. Vet Sci. 2023;10(5):324. doi:10.3390/vetsci10050324
- Tirillini F, Montinaro V, Cipolla E, Cinti F. Management of chronic pleural effusion in dogs and cats using the PleuralPort device: complications and outcome of 30 cases (2005-2024). N Z Vet J. 2025;73(4):288-295. doi:10.1080/00480169.2025.2461150
- Ishigaki K, Nagumo T, Sakurai N, Asano K. Triple-combination surgery with thoracic duct ligation, partial pericardiectomy, and cisterna chyli ablation for treatment of canine idiopathic chylothorax. J Vet Med Sci. 2022;84(8):1079-1083. doi:10.1292/jvms.22-0043
_____________________________________________________________________________________________________________________________________________________
DVM 360 News|Articles| September 29, 2025
Managing brachycephalic obstructive airway syndrome
Author(s)Katie Hoddinott, DVM, DVSc, BSc, DACVS-SA
Fact checked by: Kristen Coppock Crossley, MA
Surgery can help improve respiratory and gastrointestinal clinical signs of the disease.
Brachycephaly is a breed conformation that consists of short and wide skull proportions.1 The short and wide conformation of the skull results in compression of the nasal and laryngeal airways, which may lead to reduced or absent frontal sinuses, aberrant nasal turbinates, and a reduction in overall airway space caused by a lack of alteration in the local soft tissue structures. The tongue, tonsils, soft palate, and nasopharyngeal mucosa are proportionally larger than the space within which they are housed, reducing the area for airflow through the upper respiratory tract.1
Despite these conformational changes, not all brachycephalic dogs will be affected by brachycephalic obstructive airway syndrome (BOAS). Breeds with more severe brachycephalic conformation, including French bulldogs, pugs, and other bulldog breeds, are at greatest risk.1-3 BOAS is a spectrum of disease characterized by multilevel, partial, or complete obstruction of the upper airway secondary to brachycephalic anatomic conformation and progressive secondary changes to the local anatomy.1,3
The primary anatomic components of BOAS include the following1,3:
- Stenotic nares
- Elongated and hyperplastic soft palate
- Hypoplastic trachea
- Aberrant nasal turbinates
The secondary anatomic components of BOAS include the following1,3:
- Everted laryngeal saccules (stage 1 laryngeal collapse)
- Hiatal hernia
- Everted tonsils
BOAS primarily affects the respiratory system, with secondary consequences to the gastrointestinal system.1,3 A less commonly considered consequence of BOAS is sleep-related disorders, particularly obstructive sleep apnea, which can impact overall quality of life.4
Clinical signs of BOAS include stertorous breathing, snoring, stress, heat and exercise intolerance, regurgitation, sleep disturbances, and, in extreme circumstances, respiratory distress, collapse, and cyanosis.1,3 These clinical signs result from the overall reduction in airway diameter because of the brachycephalic conformation. Stenotic nares and aberrant nasal turbinates reduce the size of the nasal passages, making movement of air through the nasal cavity challenging, which often results in open-mouth breathing.1,3 Movement of air through the nasal passages is important for thermoregulation, making these animals prone to hyperthermia.1,3 The elongated and hyperplastic soft palate extends across the rima glottidis, resulting in a reduction in upper airway diameter, thus increasing the negative pressure required for air to pass. The increased negative airway pressures can result in eversion of the laryngeal saccules and tonsils locally but also result in increased negative pressure within the thoracic cavity, leading to gastroesophageal reflux and, in severe circumstances, hiatal hernias.1,3 The hypoplastic tracheal component of this disease may exacerbate the respiratory signs but likely plays a lesser role in the airway obstruction.3
Many dogs exhibit mild signs such as stertor or snoring early in life, which many owners may consider normal for the breed. It is important to begin discussing BOAS early in these animals’ lives, so that owners can better understand the consequences of the disease and monitor for progression of clinical signs. Early intervention when clinical signs are mild is ideal, as the longer the upper airway tissues are exposed to increased upper airway pressures, the more affected they become (ie, further hyperplasia, redundancy, and eversion of local soft tissues), leading to more severe clinical signs.1,3
Diagnostic evaluation for BOAS can vary greatly based on the severity of clinical signs, the disease’s chronicity, and the surgeon’s preferences. Stenotic nares are easily identified on routine physical examination; however, further investigation of the nasal passages for aberrant nasal turbinates requires advanced imaging such as a CT scan or rhinoscopy.1,3 Evaluation of the palate, laryngeal saccules, tonsils, and other nasopharyngeal tissues can be achieved with a sedated oral and laryngeal examination. This can be performed with or without endoscopic equipment. However, greater assessment of soft palate thickness and overall airway diameter can be achieved via CT scan.1,3,5 Thoracic imaging (3-view thoracic radiographs or CT) can rule out aspiration pneumonia because of gastroesophageal reflux/regurgitation and can be used to assess for evidence of a hiatal hernia. Hiatal hernias can be dynamic in nature and therefore may not always be identified on static imaging.1,3
Surgical interventions are typically aimed at the respiratory components of the disease, with the goal of reducing the negative pressures within the upper airway, thus improving air flow.1,3 By reducing the upper airway pressures, the negative intrathoracic pressures may also be improved, resulting in the improvement or resolution of gastrointestinal-related clinical signs. However, if gastrointestinal signs persist, medical management with antacids and promotility agents may be required, or direct surgical intervention for treatment of a hiatal hernia may be warranted.3
Surgical interventions may include any combination of the following procedures1,3:
- Rhinoplasty for stenotic nares
- Partial staphylectomy for elongated and hyperplastic soft palate
- Laryngeal sacculectomy for everted laryngeal saccules
- Laser ablation turbinectomy for aberrant nasal turbinates
- Tonsillectomy for enlarged and everted tonsils
The individual dog’s anatomic abnormalities will determine the combination of procedures. Surgeon preference and experience will dictate the specific surgical techniques used to address each component of the disease.
Preoperatively, a brachycephalic risk score may be determined to understand the relative risk for a negative outcome following BOAS surgical interventions.6 This score can help set appropriate expectations for owners and inform the veterinary team of the individual risk for each dog undergoing surgical treatment for BOAS. In addition to the specific risk associated with airway surgery, brachycephalic dogs are at increased risk of complications associated with anesthesia.7 Preanesthetic interventions are recommended and often include medications used to reduce the risk of vomiting and regurgitation, as aspiration pneumonia is the most common complication following brachycephalic anesthesia.7,8
In addition to aspiration pneumonia, the risk for postoperative upper airway obstruction following BOAS surgery is significant. There are many ways to reduce the risk of upper airway obstruction, such as administration of injectable steroids, topical application of mannitol in the oropharynx, and a slow and controlled anesthesia recovery. However, being prepared for an emergency temporary tracheostomy is always recommended.3,8
Takeaway
The overall prognosis for dogs following BOAS surgery remains unclear because of the multilevel nature of the airway obstruction with this disease.1,3 Overall, dogs who recover from BOAS surgery should show improvement in respiratory and gastrointestinal clinical signs. If clinical signs persist, further investigations and interventions may be warranted. It is important to note that not all components of BOAS may be addressed with surgical interventions; thus, a return to normal airway function (nonbrachycephalic breeds) is unrealistic.
REFERENCES
- Ekenstedt KJ, Crosse KR, Risselada M. Canine brachycephaly: anatomy, pathology, genetics and welfare. J Comp Pathol. 2020;176:109-115. doi:10.1016/j.jcpa.2020.02.008
- Tomlinson F, O’Neill E, Liu NC, Sargan DR, Ladlow JF. BOAS in the Boston terrier: a healthier screw-tailed breed? PLoS One. 2024;19(12):e0315411. doi:10.1371/journal.pone.0315411
- Krainer D, Dupré G. Brachycephalic obstructive airway syndrome. Vet Clin North Am Small Anim Pract. 2022;52(3):749-780. doi:10.1016/j.cvsm.2022.01.013
- Hynes JM, Menard JV, Lopez DJ. Quality of life improvement in 3 dogs with sleep-disordered breathing managed by permanent (crico)tracheostomy. Am J Vet Res. 2024;86(2):1-9. doi:10.2460/ajvr.24.09.0270
- Stordalen MB, Bray S, Stringer F, Stonebrook C, Guilherme S, Bray JP. Awake 160-slice computed tomography for upper airway evaluation in 17 dogs. Vet Sci. 2024;11(8):342. doi:10.3390/vetsci11080342
- Tarricone J, Hayes GM, Singh A, Davis G. Development and validation of a brachycephalic risk (BRisk) score to predict the risk of complications in dogs presenting for surgical treatment of brachycephalic obstructive airway syndrome. Vet Surg. 2019;48(7):1253-1261. doi:10.1111/vsu.13291
- Gruenheid M, Aarnes TK, McLoughlin MA, et al. Risk of anesthesia-related complications in brachycephalic dogs. J Am Vet Med Assoc. 2018;253(3):301-306. doi:10.2460/javma.253.3.301
- Hill M, Lepiz ML, Ida KK, Rutter C, Mankin KT, Dickerson V. Implementation of a standard perioperative protocol reduces postoperative respiratory distress events in dogs undergoing surgical correction of brachycephalic obstructive airway syndrome. J Am Vet Med Assoc. 2025;263(5):590-598. doi:10.2460/javma.24.09.0598
_____________________________________________________________________________________________________________________________________________________
TVP – Today’s Veterinary Practice
Emerging One Health Importance: Gastrointestinal Parasites
Reports of Echinococcus multilocularis (fox tapeworm) and Baylisascaris procyonis (raccoon roundworm) are increasing in North America and pose a zoonotic risk.
August 12, 2025 | Issue: September/October 2025
Michelle Evason, BSc, DVM, DACVIM (SAIM), MRCVS
Christian M. Leutenegger, BSc, DrMedVet, PhD, FVH
Abstract
Reports of the tapeworm Echinococcus multilocularis are increasing in the United States and Canada. Although parasite risk appeared to be confined to focal regions of Canada, infection is now being detected in dogs, humans, and wildlife in regions of the United States. Similarly, increased fecal detection of the raccoon roundworm Baylisascaris procyonis has enabled broader description of parasite geographic range. These parasites are emerging One Health concerns (animal, human, and environment), and fecal detection in dogs and cats requires immediate education of pet owners and, in some locations, reporting to public health authorities.
Take-Home Points
- Detection of E multilocularis is increasing in domestic dogs and cats, humans, and wildlife in the United States and Canada.
- Dogs and humans infected with E multilocularis can develop alveolar echinococcosis, which is often fatal.
- Most E multilocularis infections in dogs are detected through molecular fecal examination but not conventional fecal flotation tests. Dogs may or may not have clinical signs of disease.
- Increased fecal detection of B procyonis has provided a broader description of parasite geographic range and clinical features of infection.
- B procyonis infection is typically subclinical in dogs and cats; however, severe neurologic disease has been reported in humans and dogs.
- Both E multilocularis and B procyonis are One Health concerns.
- Dogs and cats serve as sentinels of disease risk for humans.
- Fecal detection of E multilocularis or B procyonis in dogs or cats requires immediate communication with pet owners.
Gastrointestinal (GI) parasites, such as tapeworms and roundworms, are frequently detected in dogs and cats by veterinary teams in day-to-day small animal practice. However, the disease risk to dogs and cats and zoonotic risk to owners vary significantly depending on the species of tapeworm or roundworm identified. For tapeworms like Echinococcus species and for the raccoon roundworm, Baylisascaris procyonis, fecal detection in a dog or cat has significance beyond their typically subclinical effects on the pet’s health status. Detection of these parasites carries significant public health (zoonotic) concern, and prompt, clear communication of disease risk, as well as counseling owners to communicate with human healthcare providers, is necessary.
Recent veterinary, human, and wildlife research has drawn attention to the emergence and range expansion of Echinococcus multilocularis.1-13 These studies, as well as case reports on E multilocularis infections and new data on B procyonis, have One Health implications. Detection of E multilocularis through routine veterinary fecal surveillance has also raised antimicrobial use questions associated with multidrug parasite protection products, warranting consideration of antimicrobial (antiparasitic) and diagnostic stewardship.
Echinococcus multilocularis
Echinococcus species are tapeworms of human, veterinary, and environmental concern in the United States and Canada.1-13 These tapeworms have complex life cycles, with domestic dogs and wild canids (coyotes, foxes, wolves) serving as definitive hosts for E multilocularis and Echinococcus granulosus sensu lato. Cystic echinococcosis can occur in humans, cervids, and livestock after infection with E granulosus sensu lato. Humans, dogs, and rodents infected with E multilocularis are at risk of alveolar echinococcosis.
Infection and Disease
Wild and domestic dogs can become infected with E multilocularis by ingesting a rodent containing a metacestode (larval stage).1-7 Dogs can also infect themselves through the oral route, typically through grooming. Development into the adult stage of the tapeworm occurs within the small intestine, and eggs are shed in the feces.1-6 These eggs are immediately infective and environmentally resistant. Recent studies have reported GI signs (diarrhea) in dogs with enteric E multilocularis; however, infections are thought to be typically subclinical in dogs.1,2,7 A recent report of PCR detection of enteric E multilocularis in a cat described GI signs (diarrhea) and resolution of E multilocularis detection with praziquantel treatment.13
Dogs—and humans—can also act as “dead-end” hosts for E multilocularis after consumption of eggs from contaminated water or food or the environment. In these cases, alveolar echinococcosis, characterized by tumor-like lesions (alveolar hydatid cysts), can develop. Alveolar echinococcosis is typically diagnosed when abdominal imaging shows these cysts within the liver, although they can form in other body locations as well.1-9 Unfortunately, alveolar echinococcosis is often fatal in both dogs and humans if undiagnosed.1-9
While detection of E multilocularis in dogs and humans remains rare, reports are increasing, particularly in regions such as Alberta, Canada, where a relatively high prevalence has been detected in wild dogs.1,2,5,11 One U.S. study in coyotes identified E multilocularis in new areas, including Illinois, Indiana, Kansas, and Missouri.12 Additionally, this study reported detection as ranging between 16% and 47%, indicating the potential for regional “spillover” into pet dogs, cats, and humans.12 Another recent publication reported fecal quantitative PCR (qPCR) results, management, preventive care status, and outcome in 26 pet dogs from Canada and the United States.1 In this research, E multilocularis was detected in dogs from novel regions of the United States, including Colorado, Idaho, Illinois, Kansas, Montana, Nevada, Oregon, Washington, and Wyoming.1
Zoonotic Risk
Zoonotic infection with E multilocularis arises from human consumption of eggs shed in dog feces.1-9 Routine fecal screening in dogs, and potentially in cats, as advised by parasite guidelines from the CAPC and Canadian Parasitology Expert Panel (CPEP), can inform the risk of human E multilocularis infection in known endemic regions and alert to emerging risks.14,15
In veterinary medicine, fecal centrifugal flotation combined with microscopy is often used for fecal screening. However, this test, even when performed by reference laboratories, has low sensitivity for Echinococcus species. Further, eggs of Taenia species cannot be microscopically differentiated from those of Echinococcus species.1-9 The CAPC and CPEP advise use of fecal qPCR testing for dogs living in (or traveling to) E multilocularis endemic regions, for dogs in regions considered high risk, and whenever taeniid eggs are found in dog feces to alert to both dog and human risk.14,15
In some regions, human health guidelines advise a risk assessment be performed for humans in contact with a dog shedding E multilocularis eggs.16
Few reports of E multilocularis detection in cats are available, aside from morphologic descriptions of worms observed in necropsy findings in Saskatchewan and North Dakota from the 1970s.17 E multilocularis prevalence in cats in the United States and Canada is currently unknown. In endemic regions in Europe, where prevalence rates in foxes are 35% (France), 48.2% (Poland), and 53% (Switzerland), detection of E multilocularis in cats has been between 7% and 9.3%, 6%, and < 1%, respectively.18-20 A recently published report of E multilocularis detection in a cat through fecal qPCR testing provides much-needed outcome information on resolution of detection after praziquantel treatment and serves to highlight the infection risk for cats with outdoor access or hunting behaviors as well as a reminder of the zoonotic risk potential in felines.13
Public Health, Antimicrobial Use, and Stewardship Considerations
If a dog or cat tests positive for E multilocularis, the veterinary team should immediately inform the owners of zoonotic risk potential and advise them to promptly connect with their healthcare provider to decide on next diagnostic or medical decisions. In some U.S. states and Canadian provinces, detection is also reportable to public health authorities. Other dogs, and potentially cats, in the household of an infected animal should also be tested and treated, along with the affected pet, with praziquantel.1,14-16
The commercial availability of multidrug parasite preventives has raised questions surrounding antimicrobial (antiparasitic) and diagnostic stewardship. While tapeworm resistance is an evolving concern for Dipylidium caninum, it is not known if this will be a concern for Echinococcus species. As such, recommendations surrounding screening and prevention (i.e., test and then treat, treat every 3 months, or treat monthly) are not yet clear, and further research is indicated.
Baylisascaris procyonis
B procyonis is highly prevalent in raccoons, and humans and dogs may become infected after ingestion of eggs. In the United States and Canada, the parasite is typically found in dogs incidentally during fecal screening and may reflect either transient egg shedding (due to coprophagy) or true enteric infection.20,21 GI infection with B procyonis is not thought to cause clinical disease in dogs or cats. However, rare case reports of neural larva migrans have been described in infected dogs, and neurologic disease, in some cases fatal, has been reported in humans.22,23
Veterinary clinics that use fecal flotation and microscopy as the sole method of screening in dogs and cats are at risk of misidentifying B procyonis as Toxocara canis or Toxocara cati. These two roundworms can be differentiated through fecal qPCR testing or by very careful microscopic review (e.g., slight size and color difference in eggs and outer shell).
Zoonotic Risk
High human B procyonis seroprevalence was described in a study from California, indicating exposure risk for people and dogs from common environmental sources (e.g., raccoon latrines).23 Recent fecal qPCR surveillance in dogs and cats has detected B procyonis in multiple regions of the United States and Canada.21
B procyonis eggs observed on microscopy in dogs or cats living in or travelling from endemic regions must be promptly differentiated from T canis and T cati eggs. Additionally, animals infected with B procyonis should be immediately treated with routine dewormers due to zoonotic risk, and testing other household pets should be considered.15,20,23,24
As with E multilocularis, fecal detection of raccoon roundworm in a dog or cat should be immediately communicated to the owners so they may promptly contact their healthcare provider. Owners should be made aware of the risk for zoonotic infectious disease and counseled to contact their healthcare provider for potential consideration of prophylactic therapy.15,20-25 They should also be educated about how to reduce exposure to raccoons (e.g., removal of raccoon latrines) and to promptly dispose of dog and cat feces.
Summary
Routine veterinary fecal screening in dogs and cats will be a critical component of ongoing One Health surveillance for emergence of E multilocularis, as well as for raising awareness of dog and human disease risk associated with both B procyonis and Echinococcus species. Further interdisciplinary efforts and collaboration between wildlife, human, and veterinary researchers is indicated to assist in provision of information that benefits pet and human health.
References
- Evason MD, Peregrine AS, Jenkins EJ, et al. Emerging Echinococcus tapeworms: fecal PCR detection of Echinococcus multilocularis in 26 dogs from the United States and Canada (2022-2024). JAVMA. 2024;263(2):1-5. doi:10.2460/javma.24.07.0471
- Kolapo TU, Hay A, Gesy KM, et al. Canine alveolar echinococcosis: an emerging and costly introduced problem in North America. Transbound Emerg Dis. 2023;2023:5224160. doi:10.1155/2023/5224160
- Williams LBA, Walzthoni N. Diagnosis, treatment, and outcome of four dogs with alveolar echinococcosis in the northwestern United States. JAVMA. 2023;261(7):1-6. doi:10.2460/javma.22.12.0540
- Kotwa JD, Greer T, Jardine CM, et al. Evaluation of the prevalence of Echinococcus multilocularis in dogs that visit off-leash dog parks in southern Ontario, Canada. Zoonoses Public Health. 2021;68(5):533-537. doi:10.1111/zph.12792
- Toews E, Musiani M, Smith A, Checkley S, Visscher D, Massolo A. Risk factors for Echinococcus multilocularis intestinal infections in owned domestic dogs in a North American metropolis (Calgary, Alberta). Sci Rep. 2024;14(1):5066. doi:10.1038/s41598-024-55515-6
- Peregrine AS, Jenkins EJ, Barnes B, et al. Alveolar hydatid disease (Echinococcus multilocularis) in the liver of a Canadian dog in British Columbia, a newly endemic region. Can Vet J. 2012;53(8):870-874.
- Evason MD, Jenkins EJ, Kolapo TU, Mitchell KD, Leutenegger CM, Peregrine AS. Novel molecular diagnostic (PCR) diagnosis and outcome of intestinal Echinococcus multilocularis in a dog from western Canada. JAVMA. 2023;261(9):1-3. doi:10.2460/javma.23.03.0179
- Joyce J, He XO, Rozovsky K, Stefanovici C, Fanella S. Disseminated Echinococcus multilocularis infection without liver involvement in child, Canada, 2018. Emerg Infect Dis. 2020;26(8):1856-1859. doi:10.3201/eid2608.191644
- Zhang N, Vuppala NK, Boney CP, et al. Primary pulmonary echinococcosis in the United States: a case report and review of the literature. Cureus. 2024;16(3):e55591. doi:10.7759/cureus.55591
- Polish LB, O’Connell EM, Barth TFE, et al. European haplotype of Echinococcus multilocularis in the United States. N Engl J Med. 2022;387(20):1902-1904. doi:10.1056/NEJMc2210000
- Catalano S, Lejeune M, Liccoli S, et al. Echinococcus multilocularis in urban coyotes, Alberta, Canada. Emerg Infect Dis. 2012;18(10):1625-1628. doi:10.3201/eid.1810.120119
- Miller K, Raya B, Kollasch T, Ryan W, Herrin B. Detection of Echinococcus spp. in coyotes in the Midwestern United States. Presented at: American Association of Veterinary Parasitologists 68th Annual Meeting; June 10-13, 2023; Lexington, Kentucky.
- Langs Rund L, Evason MD, Weese JS, Leutenegger CM. Echinococcus multilocularis in a cat: novel report of molecular detection (quantitative polymerase chain reaction) and management in a domestic cat from Canada. Am J Vet Res. 2025;86(7):ajvr.25.02.0072. doi:10.2460/ajvr.25.02.0072
- Echinococcus spp. Companion Animal Parasite Council. Updated February 7, 2025. Accessed May 2024. https://capcvet.org/guidelines/echinococcus-spp
- Conboy G, Prada CF, Gilleard J, et al. Canadian Parasitology Expert Panel guidelines for the management of parasites in dogs and cats. Accessed May 2024. https://research-groups.usask.ca/cpep/index.php
- Ontario Ministry of Health. Management of Echinococcus multilocularis infections in animals guideline, 2025. Accessed June 10, 2025. https://www.ontario.ca/files/2024-12/moh-guidelines-management-of-echinococcus-multilocularis-infections-animals-en-2025-01-02.pdf
- Massolo A, Liccoli S, Budke C, Klein C. Echinococcus multilocularis in North America: the great unknown. Parasite. 2014;21:73. doi:10.1051/parasite/2014069
- Karamon J, Sroka J, Dabrowska J, et al. First report of Echinococcus multilocularis in cats in Poland: a monitoring study in cats and dogs from a rural area and animal shelter in a highly endemic region. Parasit Vectors. 2019;12(1):313. doi:10.1186/s13071-019-3573-x
- Furtado Jost R, Müller N, Marreros N, et al. What is the role of Swiss domestic cats in environmental contamination with Echinococcus multilocularis eggs? Parasit Vectors. 2023;16(1):353. doi:10.1186/s13071-023-05983-y
- French SK, Pearl DL, Peregrine AS, Jardine CM. Baylisascaris procyonis infection in raccoons: a review of demographic and environmental factors influencing parasite carriage. Vet Parasitol Reg Stud Reports. 2019;16:100275. doi:10.1016/j.vprsr.2019.100275
- Teng E, Castro JP, Evason M, et al. Baylisascaris procyonis (raccoon roundworm) detection in dogs and cats from the United States and Canada. Poster presented at: ACVIM Forum 2025; June 19-21, 2025: Louisville, Kentucky.
- Lipton BA, Oltean HN, Capron RB, et al. Baylisascaris procyonis roundworm infection in child with autism spectrum disorder, Washington, USA, 2022. Emerg Infect Dis. 2023;29(6):1232-1235. doi:10.3201/eid2906.230290
- Weinstein SB, Lake CM, Chastain HM, et al. Seroprevalence of Baylisascaris procyonis infection among humans, Santa Barbara County, California, USA, 2014-2016. Emerg Infect Dis. 2017;23(8):1397-1399. doi:10.3201/eid2308.170222
- Baylisascaris procyonis. Companion Animal Parasite Council. Updated March 27, 2023. Accessed May 2024. https://capcvet.org/guidelines/baylisascaris-procyonis
- Davidson RK, Øines Ø, Hamnes IS, Schulze JE. Illegal wildlife imports more than just animals—Baylisascaris procyonis in raccoons (Procyon lotor) in Norway. J Wildl Dis. 2013;49(4):986-990. doi:10.7589/2012-06-154
______________________________________________________________________________________________________________________________________________________________
Frontiers Vet. Sci., 22 April 2025 / Sec. Veterinary Imaging
Orthopedic applications of 3D printing in canine veterinary medicine
Objective: This case series investigates the application of 3D printing in veterinary orthopedic surgeries, emphasizing its potential to enhance preoperative planning, intraoperative precision, and postoperative outcomes.
Animals: Three canines—German Shepherd, Basset Hound, and Labrador Retriever—were included in this study.
Materials and methods: Three canine cases involving complex orthopedic deformities were selected to illustrate different uses of 3D printing in veterinary surgery. CT scans were segmented using Materialise Mimics 26.0, followed by virtual surgical planning and creation of 3D printed models and guides.
Results: In Case 1, a 2-year-old German Shepherd with a congenital right tibial deformity underwent successful surgical correction aided by a preoperatively prepared external fixator frame, saving approximately 1 h of OR time. In Case 2, a 1-year-old Basset Hound with a left antebrachial deformity had a double wedge osteotomy performed with the assistance of patient-specific cutting and reconstruction guides, leading to optimal alignment and reduced surgical time. Case 3 involved a young, less than 1-year-old Labrador Retriever rescue with severe bilateral tibiofemoral deformity, where 3D printed models helped the surgeon determine that surgery was not the best option, potentially preventing a poor outcome.
Clinical relevance: This case series highlights the transformative potential of 3D printing in veterinary orthopedic surgery, illustrating its ability to improve aid surgical outcomes, reduce operative times, and be a valuable tool in preoperative decision-making. This technology allows for tailored surgical interventions, enhancing the precision and effectiveness of treatment plans in veterinary medicine.
Introduction
Additive manufacturing, commonly referred to as 3D printing, is the process by which virtual digital files can be manifested as physical objects. Within medicine, advanced volumetric imaging, such as computed tomography (CT) and magnetic resonance imaging (MRI), can be virtually translated from 2D sliced images to reconstruct 3D objects. These 3D objects can then be 3D printed in materials of varying qualities using additive manufacturing technologies.
Commonly printed models include physical representations of complex anatomy utilized for presurgical planning (1–4), anatomical education (5–9), and simulation (9–13). With the development of 3D printing materials that can be sterilized, more intraoperative models are being produced and utilized such as cutting and reconstruction guides to aid precise osteotomies and hardware placement (5, 9, 11). These techniques are already heavily adopted for human medical care, particularly in complex or unique cases, but there is still limited literature supporting their use in veterinary medicine. Veterinary medicine stands to benefit greatly from 3D printing through enhanced understanding of animal pathology for veterinary surgeons, trainees, and animal owners.
Historically, the anatomical heterogeneity across diverse animal species has posed challenges in the field of veterinary orthopedics. The advent of medical 3D printing has provided means to navigate these challenges by enabling the translation of high-fidelity medical imaging datasets into tangible 3D anatomic models. Advancements in veterinary medicine have witnessed the beginning stages of integration of 3D printing technologies, catalyzing transformative changes in the approach to a limited number of complex orthopedic cases (10–13).
A systematic review of veterinary orthopedic literature by Memarian et al. found benefits of 3D printing technologies in education, preoperative planning, client communication, custom-made orthopedic implants (total joint replacement—hip, knee, patellar groove, and elbow), limb-sparing surgery, corrective osteotomies, arthrodesis, as well as customized scaffolds for forays into 3D bioprinting (11). Kamishina et al. demonstrated clinical benefits of using patient-specific titanium plates in 83.3% (15/18) of spinal stabilization surgeries with most benefit being gained from accurate placement of screws and minimization of complications during technically demanding parts of surgery (12). Fracka et al. found that patient-specific guides had the potential to improve the accuracy of tibial and femoral cut alignment in canine total knee replacement (TKR), especially in cases where surgeons had limited prior experience with TKR surgery (13). Altwal et al. highlighted presurgical modeling as a major emerging use of 3D printing in small-animal surgery, supporting the need for case-based evidence such as we will present here (1).
This case series will examine three distinct applications of 3D printing in veterinary orthopedics. We emphasize the creation of detailed anatomic models and their role in guiding orthopedic surgeries with heightened precision. We offer an example of the diagnostic potential of 3D printing as a preoperative decision-making tool, assessing its capacity to potentially prevent unnecessary surgeries and enhance overall patient care.
Materials and methods
Case selection
Three canines—German Shepherd, Basset Hound, and Labrador Retriever—were included in this study. These cases were selected from our recent clinical caseload to highlight a range of applications where 3D printing was utilized. Owner consent was obtained for all three canines in this case series.
Volumetric CT images were obtained at a slice thickness of 1 mm or thinner to optimize accuracy. To begin, the Digital Imaging and Communications in Medicine (DICOM) data from these scans was anonymized and uploaded into Materialise Mimics 26.0 segmentation software (Leuven, Belgium) or DICOM to PRINT® (D2P) (3D Systems, Inc. Rock Hill, SC), both of which are FDA-cleared—a necessity given that 3D anatomical models are a class II medical device and require regulatory clearance. Once in the software, specific anatomy was isolated by selecting Hounsfield unit (HU, a measure of radiodensity) ranges. Soft tissues tended to be in the range of 40–50 HU, bone >1,000 HU, and fat −100 to −155 HU. Each CT slice was manually visualized to ensure that anatomic integrity and accuracy were maintained and serve the purpose desired for the eventual 3D model. Once the segmentation was complete, stereolithography (.STL) files were exported and overlaid on the original scan to verify the accuracy of the contours against the imaging data.
Computer aided design (CAD)
When necessary, the. STL file was exported to Materialise 3-matic 13.0 (Leuven, Belgium) CAD software program to manipulate the files such as creating magnet holes for part retention, smoothing or hollowing the models, adding support structures to allow for accurate printing, and creating surgical guides that are shaped to the specific anatomy in an individual case. For these design tasks, we created parts using primitive shapes that were then morphed to the contours of the anatomy they were correcting. Boolean operations of addition and subtraction, which allow separate parts to become a union or to ensure there is no overlap, were heavily utilized to perform these functions. In cases where magnet holes were necessary, a tolerance of ± 0.2% of the hole size was incorporated to account for inter-print variability in accuracy.
Surgical planning
These 3D models were uploaded to Materialise Viewer (Leuven, Belgium), an online platform used to view and mark. STL files, and shared with the veterinary surgeons for surgical planning. In this software, the veterinarian could mark which planes and angles were desired for cutting, which pieces of anatomy they would theoretically want to manipulate during surgery, and, most importantly, it served as an easy point of collaboration that could be easily referenced across geographies.
3D printing
Once anatomy and/or cutting guides were finalized and agreed upon,.STL files were uploaded to Formlabs 3B printer’s proprietary software PreForm v3.21.0 (Formlabs Inc. Somerville, MA). Within this software, support structures were automatically added and ideal printing orientations were algorithmically chosen by the software to optimize print quality and shorter printing times. All 3D models were printed using a Formlabs Form 3B desktop 3D printer (Formlabs Inc. Somerville, MA), which utilizes stereolithography (SLA) printing with resin. These SLA resin 3D printers used a laser to cure liquid resin into hardened plastic in a process called photopolymerization. Osseous anatomic models were printed using Formlabs White resin and intraoperative guides were printed using Formlabs Biocompatible Surgical Guide resin. Models and guides were provided to the veterinarian surgeons 1–2 days prior to surgery to allow for adequate preparation.
Case 1
A 2-year-old German Shepherd with congenital right tibial deformity was undergoing surgical correction and the orthopedic veterinarian consulted for an anatomic model. Segmentation isolated the right tibia and fibula and took approximately 0.5 h to segment, 13 h to print, and used 32 mL of white resin.
The veterinarian used the anatomic model for surgical planning and they were able to prepare the complex external frame (Imex external fixator, Longview, TX) ahead of time (Figure 1a) and achieve satisfactory alignment (Figure 1b). This saved approximately 1 h of OR time compared to the veterinarian’s historic average for similar cases. This canine is doing well 12 months after surgery despite the lack of bony union and bone plating after external fixator removal.
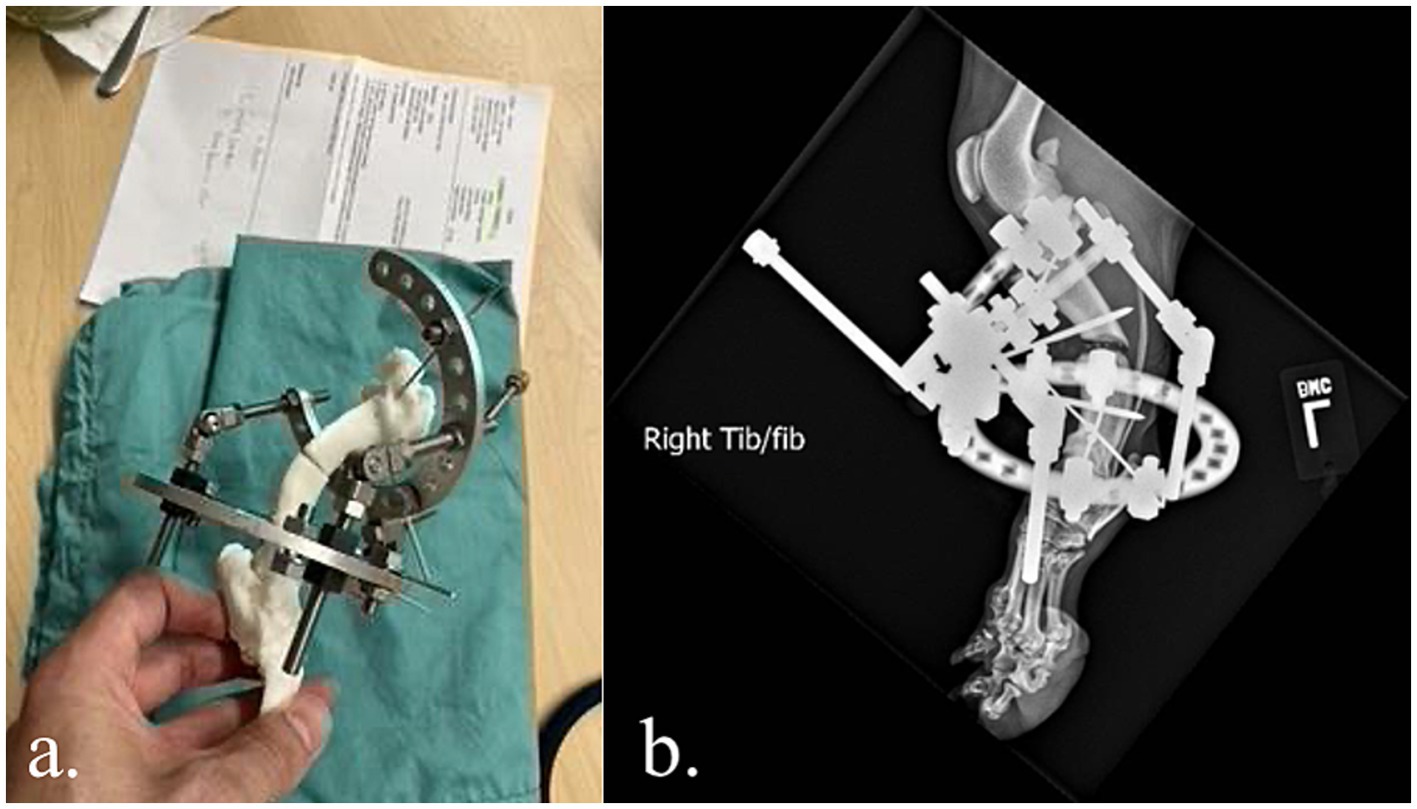
Case 2
A 1-year-old Basset Hound with left antebrachial deformity was undergoing surgical correction. The orthopedic veterinarian requested virtual surgical planning, anatomic models, and surgical guides for intraoperative pin placement to correctly approximate bone segments. Initially, the forelimbs were segmented, which took approximately 0.5 h. Virtual surgical planning was performed with the veterinarian surgeon for double wedge osteotomies with desired locations, orientations, and width of the wedges. The bone segments were then aligned with the feedback of the veterinarian surgeon to optimally correct for the curvature and rotation (Figure 2).
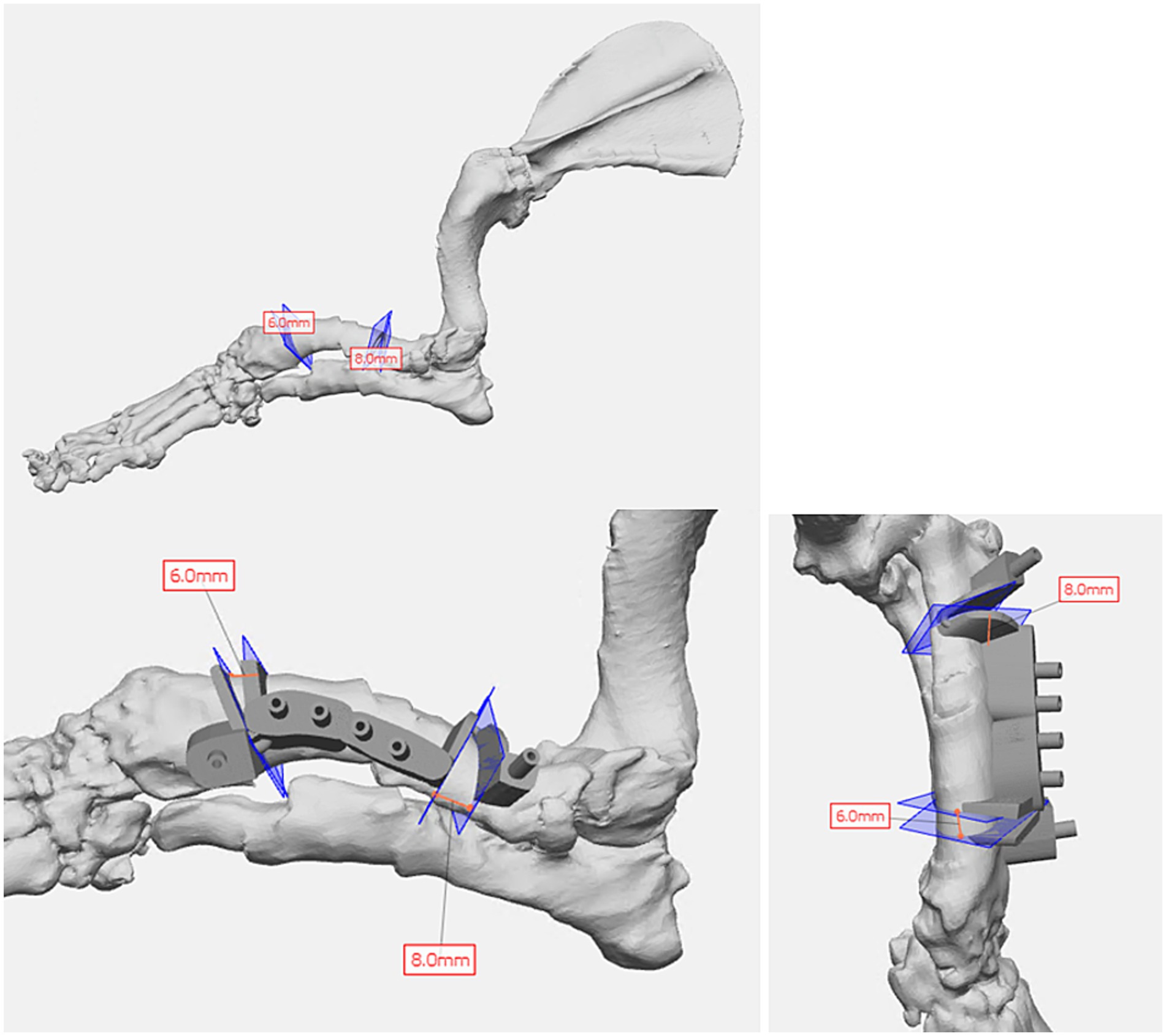
Once the plan was finalized, a single piece cutting guide was designed for the planned double wedge osteotomy with pin holders according to the anticipated tissue exposure as well as the need for the fixation pins to be placed laterally (Figure 2). A reconstruction guide was designed to align the bone segments in the planned positions using the same fixation pins (Figure 3). The cutting guide and reconstructed model with the reconstruction guide were printed in biocompatible surgical guide FormLabs resin (Figures 4a,b). The reconstructed anatomy took 11 h to print and used 88 mL of white resin. The cutting guide took 5.5 h to print and used 49 mL of biocompatible surgical resin. These models were printed concurrently on separate printers. The models and guides were sterilized for intraoperative use with the placement of the cutting guide to execute the double wedge osteotomies. After the osteotomies, the reconstruction guide was applied to align the bone segments as planned for plate fixation (Figure 5). Proper alignment was achieved and the severe curvature of the left forelimb was corrected (Figure 6).
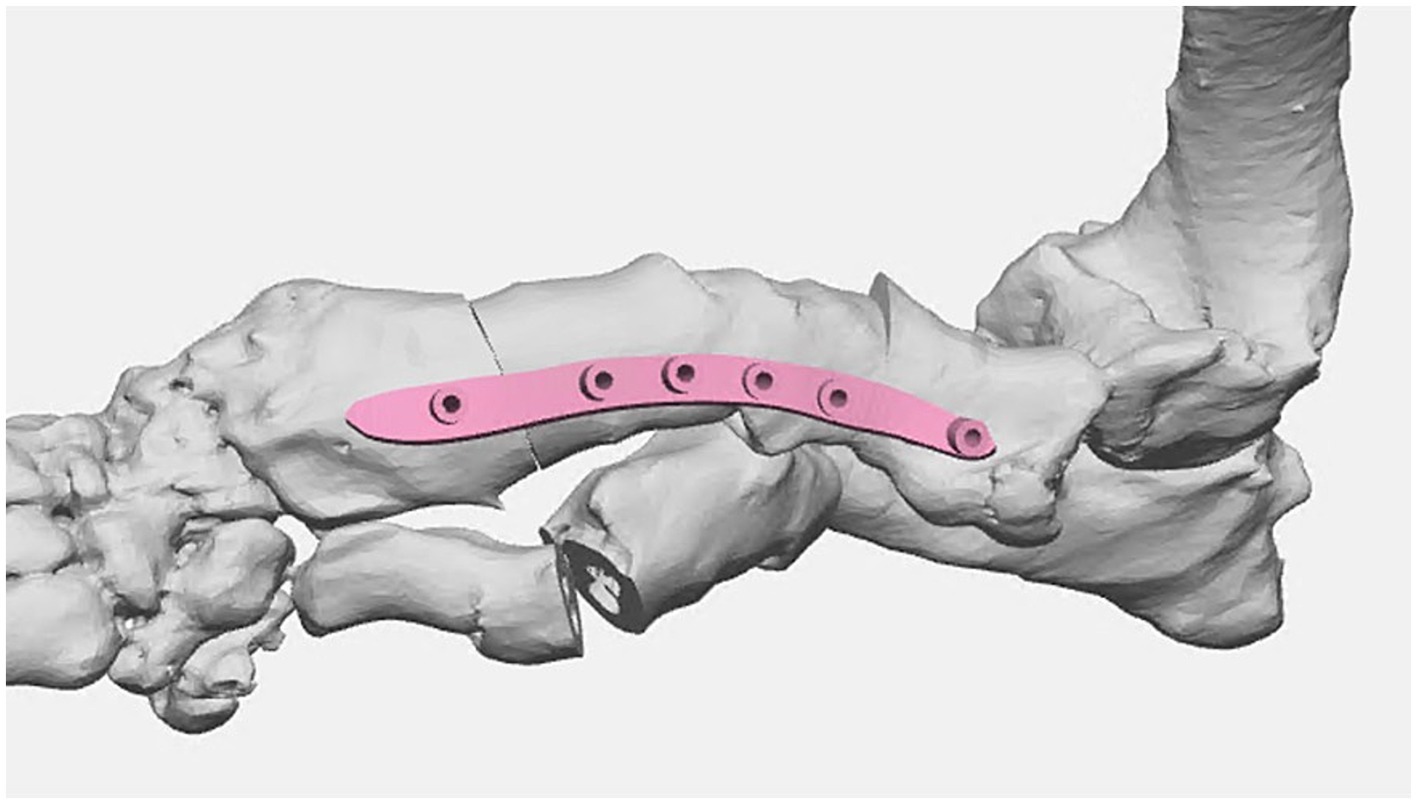
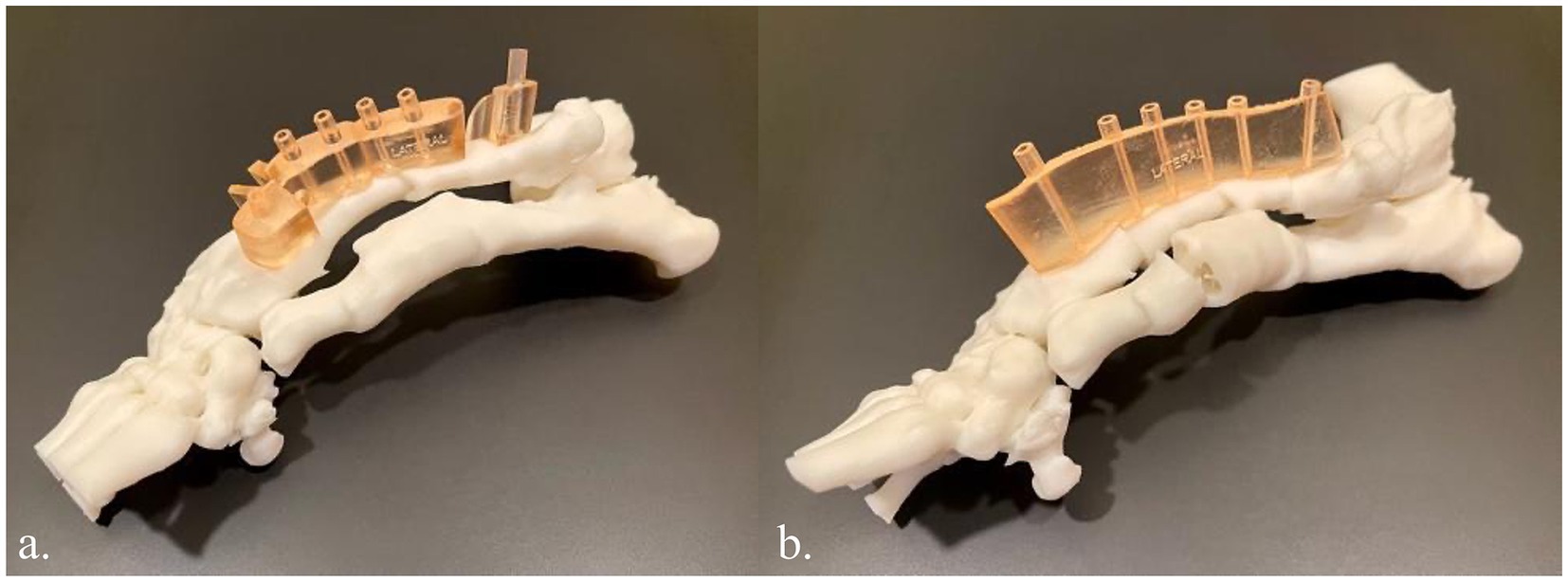
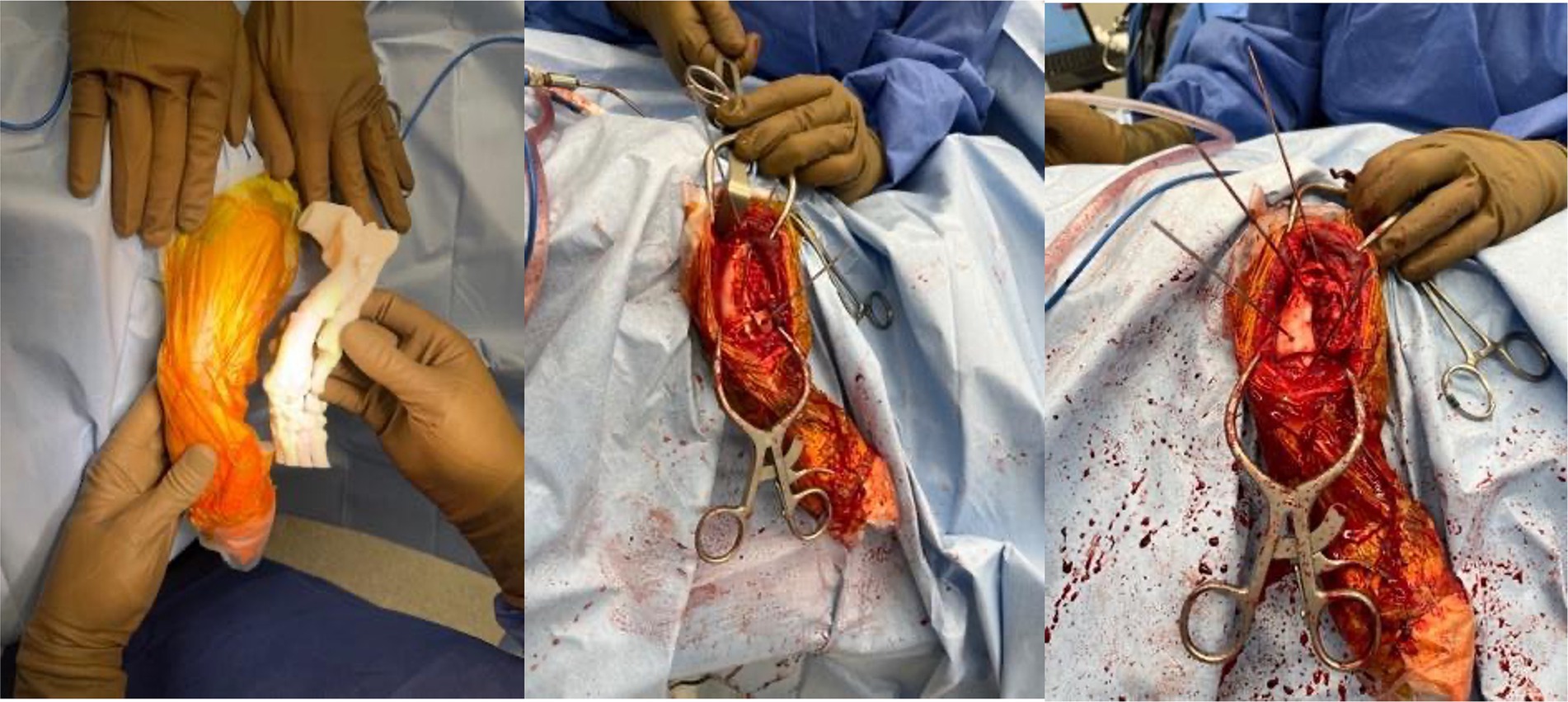
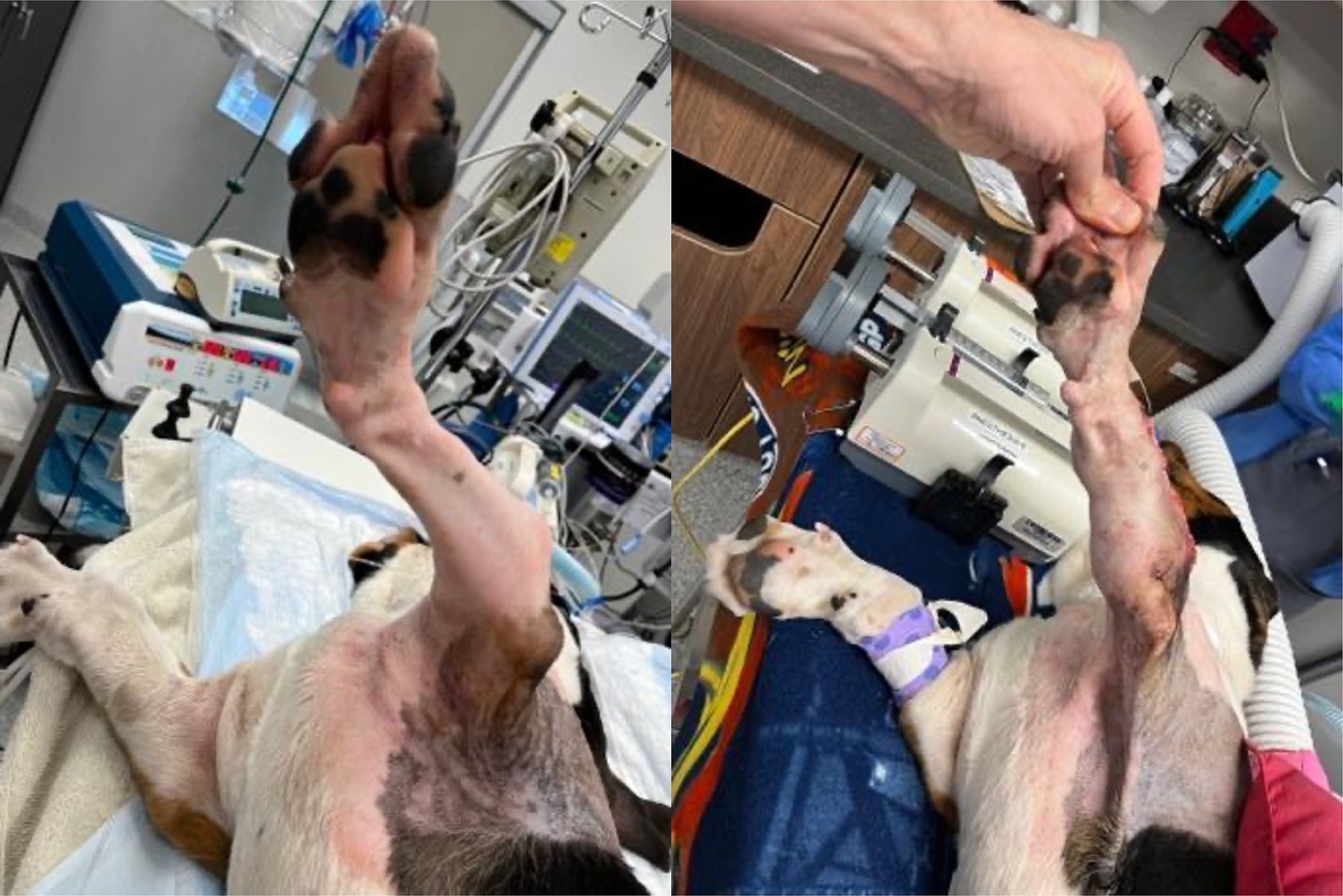
The surgeon reported that the models and guides saved him over 1 h of surgery time and ensured optimal surgical correction and outcome. He also remarked how he was able to use anatomic landmarks on the model to guide him during surgery where landmarks became obscured by tissue and blood. This canine is doing well over 12 months after surgery.
Case 3
A young, less than 1-year-old, Labrador Retriever rescue with severe bilateral hindlimb deformity was being evaluated for possible surgical correction and an anatomic model was requested. The volume rendering of his hindlimbs from his CT demonstrated severe malalignment of his tibiofemoral joints bilaterally (Figure 7). The hindlimbs were segmented and modified for magnetic retention to maintain the native anatomic orientation. The 3D model assisted the surgeon in determining that surgical correction was not the optimal treatment plan and could possibly result in a worse outcome.
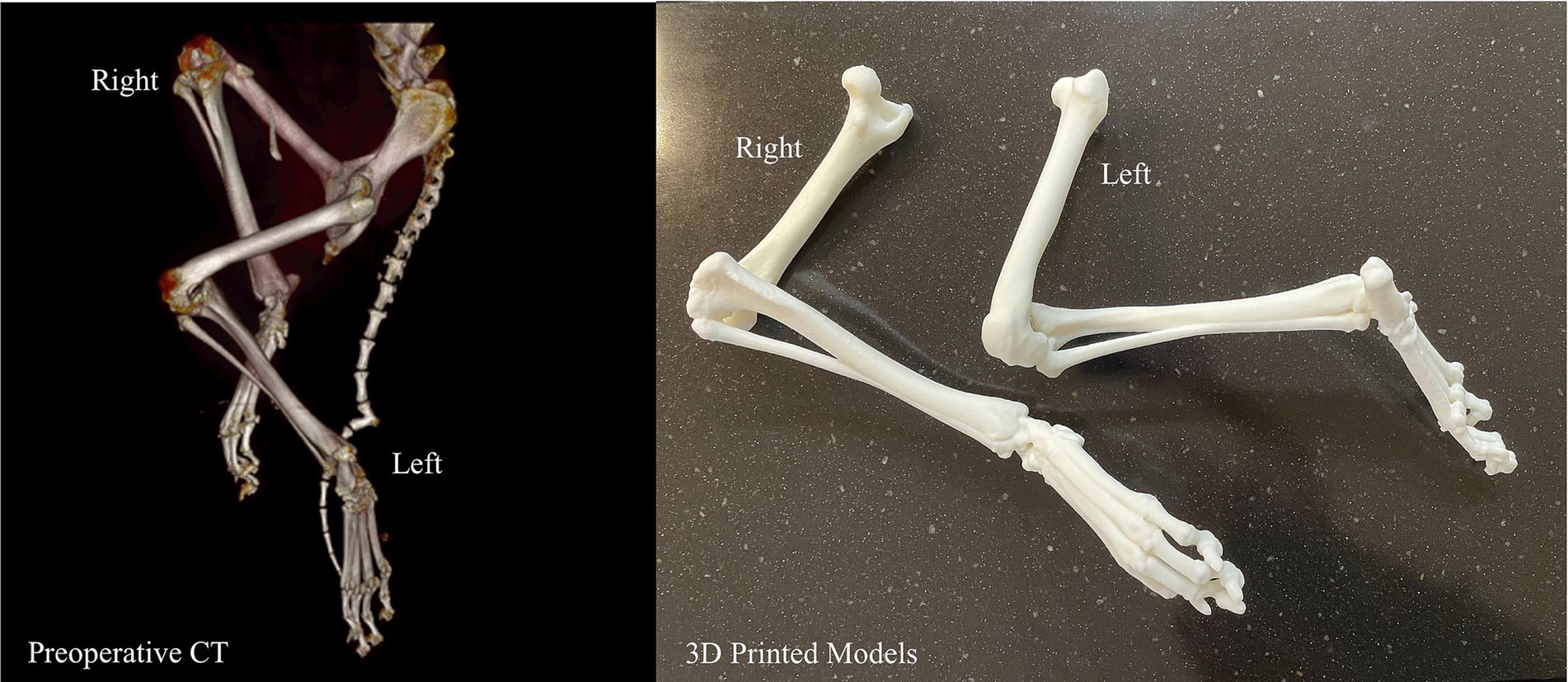
A summary of all canines included in this study can be found in Table 1.
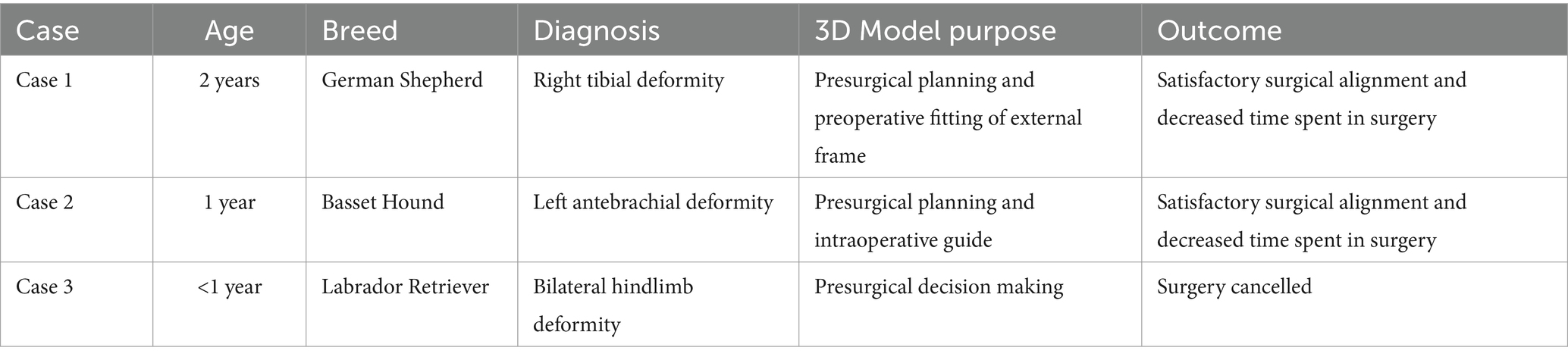
This case series of three orthopedic applications of 3D printing in veterinary medicine highlights the potential benefits of utilizing this technology, as well as providing specific examples of instances where it can effectively be used. Specifically, 3D printing in veterinary medicine can be used to preoperatively prepare external frames for complex orthopedic injuries, create detailed anatomic models for osteotomies as well as surgical guides to steer pin placement for adequate realignment, and provide an avenue whereby operative judgment can be exercised ahead of time, possibly preventing unnecessary surgery. These models assisted veterinary surgeons in optimizing their surgeries through enhanced anatomic understanding, better preparation, and confidence in their treatment plans.
Notably, despite optimal alignment and reduced surgery time, Case 1 resulted in a non-union at the osteotomy site (treated with an external fixator without subsequent plating). The canine remained clinically stable and pain-free at 12 months, indicating a functional outcome even without complete bony union. Similar to our results in Case 2, a previous study by Jeong et al. planned a corrective osteotomy for treatment of a medial patellar luxation in a canine using a 3D-printed bone model for pre-contouring and demonstrated improvement of limb function without relaxation of the patella through increased surgical accuracy (10). In Case 3, the anatomic models aided in decision-making to avoid a likely poor surgical outcome. Our cases add to the existing literature that supports the notion that 3D printed models can reduce OR time, decrease time under anesthesia, and avoid potential complications from improvising during surgery.
The veterinary surgeons in our unique cases remarked on how the printed physical models allowed them to examine the anatomy from different angles in order to create a treatment plan that was optimized for each specific canine. Though this case series is small, in these specific instances, the 3D models decreased operation time and, in some cases, allowed the veterinary surgeons to have a tactile appreciation during the surgery. Surgery requires an appreciation for haptic feedback, which allows opportunity for the insertion of 3D printed models into the presurgical and intraoperative process. Previous 3D printing case series have consistently supported the use of anatomic models in determining the margins of a mass, both before and during surgery, which assists with successful resection with proper margins (1, 2, 11). This can be especially helpful in veterinary medicine where numerous species of animals, as well as breeds within those species with varying anatomic composition, exist. With this variety, anatomic landmarks during surgeries may largely differ, and can require intimate understanding of spatiality.
An important consideration in orthopedic-oriented medical 3D printing is that imaging parameters are commonly limited by slice thickness. Currently, most of the diagnostic imaging parameters are not optimized for 3D printing. The thicker the slices, the less detailed the models. Artifacts, especially scatter artifacts related to existing metallic hardware, can be a barrier in orthopedic care where they are more commonplace. Even though metal artifact reduction algorithms can improve image quality, it can also sometimes generate new artifacts. Thus, it is important to review the images with a radiologist or imaging specialist in multiplanar reformats to avoid misinterpreting newly generated artifacts as true anatomy or pathology. Additionally, the specific need for the model determines which imaging modalities and parameters are most appropriate. An imaging specialist, such as a radiologist, is uniquely qualified to assess parameter appropriateness.
Our study was limited by being a three-case series, which makes these results difficult to extrapolate. As a case series without a control group, we cannot definitively quantify the advantages. Rather, our report is intended only to demonstrate the feasibility and potential benefits of this technology. The outcomes observed, while promising, should be interpreted as illustrative examples that will require validation in larger studies. Current general limitations to 3D printing in veterinary surgery include cost and necessary printing time and post-processing. Segmentation itself is completed using software that can range in cost from free to thousands of dollars per year for one license. When used for medical purposes, it is encouraged to utilize software that is specialized for use with advanced imaging for the purpose of optimizing image data and achieving accuracy.
Furthermore, desktop 3D printers are becoming increasingly accessible and affordable. These desktop 3D printers are recommended for simple anatomic models mainly due to these printers typically being single-nozzle with a few color options available and less capability overall. Industrial grade 3D printers cost a minimum of a few thousand dollars and range up to hundreds of thousands of dollars. These printers tend to have more material options, color varieties, and accurate printing specifications, often down to microns. However, the more accuracy a model requires, the thinner the 3D printed layers will be, which will increase the amount of time needed for printing algebraically. Once printed, these models at a minimum usually require separation of the 3D object from its 3D printed support structures, as well as the possibility of additional sanding, painting, magnet insertion, etc. These limitations are being addressed by newer 3D printers, but still remain a very present constraint for accessibility and usage.
Future applications of 3D printing in orthopedic veterinary surgery cases should quantify data by assessing data such as blood loss, OR time, time under anesthesia, and complication rate against a matched control group of canines where 3D printed models are not utilized. Additional information can be obtained from studies specifically studying anatomic models and usage in presurgical planning or surgical cutting guides and their possible optimization of the surgical process. These two usage scenarios often go hand in hand. Implementation of this technology in veterinary medicine should seek to develop case series with larger study populations so that both internal and external validity can be assessed. Furthermore, a wide variety of usages in diverse animal populations can be investigated to continue finding productive applications of additive manufacturing. Supplemental research can also aim to explore other burgeoning technologies that are related, such as virtual reality, augmented reality, and photogrammetry.
In conclusion, the integration of 3D printing technology into veterinary orthopedic surgeries has the potential to reshape traditional treatment paradigms and can possibly elevate the standard of care for animal patients. The multifaceted applications explored in this paper underscore the substantial impact 3D printing can have on presurgical planning, intraoperative precision, and postoperative assessment. The customization of external frames, surgical guides, and implants through 3D printing stands out as a hallmark advancement, allowing veterinary surgeons to tailor interventions to the unique anatomical nuances of each patient. This level of personalization not only elevates the precision of surgeries, but also contributes to postoperative recoveries that align more closely with the individual needs of the animal.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethical approval was not required for the studies involving animals in accordance with the local legislation and institutional requirements because owner consent was obtained and surgeries were performed by veterinarian at an outside academic institution. Written informed consent was obtained from the owners for the participation of their animals in this study.
CT: Data curation, Investigation, Methodology, Software, Visualization, Writing – original draft, Writing – review & editing. PA: Conceptualization, Data curation, Investigation, Methodology, Supervision, Writing – review & editing. DN: Conceptualization, Investigation, Methodology, Supervision, Visualization, Writing – review & editing. YHH: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Software, Supervision, Visualization, Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Altwal, J, Wilson, CH, and Griffon, DJ. Applications of 3-DIMENSIONAL printing in small-animal surgery: a review of current practices. Vet Surg. (2022) 51:34–51. doi: 10.1111/vsu.13739
2. Hespel, A, Wilhite, R, and Hudson, J. Invited review-applications for 3d printers in veterinary medicine. Vet Radiol Ultrasound. (2014) 55:347–58. doi: 10.1111/vru.12176
3. De Armond, CC, Lewis, DD, Kim, SE, and Biedrzycki, AH. Accuracy of virtual surgical planning and custom three-dimensionally printed osteotomy and reduction guides for acute uni- and biapical correction of antebrachial deformities in dogs. JAVMA. (2022) 260:1–9. doi: 10.2460/javma.21.09.0419
4. Huang, YH, Lee, B, Chuy, JA, and Goldschmidt, SL. 3D printing for surgical planning of canine oral and maxillofacial surgeries. 3D Print Med. (2022) 8:17. doi: 10.1186/s41205-022-00142-y
5. Gorham, DMQ, and Khan, MJ. Thinking outside of the box: the potential of 3D printing in veterinary medicine. J Vet Sci Technol. (2016) 7:360. doi: 10.4172/2157-7579.1000360
6. Suñol, A, Aige, V, Morales, C, López-Beltran, M, Feliu-Pascual, AL, and Puig, J. Use of three-dimensional printing models for veterinary medical education: impact on learning how to identify canine vertebral fractures. J Vet Med Educ. (2019) 46:523–32. doi: 10.3138/jvme.0817-109r
7. Leonardi, L, Marsili, R, Bellezza, E, Angeli, G, Emiliani, C, Ricci, A, et al. A pilot study on the use of 3D printers in veterinary medicine. Braz J Vet Pathol. (2021) 14:159–64. doi: 10.24070/bjvp.1983-0246.v14i3p159-164
8. Klasen, JRS, Thatcher, GP, Bleedorn, JA, and Soukup, JW. Virtual surgical planning and 3D printing: methodology and applications in veterinary oromaxillofacial surgery. Front Vet Sci. (2022) 9:971318. doi: 10.3389/fvets.2022.971318
9. Dundie, A, Hayes, G, Scrivani, P, Campoy, L, Fletcher, D, Ash, K, et al. Use of 3D printer technology to facilitate surgical correction of a complex vascular anomaly with esophageal entrapment in a dog. J Vet Cardiol. (2017) 19:196–204. doi: 10.1016/j.jvc.2016.10.003
10. Jeong, B, Jung, J, Park, J, Jeong, SM, and Lee, H. 3D-printing bone model for surgical planning of corrective osteotomy for treatment of medial patellar luxation in a dog. J Vet Clin. (2016) 33:385–8. doi: 10.17555/jvc.2016.12.33.6.385
11. Memarian, P, Pishavar, E, Zanotti, F, Trentini, M, Camponogara, F, Soliani, E, et al. Active materials for 3D printing in small animals: current modalities and future directions for orthopedic applications. IJMS. (2022) 23:1045. doi: 10.3390/ijms23031045
12. Kamishina, H, Sugawara, T, Nakata, K, Nishida, H, Yada, N, Fujioka, T, et al. Clinical application of 3D printing technology to the surgical treatment of atlantoaxial subluxation in small breed dogs. PLoS One. (2019) 14:e0216445. doi: 10.1371/journal.pone.0216445
Keywords: 3D printing, orthopedics, additive manufacturing, intraoperative guide, presurgical planning, anatomic modeling, canine surgery
Citation: Thomas C, Amsellem P, Nascene D and Huang Y-H (2025) Orthopedic applications of 3D printing in canine veterinary medicine. Front. Vet. Sci. 12:1582720. doi: 10.3389/fvets.2025.1582720
Edited by:
Nedzad Hadziomerovic, University of Sarajevo, Bosnia and Herzegovina
Reviewed by:
Sokol Duro, Agricultural University of Tirana, Albania
Alan Maksimovic, University of Sarajevo, Bosnia and Herzegovina
Copyright © 2025 Thomas, Amsellem, Nascene and Huang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yu-Hui Huang, huan2098@umn.edu
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
________________________________________________________________________________________________________________________________
AVMA I News
Monoclonal antibodies show promise as canine parvovirus treatment
Therapy marks a shift away from treating symptoms to targeting the virus
AVMA guidelines for the use of telehealth in veterinary practice
IMPLEMENTING CONNECTED CARE
https://www.avma.org/sites/default/files/2021-01/AVMA-Veterinary-Telehealth-Guidelines.pdf
Feds approve drugs treating canine osteosarcoma, ornamental fish parasites
https://www.avma.org/news/feds-approve-drugs-treating-canine-osteosarcoma-ornamental-fish-parasites
